Abstract
This study examined the feasibility and impact of implementing routine depression screening in pharmacy settings through a 4-phase approach. A systematic literature review identified evidence supporting pharmacist-led screenings as effective and acceptable, with studies reporting improved detection rates and positive patient outcomes. Legislative analysis revealed strong international frameworks, particularly in the United States and United Kingdom, emphasizing pharmacists’ role in mental health care. A situational analysis in Bulgaria highlighted a significant unmet need, with high prevalence of depressive symptoms among chronically ill patients and limited access to mental health specialists. These findings underscore the potential of community pharmacies to bridge care gaps. Based on the evidence, key recommendations include developing pharmacist training programs, advocating for supportive legislation and funding, launching public awareness campaigns to reduce stigma, and establishing collaborative referral networks with other healthcare providers. Implementing these measures can facilitate early detection, timely intervention, and improved public health outcomes by leveraging the accessibility and expertise of pharmacists in mental healthcare.
Keywords
Introduction
Depression is a common mental health disorder affecting 5% of the population globally that causes a significant burden to the affected individuals and society. 1 In most countries, 8% to 12% of the population develops clinically significant depression at some time in their lives, making it the third most common reason for consultation in general practice and a leading cause of disability. Only 5% of patients identified with depression are referred to psychiatrists, and only 2% receive inpatient care, which is one of the leading reasons 1 in 8 diagnosed patients develop chronic depression. 2 Data from the European Health Interview System found that 3 out of every 100 people (3%) in Bulgaria are living with chronic depression. 2
Depression often accompanies patients with chronic illnesses. Among specific populations, such as patients with recent acute myocardial infarction, the prevalence is as high as 33%, and among patients with cancer, the prevalence can be as high as 47%. 3 World Health Organization predicts that by 2030, depression will become the leading cause of complicating the health state of patients with chronic and comorbid conditions. Gender differences play a significant role in the epidemiology and manifestation of depression. Globally, women are nearly twice as likely to experience major depressive episodes as men, and perinatal depression affects up to one in 5 mothers, underscoring the importance of early detection and community-based support systems for this population. 4 In contrast, suicide mortality remains substantially higher among men, with more than half of global suicides occurring before the age of 50, highlighting an urgent need for accessible mental health interventions targeting working-age male populations (World Health Organization, 2023). 5
The total economic burden of depression is estimated to be $210.5 billion per year, an increase of 21.5% or $37.3 billion per year from the last decade. 6 Nearly half of the total cost is direct medical costs of treating depression (45%-47%). Indirect economic costs arise from premature mortality due to the disease and from reduced productive capacity of individuals, accounting for about 50% of total costs. 7 Effective screening programs are essential for early diagnosis as well as for preventing depression and its comorbidities. Pharmacists, as health professionals in direct daily contact with patients, play a crucial role in the healthcare system. Their accessibility allows them to provide essential services and actively participate in early screening for various health conditions, including depression. Early screening is a simple, cost-effective method for identifying depression, facilitating timely intervention, and improving patient outcomes. This study aims to provide actionable insights for implementing early depression screening as a routine pharmacy practice. The second aim is to identify the legal, systemic, and contextual enablers and barriers to implementing pharmacist-led depression screening, with particular attention to the Bulgarian setting and transferable international models.
Materials and Methods
This study involves a multiphase approach structured into 4 distinct phases. Phase 1 is a systematic literature review (SLR) that aims to gather and synthesize existing research on the feasibility, effectiveness, and outcomes of early depression screening conducted by pharmacists in community pharmacies. Phase 2 aims to analyze the legislative and regulatory frameworks that support the implementation of depression screening programs in pharmacies across different countries. Phase 3 situational Analysis and implementation Strategy in Bulgaria aims to understand the current landscape, opportunities, and challenges for implementing early depression screening in Bulgarian pharmacies, providing a detailed understanding of the local context. Phase 4 integrates findings into coherent recommendations aimed at facilitating the adoption and successful implementation of early depression screening programs in pharmacies.
