Abstract
This study was designed to determine the quality of life, diagnostic, and illness-related experiences of patients who self-report a diagnosis with Lyme disease (LD) and/or who are experiencing chronic illness in Texas, a state considered non-endemic for tick-borne illness. This exploratory study found that self-reported LD respondents have multisystem health problems that result in very poor quality of life. Lyme disease respondents experience multiple and severe symptoms, particularly flu-like illness, extreme fatigue, back and neck pain, and anxiety and depression. These symptoms were present at similar levels among all LD respondents, whether their diagnosis was clinical or serological. For all LD respondents, this study points to quality of life experiences that are powerfully negative. Practitioners and disease surveillance experts may consider LD when multisystem symptoms are severe, other etiologies are ruled out, and quality of life is threatened.
Introduction
The aim of this study was to capture the experiences of Lyme disease (LD) patients regarding symptoms, diagnosis, and quality of life and compare experiences by diagnostic indicators. The complexity of LD is presented through nonspecific symptoms that mimic many other illnesses. Controversies arise in diagnosis when the early stages of LD are missed and disease progresses to later stages, which include multisystem involvement that often leads to severe and chronic symptoms. Stricker and Fesler offer a working definition of chronic Lyme disease (CLD): “CLD is a multisystem illness with diverse musculoskeletal, neuropsychiatric, and/or cardiovascular manifestations that result from ongoing infection with pathogenic members of the Borrelia spirochete complex often associated with other tick-borne disease (TBD) pathogens. To qualify for the diagnosis of CLD, patients must have Lyme-compatible symptoms and signs that are either consistently or variably present for six or more months” (1).
The Centers for Disease Control (CDC) reports 329,000 cases of LD annually in the United States, but the actual number may be considerably higher (4 –6). Lyme disease, like other TBD, such as ehrlichiosis, anaplasmosis, babesiosis, and tularemia, is zoonotic in origin. Lyme disease is spread by the pathogen, Borrelia burgdorferi (3). Causing frustration for patients, LD does not always produce a bull’s eye rash; other LD rashes may not be classic bull’s eye in appearance, and the numerous nonspecific symptoms can mimic other diseases (7). Indeed, many LD patients cannot recall a tick bite and among those surveyed in a study by Aucott et al, “a mean of 20.5% of participants was able to correctly identify the four nonclassic erythema migrans” (7)(p.1).
With testing protocols in debate, diagnosis of LD becomes more elusive and misdiagnosis can lead to symptoms associated with later stages of the disease. Lyme disease mimics symptoms found in other illnesses, such as multiple sclerosis, demyelinating diseases, and rheumatoid arthritis (8). False negative tests are recognized concerns, and the serological testing required by the CDC is often found to be subjective. In a recent study, Lantos et al found that “fewer than 20% of positive LD tests are obtained from patients with clinically likely LD” (9). The CDC recently revised their testing guidelines and offered new protocols beginning August, 2019 (4).
Without diagnosis, late-stage disseminated LD becomes problematic for patients, medical providers, insurance companies, and society. The economic burden of LD is considered significant (10). Adrion et al also found “total direct medical costs attributable to LD and (posttreatment Lyme disease syndrome [PTLDS]) could be somewhere between $712 million and $1.3 billion each year” (10). Thus, understanding LD patient experience, symptoms, and quality of life indicators is necessary to improve outcomes for LD patients and ultimately cost savings. Furthering this knowledge can also assist the medical community in understanding the role of chronic symptoms in a patient’s life. In addition to the basic diagnostic challenges of LD, the conflation of psychiatric presentation with vague and multisystem symptoms may further complicate a diagnosis. Studies on chronic illness point to the need to address the patient–physician dilemma, where “chronic” may be interpreted as faking or inflating a diagnosis (11).