Phase 1: Systematic Literature Review
Data Sources and Search Strategy
This SLR followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. The methodological quality of all included studies was assessed using the Joanna Briggs Institute (JBI) meta-aggregative approach. A comprehensive systematic review of abstracts and free full texts investigating early screening of depression in community pharmacies was performed based on: (1) identification of a research question; (2) identification of inclusion and exclusion criteria; (3) data extraction; (4) reporting results; and (5) discussion and interpretation of findings. The primary search was performed in the PubMed database, using the following keywords and Boolean operators “depression” OR “depressive disorder” AND “pharmacist” OR “pharmacy” OR “community pharmacy” AND “early diagnosis” OR “early screening” OR “screening.” Searches were carried out between June 1st, 2024, and January 31st, 2025. The search for available studies covered the period from 2006 to 2025 reflecting the emergence of the value-based healthcare concept. To enhance comprehensiveness and minimize the risk of publication bias, a supplemental search was conducted using the Elicit AI platform, which draws on the Semantic Scholar database. The guiding research question used in Elicit was “What is the effectiveness of routine depression screening by pharmacists in identifying undiagnosed depression among adult patients in community pharmacy settings?” The top 50 Elicit-generated results were screened, and 8 relevant articles were reviewed and included in the final synthesis. A flow diagram was applied to systematically present the number of studies identified, those meeting the criteria, excluded studies, and reasons for exclusion. The protocol was registered in Open Science Framework on July 15th, 2025.
Inclusion and Exclusion Criteria
Articles were included based on the following criteria: (1) studies in English; (2) no limitations to publication type; (3) full-text available or abstract containing sufficient data; (4) involved pharmacist-led depression screening in community pharmacy settings; (5) studies addressing the research questions and reporting data on depression screening. Studies were excluded if they did not: (1) report depression screening; (2) involve pharmacists in depression screening; (3) were conducted outside of pharmacy settings (eg, hospitals, clinics); and (4) provide accessible full text or adequate information in the abstract.
Eligibility Criteria
The search was conducted following the routine literature review methodologies, including PICO (Population, Intervention, Comparison, Outcomes):
- - - -
Methodological Quality Assessment
The JBI critical appraisal tool, used to assess the quality of the included SLRs, contains 10 questions with 4 rating choices (yes, no, unclear, not applicable) for each question (Supplementary Table 1).
Data Extraction and Synthesis
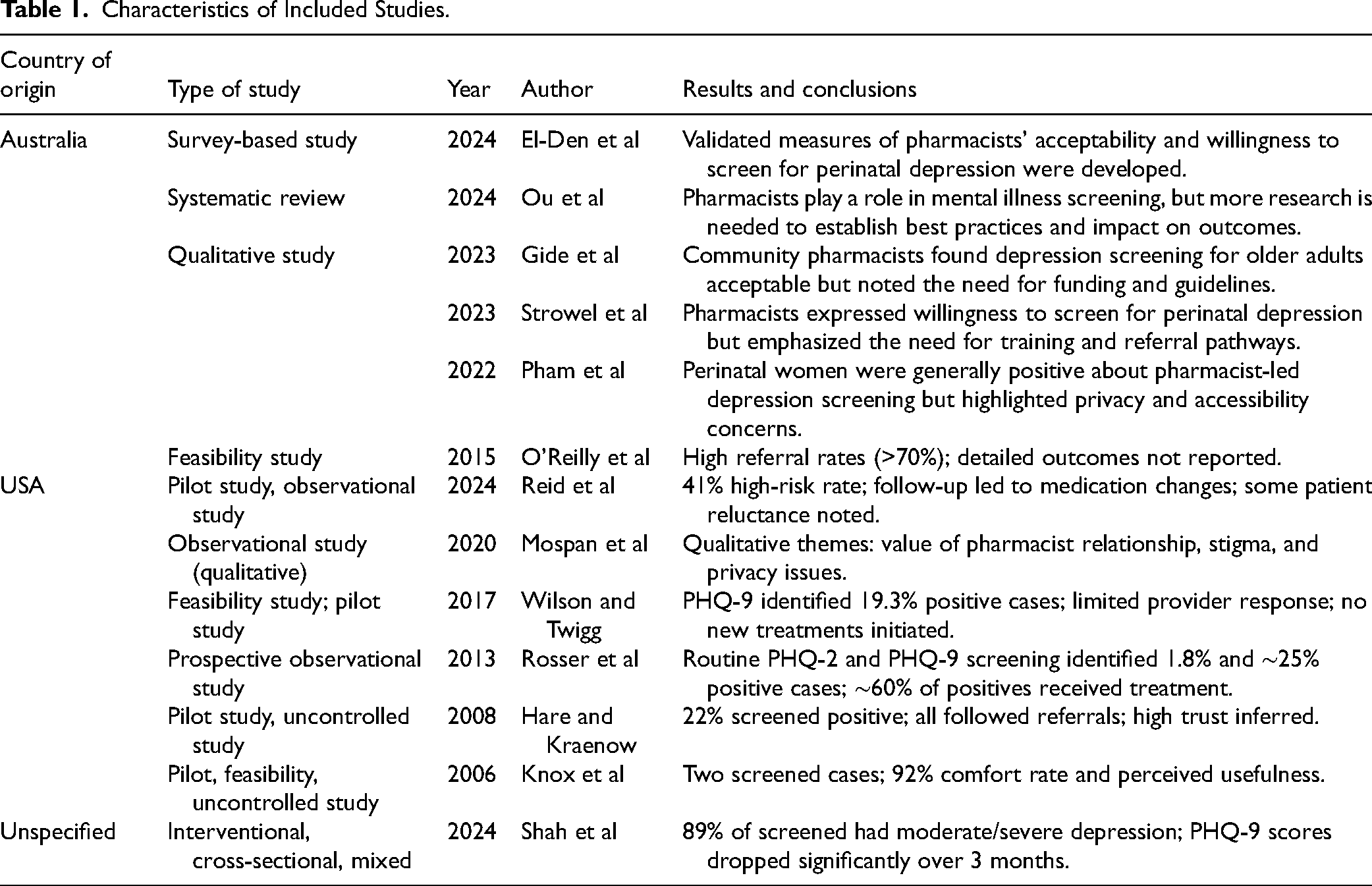
One investigator entered the study data into the extraction table, while a second one verified the information for accuracy and completeness. The following data were extracted: author, year, country of origin, type of study, results, and conclusions. Characteristics of the 13 included studies4,7–18 are presented in Table 1.
Characteristics of Included Studies.
Phase 2: Legislative Analysis
The analysis reviewed legislative documents, guidelines, and literature on early depression screening programs in pharmacy settings. Countries included the United States, United Kingdom, Australia, Canada, Netherlands, and Germany. Sources included health department websites, pharmacy associations, and legislative databases. Data extraction focused on frameworks, implementation processes, key stakeholders, and challenges (Table 2). The inclusion of these 6 countries was based on the presence of established or emerging national frameworks integrating pharmacists into mental health or preventive care services, as identified through preliminary scoping searches. Selection criteria included: (1) availability of documented legislation or national policy related to pharmacist-led health screenings; (2) recognition of pharmacists’ expanded clinical roles within primary care; and (3) relevance to transferable models for European healthcare systems. These countries were chosen to represent diverse yet well-documented approaches to implementing pharmacist-led screening programs across different regulatory and healthcare environments.
Legislative Analysis in the USA, UK, Australia, Canada, Germany, and the Netherlands.

Abbreviations: ACA, Patient Protection and Affordable Care Act; AHRQ, Agency for Healthcare Research and Quality; CPCF, Community Pharmacy Contractual Framework; CTFPHC, Canadian Task Force on Preventive Health Care; 5CPA, Fifth Community Pharmacy Agreement (Australia); GPhC, General Pharmaceutical Council (UK); IGZ, Dutch Health Care Inspectorate; GVWG, Gesundheitsversorgungsweiterentwicklungsgesetz (Health Care Development Act, Germany); KNMP, Royal Dutch Pharmacists Association; NHS, National Health Service (UK); NIVEL, Netherlands Institute for Health Services Research; NVL, National Disease Management Guidelines (Germany); PSA, Pharmaceutical Society of Australia; USPSTF, United States Preventive Services Task Force.