Quality of life presents yet another elusive diagnostic and treatment challenge for those with LD. Generally, quality of life is shown to be poor for those with multiple chronic diseases (ie, comorbidities), including psychological distress that is bidirectional (12). For LD patients, the ability to live “normally” appears to be especially poor in comparison to other chronic illnesses (13,14). In a large national study in the United States, half of the LD patients required up to 10 years for a diagnosis; traveling over 50 miles for treatment; and seeing more than seven doctors before receiving a diagnosis (13). In another recent study, Johnson et al report, “Compared to the general population and patients with other chronic disease…, patients with CLD reported significantly lower health quality status, more bad mental and physical health days, a significant symptom disease burden, and greater activity limitations” (14)(p.1).
Noting the extremely poor quality of life reported by those with LD in comparison with other chronically ill patients, health care providers may have another tool in diagnosing LD when other testing fails, that is, inquiring about the patient’s quality of life, perhaps on a scale and over time or the life course.
Method
A survey methodology was employed to gather anonymous responses to questions regarding experiences and exposure to LD. The questionnaire was administered to members of a Facebook educational campaign on LD in Texas, exploring self-reported experiences with diagnostic information, quality of life, and treatment. The survey was designed to reach participants who may have been diagnosed with LD; presumably, the reason for their visit to the site originally. The objective was to describe quality of life and symptoms by LD diagnostic type. Adult participants were provided the option of completing the survey on behalf of a minor family member.
Relatively few patient surveys, specifically for tick-borne illness are available in the literature. This exploratory study is a first step in understanding the patient experience with an LD diagnosis or suspected LD. University of Texas at Dallas Institutional Review Board approval, under minimal review criteria, was obtained prior to the survey (1). All answers were completely anonymous. No personal identification data were collected.
For 1 year in 2018, a grant funded Facebook site titled “Texans & Ticks” provided prevention and scientific information to anyone who followed the site. The Facebook page was not an advocacy or treatment-related site. The site attracted many Texas visitors who had experiences with LD. The survey was posted on the site from May 30, 2018, until December 1, 2018, using an anonymous link. Visitors to the site were not required to take the survey, nor did they expect to take the survey for anything in return, for example, membership to a group. Rather, the Facebook page was open to the public. The consent and ethics statement included information about the researcher conducting the study.
The Texans & Ticks site garnered considerable attention, resulting in over 50,000 unique visitors to the site. Posts regarding information about tick species, prevention information, such as tick testing, and disease surveillance were provided weekly. The survey link resulted in the completion of 111 surveys, with 95 of the 111 reporting a diagnosis with LD.
The survey data were collected from the social media site using Qualtrics, an online survey platform. The data were available via a password protected, university-secured site. Data were analyzed for common patterns and total responses, as well as patterns among patient symptoms, diagnostic information, treatment, and quality of life indicators. For symptoms, LD-diagnosed respondents were compared by diagnostic type. Clinical and serological diagnostic respondent categories were compared by quality of life indicators.
Results
A total of 95 respondents reported an LD diagnosis and are included in Table 1. Table 1 also presents diagnostic information regarding respondents who report they have LD. Options include clinical diagnosis based on symptoms or serological tests, specifically the Western Blot or IGeneX. IGeneX is a specialty laboratory, which typically attracts patients who are suspected of having LD. The respondents with the lowest diagnostic reports fell under “I don’t know” and “CDC positive.” Almost half of the respondents were clinically diagnosed with LD by a medical provider through symptomatic presentation and eliminating other etiologies, rather than serological testing, while 23% met CDC-positive criteria. Respondents were able to select more than 1 category. Sixteen percent did not know how they were diagnosed and others report Western blot results that do not confirm a diagnosis (ie, not CDC positive).
. Participant and Diagnostic Information.

Abbreviation: LD, Lyme disease.
Table 1 shows that over 80% who responded were between the ages of 18 and 60 years old. One-third was not working. Figure 1 presents the type of medical professional who diagnosed the respondent or respondent’s minor family member. Even though LD symptoms include psychiatric and arthritic manifestations, not 1 respondent in the present survey was diagnosed by a psychiatrist and only 1% was diagnosed by a rheumatologist. On the other hand, more than half were diagnosed by a LD specialist.