Phase 3: Situational Analysis and Implementation Strategy in Bulgaria
This phase assessed the Bulgarian context using demographic and epidemiological data, a 2020 survey of pharmacists and patients, and feasibility indicators for depression screening. A strategic implementation plan and SWOT analysis were developed based on expert opinions and patient feedback from a pilot study involving 119 chronically ill patients. 8 All participants in the pilot study provided informed consent prior to participation. Participant anonymity and data confidentiality were strictly maintained. Screening data were collected and stored in a de-identified format, accessible only to the research team. No personally identifiable information was disclosed, and participants were informed that screening results would not replace medical diagnosis.
Phase 4: Recommendations
Recommendations were based on evidence from previous phases, synthesizing findings on feasibility, legislative frameworks, and implementation strategies.
Results
Phase 1: Systematic Literature Review
Selection and Quality of Articles
The PubMed search yielded 40 records; after title and abstract screening, 34 were excluded. Full-text review of 6 articles excluded one for not involving pharmacist-led screening in a pharmacy setting, leaving 5 studies. A complementary Elicit search screened 50 records and identified 8 additional eligible studies. In total, 13 studies (5 PubMed, 8 Elicit) were included. Most studies demonstrated moderate to high methodological quality, with clear objectives and robust designs. Common limitations included incomplete reporting of demographics and variability in follow-up. Despite these, risk of bias was generally low, supporting the reliability of evidence for pharmacist-led depression screening.
Description of Included Studies
Geographic distribution: Six from Australia, 6 from the United States, and 1 unspecified (included as it fulfilled all predefined inclusion criteria and contributed valuable methodological data relevant to the review objectives). Study designs: Qualitative (n = 4), feasibility/pilot (n = 5), survey-based (n = 1), observational/mixed methods (n = 2), and one systematic review. Focus areas: Feasibility, acceptability, implementation barriers, and patient-pharmacist perspectives. Common barriers included lack of funding, time constraints, and privacy concerns. Screening tools: The PHQ-9 and PHQ-2 were most frequently used; some studies also applied GAD-7 for anxiety screening. The preference for PHQ-2 and PHQ-9 tools in pharmacy settings stems from their strong psychometric validity, ease of use, and minimal time burden. Both instruments are self-administered, require limited training for pharmacists, and are well-suited for brief patient interactions typical in community pharmacy practice. The PHQ-2 serves as an efficient initial screener to identify potential depressive symptoms, while the PHQ-9 allows for more detailed assessment and monitoring of symptom severity. These tools are validated across diverse populations and have demonstrated high sensitivity and specificity for major depressive disorder. Limitations include the reliance on self-reporting, which may lead to under- or overestimation of symptoms, and the need for appropriate referral pathways for patients with positive results. Key outcomes: Detection rates ranged from 1.8% to over 40%, with referral follow-up rates between 25% and 70%. One study (Shah et al, 2024) reported significant PHQ-9 score reductions over 3 months, indicating improved mental health outcomes.
A summary of study characteristics is provided in Table 1.
Phase 2: Legislative Analysis
United States
United States policies increasingly support pharmacist-led depression screening as part of efforts to improve mental health access. The Affordable Care Act (2010) mandates coverage of depression screening without cost-sharing, facilitating integration into care settings, including pharmacies. 19 The U.S. Preventive Services Task Force recommends routine adult screening, including for perinatal populations, influencing its adoption in primary care. 20 The Agency for Healthcare Research and Quality reviews confirm the effectiveness of standardized tools for detection, 20 and Healthy People 2030 sets goals to increase depression screening in primary care visits, emphasizing early detection as a national priority. 21
United Kingdom
The United Kingdom supports pharmacists’ involvement in mental health through several policies. The National Service Framework for Mental Health (1999) set quality standards for integrated mental healthcare. The National Health Service (NHS) Long Term Plan (2019) and Community Pharmacy Contractual Framework emphasize expanding community-based services and the pharmacist's role in early detection. 22 The General Pharmaceutical Council provides regulatory guidance on pharmacists’ responsibilities in delivering safe and effective mental health care, including screening.
Australia
Australia has integrated pharmacists into mental healthcare through national agreements and competency standards. The Fifth Community Pharmacy Agreement (2010) and subsequent agreements funded pharmacist training in mental health, 23 including programs such as Mental Health First Aid and the Pharmacy Guild's screening initiatives. The Pharmacists in General Practice (2017) program promotes collaborative care, enabling pharmacists to conduct screenings within primary care teams. 24 The 2019 National Competency Standards Framework defines required skills for delivering services such as mental health screening. 25
Canada
The Canadian Task Force on Preventive Health Care (2005) issued guidelines supporting depression screening by primary care providers. 26 Programs such as MedsCheck (Ontario, 2007) allow pharmacists to assess mental health during medication reviews, while provincial scope expansions (2012-2015) enabled pharmacists to deliver broader services, including screening. 27 Initiatives like Nova Scotia's Bloom Program integrate pharmacists into mental health teams to enhance access, early detection, and care coordination. 28
Germany
Germany's Gesundheitsversorgungsweiterentwicklungsgesetz 2021 (GVWG) expanded nonphysician roles in preventive care, enabling pharmacists to conduct mental health screenings. 29 The Apothekenbetriebsordnung regulates pharmacy operations and increasingly emphasizes preventive care, including mental health. 30 The National Disease Management Guidelines (Nationale VersorgungsLeitlinie, NVL) further support pharmacist involvement in early depression detection and management. 31
Netherlands
The Dutch Health Care Inspectorate (IGZ) and Royal Dutch Pharmacists Association (KNMP) issue guidelines supporting pharmacists’ involvement in preventive care, including mental health screening.32,33 Integrated care models position pharmacists alongside general practitioners, while the National Prevention Agreement promotes early detection of depression within primary care. 34
Comparative Analysis
Across all countries, pharmacists are integrated into primary care with roles in depression screening supported by regulatory guidelines and government initiatives. Common goals include improving early detection, public health outcomes, and care integration. Training and collaboration frameworks are widely established to enable effective referrals and follow-up (Table 3).
Legislative Analysis—Similarities and Differences.

Abbreviations: AHRQ, Agency for Healthcare Research and Quality; CPCF, Community Pharmacy Contractual Framework; NHS, National Health Service; GPhC, General Pharmaceutical Council; USPSTF, United States Preventive Services Task Force.
Scope of practice varies: Canada and Australia allow broader clinical roles, while Germany and the Netherlands rely on general preventive care policies. The United States and United Kingdom have more explicit mandates for screening through detailed legislative frameworks. The United Kingdom and Netherlands emphasize integrated care models, whereas the United States and Canada focus on pharmacist-led community-based services. Funding approaches differ—Australia and Canada provide targeted funding, while Germany relies on broader health reforms.
Shared challenges include provider engagement, patient privacy, time and resource constraints, and ensuring consistent implementation.
Phase 3: Situational Analysis and Implementation Strategy in Bulgaria
Introducing early depression screening in Bulgarian pharmacies requires careful planning, supported by situational and SWOT analyses. This approach identifies opportunities and challenges for integration into community pharmacy practice.