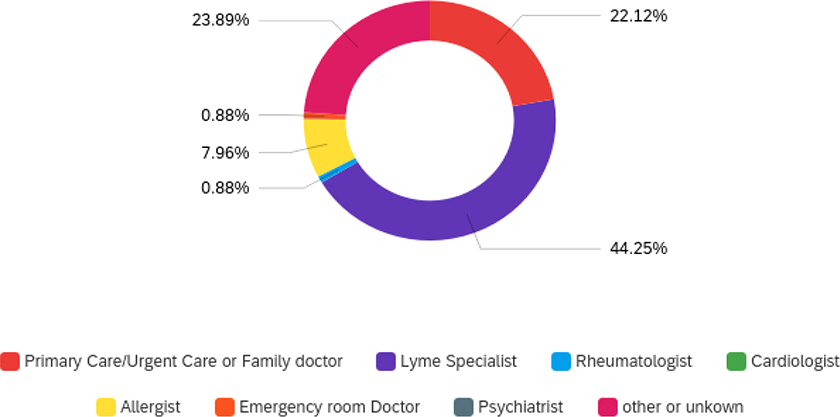
Percentage of LD self-reported respondents diagnosed by medical profession type. LD indicates Lyme disease.
Symptom questions were derived from LD literature and official sources such as the CDC. Respondents were allowed to check “all that apply” regarding their symptoms and quality of life. Respondents report LD symptoms commonly found in the literature related to later stages of the disease. For LD respondents, extreme fatigue, headaches, neck and back pain, brain fog, and depression and anxiety were the most frequently selected symptoms. The Anxiety and Depression Association of America states that over 18% of the US population has depression or anxiety (17), thus indicating a considerably higher rate for LD respondents in the present study.
Figure 2 offers self-reported respondent symptoms by diagnostic type to determine whether CDC positive cases presented differently than those reporting clinical or other serological findings. Lyme disease respondents with some positive Western blot bands or CDC positive are highest for headaches (95%). The CDC positive cases generally reflect overall patterns to other diagnostic types, except for IGeneX, with respondents reporting less severe symptoms for rash and headaches, but slightly higher for depression or anxiety. Overall, flu-like illness, extreme or unusual fatigue, neck and back pain, and brain fog stand out among all diagnostic types as consistently severe.

Percent of self-reported LD respondents by symptoms and diagnostic type. LD indicates Lyme disease.
In addition to symptoms, quality of life was measured through a history of lasting symptoms such as poor mental health or loss of friends, as noted in Figure 3. Lyme disease respondents report similar quality of life indicators across both clinical and CDC diagnoses. In both clinical and CDC positive cases, multiple symptoms with extreme fatigue were the highest reported indicators of poor quality of life. Poor physical and mental health and loss of friends were also important findings at over 60% responding. Those who were CDC positive report admission to the hospital and homecare visits more frequently than those with clinical diagnoses. Those with official diagnoses may have access to hospital admissions and homecare as a result of their diagnostic type, and future studies could investigate these differences further. In sum, both LD diagnostic types suggest a very poor quality of life with a complex web of health problems.

Quality of life over the last year by diagnosis type.
Discussion
The results covered a survey of patient experiences, including diagnostic, symptoms, and quality of life. In sum, findings suggest LD self-reported diagnosed respondents have lingering and challenging symptoms that severely affect their quality of life. Similar to LD scholarly research regarding late-stage disseminated LD, CLD, or PTLDS, survey respondents in the present study also demonstrated multisystem issues, with extreme fatigue as the most common poor quality of life indicator. Findings in this study confirm other patient surveys suggestive of extreme fatigue, neck and back pain, and overall quality of life as key factors in clinical presentations.