Situational Analysis
High prevalence of depression: A pilot study of 119 chronically ill patients found 64.9% had depressive symptoms, including 14% with severe depression, consistent with global trends. 35
Risk factors: Depression is more common among older adults, urban residents, and women. Over 50% of chronically ill patients reported mild to moderate symptoms.35,36
Underdiagnosis: 89% of affected patients had never consulted a psychiatrist, highlighting stigma, poor access to specialists, and screening gaps. 35
Limited mental health services: Mental health screenings by GPs are insufficient, leaving many patients untreated. Untreated depression is associated with higher pharmacotherapy costs. 35
No pharmacy-based screening: Current efforts rely on physicians, though evidence from other countries supports pharmacy integration as feasible and effective. 13
SWOT Analysis
Strengths
Pharmacies offer high accessibility and no appointment barriers, enabling broader depression screening coverage than primary care. Screening by trained pharmacists supports early detection, timely intervention, and improved outcomes, while reducing GP workload. This service leverages pharmacists’ underused expertise and creates opportunities for professional development.5,35
Weaknesses
Depression screening is not yet a standard pharmacy service in Bulgaria, potentially increasing workload without initial incentives. Accuracy limitations of screening tools and lack of pharmacist experience present challenges, requiring training and practice (Table 4). The service currently addresses only depression, which may seem fragmented, though it could pave the way for broader health screening programs. 5
SWOT Analysis for the Feasibility of Introducing an Early Depression Screening Program in Bulgarian Pharmacies.

Abbreviations: GPs, general practitioners; GPP, good pharmacy practice; SWOT, Strengths, Weaknesses, Opportunities, and Threats.
Opportunities
Depression screening offers pharmacies a first-mover advantage in Bulgaria, addressing an unmet need and expanding their role in preventive care. The service aligns with Good Pharmacy Practice regulations and potential legislative changes promoting health promotion activities. Implementing this innovation positions pharmacies as key players in public health and strengthens patient trust. 35
Threats
Pharmacists’ expanded roles may face skepticism from healthcare professionals and stakeholders accustomed to traditional pharmacy services. Social stigma around depression remains a barrier, potentially discouraging patients from screening participation despite assurances of confidentiality. 35
Implementation Strategy
Goals
The strategy focuses on improving pharmaceutical care through clear communication, accurate screening guidance, and quality patient service. Objectives include increasing service visibility, meeting and exceeding patient expectations, expanding the mental health care awareness, and supporting long-term service sustainability. 5
Target Population
Patients with chronic diseases have been specified as the target population, divided into 3 categories: primary target (chronic disease patients living in urban areas, over 60 years old, predominantly female), secondary target (chronic disease patients living in urban areas, over 60 years old, predominantly male), and tertiary target (all other chronic disease patients). 5
Communication Strategy
Awareness will be raised via brochures, professional events, online channels, and direct engagement with patients and healthcare providers. Endorsement by the Bulgarian Pharmaceutical Union will support credibility through education campaigns and public outreach.
Phase 4: Recommendations
Based on findings from the previous phases, the following recommendations are proposed:
Training—Equip pharmacists with skills in screening tools, symptom recognition, and referral; integrate mental health into pharmacy curricula. Legislation & Funding—Amend regulations to authorize pharmacist-led screening and secure sustainable funding, modeled on US/UK practices. Public Awareness—Educate communities, reduce stigma, and emphasize confidentiality and pharmacist support. Collaboration—Build referral networks and foster multidisciplinary care involving pharmacists. Monitoring—Track detection rates, referrals, and patient satisfaction to ensure continuous improvement.
Discussion
Interpretation
Pharmacist-led depression screening is both feasible and effective, as shown by the SLR, which reported high patient acceptance and successful identification of undiagnosed depression. Pharmacists’ accessibility positions them as trusted providers in mental health care. Legislative reviews highlighted strong policy support in countries such as the United States (Affordable Care Act) and the United Kingdom (NHS Long Term Plan). The Bulgarian situational analysis confirmed a significant unmet need, especially among chronically ill patients, while the pilot study demonstrated a high prevalence of depressive symptoms, underscoring the value of pharmacy-based screening services.