A few key findings in this exploratory study indicate the need for further research. Respondents were able to select the way in which they were diagnosed with LD, but few were officially “CDC positive.” However, almost all the respondents in this study exhibited classic LD symptoms (1), including a wide range of overlapping symptoms that appear to be lingering or chronic in nature. This finding is remarkable given the small number of CDC positive reports in Texas. However, a 2003 study reports “Unexpectedly we have found large numbers of chronically ill Borrelia burgdorferi PCR and seropositive patients in Houston, Texas, a zoonotically ‘non-endemic’ area” (2), pointing to possible underreporting or lack of testing in states generally considered non-endemic. Continued research is needed to address the decades-old mismatch between CDC-positive serological reports and scholarly findings. As with known LD patient reports, respondents’ quality of life in this study is poor, with symptoms that are highly disruptive of daily living. Finally, most of the LD respondents report diagnosis by a physician with LD training, and even though a high number of LD respondents report psychiatric symptoms, a known common feature of LD and co-infections, not one was diagnosed by a psychiatrist.
Conclusion
Findings indicate that LD symptoms are similar and generally equally severe across all diagnostic LD groups. Those diagnosed clinically match those with CDC-positive diagnoses in symptoms and level of severity. The CDC-positive respondents are more likely to have been admitted to a hospital or have home care, a finding that merits further attention.
The strengths of this exploratory study indicate a distinct need to identify and assist patients who do not meet CDC LD serological testing criteria, but who report poor quality of life and multisystem symptoms. Rather than questioning the legitimacy of their symptoms, physicians may need to render a new diagnosis, eliminate other etiologies, and/or diagnose clinically. For all LD respondents, this study highlights the quality of life experiences that those with either a clinical or a serological LD diagnosis face, and those experiences are powerfully negative.
In practical terms, this study hints at the need for further clinician training, better testing protocols, and involvement of medical providers outside of their specialty areas. Lyme disease psychiatric assessment or screening tools that include LD neuropsychiatric indicators could be implemented. If patients presenting with neuropsychiatric symptoms who also report migrating joint pain, brain fog, extreme fatigue, and a known tick-bite or outdoor behaviors such as hunting, camp attendance, sports, or gardening are asked about these indicators, a referral to a specialist may be a commonsense approach.
The practical implications of additional screening tools are simple, cost effective, and easy. Public policy will most likely not acquiesce given the opposing need to practice good stewardship with antibiotic resistance. Like all chronic and challenging illnesses, a one-size-fits-all treatment approach is elusive. Also elusive is the full understanding by the medical community and policy makers regarding the possibility of LD and the extremely poor quality of life faced by those reporting an LD diagnosis. Community health providers who serve those at risk may consider improved prevention and education about tick-borne illness.
Supplemental Material
Supplemental Material, Consent_Form_Appendix - The Elusive Understanding of Lyme Disease in Non-Endemic Geographic Areas: An Exploratory Survey of Patients With Chronic Symptoms in Texas
Supplemental Material, Consent_Form_Appendix for The Elusive Understanding of Lyme Disease in Non-Endemic Geographic Areas: An Exploratory Survey of Patients With Chronic Symptoms in Texas by Sarah P Maxwell in Journal of Patient Experience
Supplemental Material
Supplemental Material, Cover_Letter_JPatientExperience - The Elusive Understanding of Lyme Disease in Non-Endemic Geographic Areas: An Exploratory Survey of Patients With Chronic Symptoms in Texas
Supplemental Material, Cover_Letter_JPatientExperience for The Elusive Understanding of Lyme Disease in Non-Endemic Geographic Areas: An Exploratory Survey of Patients With Chronic Symptoms in Texas by Sarah P Maxwell in Journal of Patient Experience
Supplemental Material
Supplemental Material, Respoonse_to_Reviewers_April_21_2020 - The Elusive Understanding of Lyme Disease in Non-Endemic Geographic Areas: An Exploratory Survey of Patients With Chronic Symptoms in Texas
Supplemental Material, Respoonse_to_Reviewers_April_21_2020 for The Elusive Understanding of Lyme Disease in Non-Endemic Geographic Areas: An Exploratory Survey of Patients With Chronic Symptoms in Texas by Sarah P Maxwell in Journal of Patient Experience
Footnotes
Authors’ Note
Institutional Review Board approval was obtained prior to the survey. All answers are completely anonymous and patients consented to voluntarily take the survey. No personal identification data were collected.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