Comparison With Other Studies
The findings of this study align with previous research conducted in countries such as the United States and Australia, where pharmacists have been successfully integrated into mental health screening initiatives. For example, high acceptance rates of depression screenings conducted by pharmacists, as well as improvements in the early detection of mental health conditions are reported.4,13,15,16 However, unlike these countries, Bulgaria's legislative and healthcare system presents unique challenges, such as the absence of structured funding mechanisms and limited experience among pharmacists in conducting screenings. While the pilot study in Bulgaria reflects trends observed internationally, it also emphasizes the localized barriers that need to be addressed to ensure the successful implementation of such programs. 8
Limitations and Strengths
This study demonstrates the feasibility and potential impact of pharmacy-based depression screening but has several limitations. Findings may not be fully generalizable, as the analysis focuses on the Bulgarian healthcare and pharmacy context and draws on studies primarily from high-income countries. Bulgaria, an EU member state with an upper-middle-income and steadily growing economy, provides a valuable environment for piloting pharmacist-led innovations. The pilot study involved a smaller sample, limiting demographic diversity, yet it represents the first phase of a broader PhD research project that is currently expanding in scope and sample size. While large-scale pharmacy screening models are not yet established, the ongoing work aims to generate more robust, generalizable evidence.
Despite these contextual factors, the study offers transferable lessons for other countries seeking to integrate early depression screening into community pharmacy practice. Elements such as pharmacist accessibility, patient trust, and use of validated screening tools are widely transferable, whereas implementation speed, funding mechanisms, and integration pathways may vary according to national health policy and system structure.
Strengths include a multiphase approach combining SLR, legislative review, and situational analysis for a comprehensive perspective. Cross-country comparisons identify transferable best practices, while the Bulgarian pilot provides real-world insight. The SWOT analysis offers a structured basis for actionable recommendations. Overall, this study underscores the pharmacist's role as an accessible provider capable of improving mental health outcomes through early detection.
Implications
The findings of this study have several implications for practice and policy. Implementing pharmacist-led depression screening requires coordinated efforts between healthcare providers, policymakers, and professional organizations. Priorities include establishing structured training programs, introducing supportive legislation and funding mechanisms, and creating clear referral and follow-up pathways. Public education campaigns are also needed to reduce stigma and improve awareness of mental health services. Together, these measures can strengthen the role of community pharmacies in early detection and integrated mental health care.
Conclusion
This study demonstrates that community pharmacies represent an underused yet highly accessible setting for early depression screening. By combining evidence from international frameworks and local pilot data, the research provides a practical model for implementing pharmacist-led screening programs in Bulgaria and similar European contexts. The results highlight both feasibility and the need for continued expansion, including larger-scale implementation and outcome evaluation. Ongoing research under the associated PhD project will further validate and refine this model to support the sustainable integration of mental health services in pharmacy practice.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735261415769 - Supplemental material for Implementing Early Depression Screening as Routine Practice in Pharmacy Settings
Supplemental material, sj-docx-1-jpx-10.1177_23743735261415769 for Implementing Early Depression Screening as Routine Practice in Pharmacy Settings by Yoanna Vutova, Georgi Slavchev, Desislava Ignatova and Мaria Kamusheva in Journal of Patient Experience
Supplemental Material
sj-docx-2-jpx-10.1177_23743735261415769 - Supplemental material for Implementing Early Depression Screening as Routine Practice in Pharmacy Settings
Supplemental material, sj-docx-2-jpx-10.1177_23743735261415769 for Implementing Early Depression Screening as Routine Practice in Pharmacy Settings by Yoanna Vutova, Georgi Slavchev, Desislava Ignatova and Мaria Kamusheva in Journal of Patient Experience
Footnotes
Acknowledgments
The authors would like to thank all participating pharmacists and patients for their valuable insights and contributions to the pilot study phase of this research.
Ethical Approval and Informed Consent Statements
Ethical approval was not required for this study, as it did not involve any interventions or personal data beyond anonymized survey responses. Patient consent was not required for this study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is financed by the European Union NextGenerationEU, through the National Recovery and Resilience Plan of the Republic of Bulgaria, project № BG-RRP-2.004-0004-C01.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data supporting this study's findings are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
