Abstract
Play is crucial for childhood development and quality-of-life, but for children with medical complexity (CMC), availability of recreational programming is rare. A home healthcare organization in Toronto, Ontario collaborated with parents to co-design a novel 6-week playgroup that offered accessible, medically safe, and enjoyable play experiences for CMC. A mixed methods approach was undertaken to assess the early impact of the playgroup on participating CMC's quality-of-life functioning. The Pediatric Quality-of-Life Inventory was administered as a pre-post-measure followed by semi-structured interviews with parents. Sixteen parents corresponding to 18 registered CMC participated in the study. Early outcomes indicated an increase in overall quality-of-life functioning (27.4%
Introduction
Children with medical complexity (CMC) are a vulnerable health population, characterized by their medical fragility, usually with one or more complex, chronic health conditions, significant limitations with ambulation, and reliance on medical technology such as tracheostomies, leaving them physically dependent on others for activities of daily living. 1
This can include a wide range of children who have “congenital or acquired multisystem disease, a severe neurological condition with marked functional impairment, or patients with cancer/cancer survivors with ongoing disability in multiple areas.”
2
They require extensive healthcare support from multiple professional
Physical and sensory activities have empirically shown to improve sleep patterns, behaviors, and feelings of inclusion and self-esteem among children with special health needs,7–10 which suggests that specialized recreational programming may be an effective intervention to increase quality-of-life for CMC.
The PlayDate Program
To address programming gaps for CMC, a homecare organization in the Greater Toronto Area supporting this pediatric population, co-designed
To understand the program's early impact and feasibility
Methods
Design
A mixed methods approach informed this pilot study. A pre-post self-administered questionnaire was used to assess participants’ quality-of-life functioning. Brief post interviews with parents captured their perspectives and experiences. The study received institutional ethics approval from the University of Toronto.
Participants
Participants were recruited through convenience sampling; registered parent–child dyads from two
During the first week of each playgroup, the study team was onsite to answer questions and obtain informed consent from parents; parents were designated as substitute decision-makers for CMC when enrolling into the study and maintained throughout study activities. Of 22 registrants, 16 parents corresponding to 18 CMC were enrolled into the study; two parent–child dyads included a set of twins.
Data Collection
All data was collected from parent–child dyads over the course of each 6-week program by the first author, trained in qualitative research and social work. Program intake documents provided demographic and medical complexity details.
The PedsQL™ Inventory 4.0
11
was administered as a pre- and post-measure. The questionnaire is completed by a caregiver-proxy and consists of 23 statements which assess four domains: physical, emotional, social, and school functioning. Two composite scores are also available: a total score (all questionnaire items) and a psychosocial functioning score (emotional, social, and school functioning subscales). Parents rate their child's perceived quality-of-life by evaluating the frequency, in the previous 1 month, where each item have presented as problems ranging from never to almost always.
11
Completed baseline assessments were returned to researchers by the first week of
Brief post-program interviews with 16 parents were conducted via telephone. An interview guide facilitated conversations about quality-of-life and experience in the program. Interviews were approximately 15 minutes long and were audio-recorded with participant permission.
Data Analysis
Out of 18 potential pre- and post-questionnaires completed; 16 were returned to the study team and four were eliminated due to missing data, rendering 12 pre-post tests available for analysis. Raw scores were reverse scored, converted to a 0 to 100 scale, excluded if over half the responses were missing, and removed if multiple Likert options were selected. The psychosocial functioning subscale was the arithmetic mean of the emotional, social, and school functioning subscales. Changes were calculated as the difference between post-test and baseline scores. A paired
Interviews were transcribed and de-identified prior to analysis. Thematic analysis was guided by a team-based method 12 led by the first author and supported by two research assistants. Transcripts were equally distributed among the analysts for review of salient patterns and concepts. Analysis occurred over six stages: dynamic reading of transcripts, engaged codebook development, participatory coding, inclusive reviewing and summarizing of categories, collaborative analyzing, and data translation. 12 A pilot codebook was developed to organize codes into categories and sub-categories and applied to eight transcripts before a final codebook was established. The remaining eight transcripts were double coded using the final codebook with two members working independently; coded data was extracted and synthesized into themes with exemplar participant quotes. The analysis team held frequent, reflexive group meetings to reflect on interpretations until consensus was reached on themes. Member-checking was facilitated with the advisory committee and participants to validate interpretation and reporting of our findings.
Results
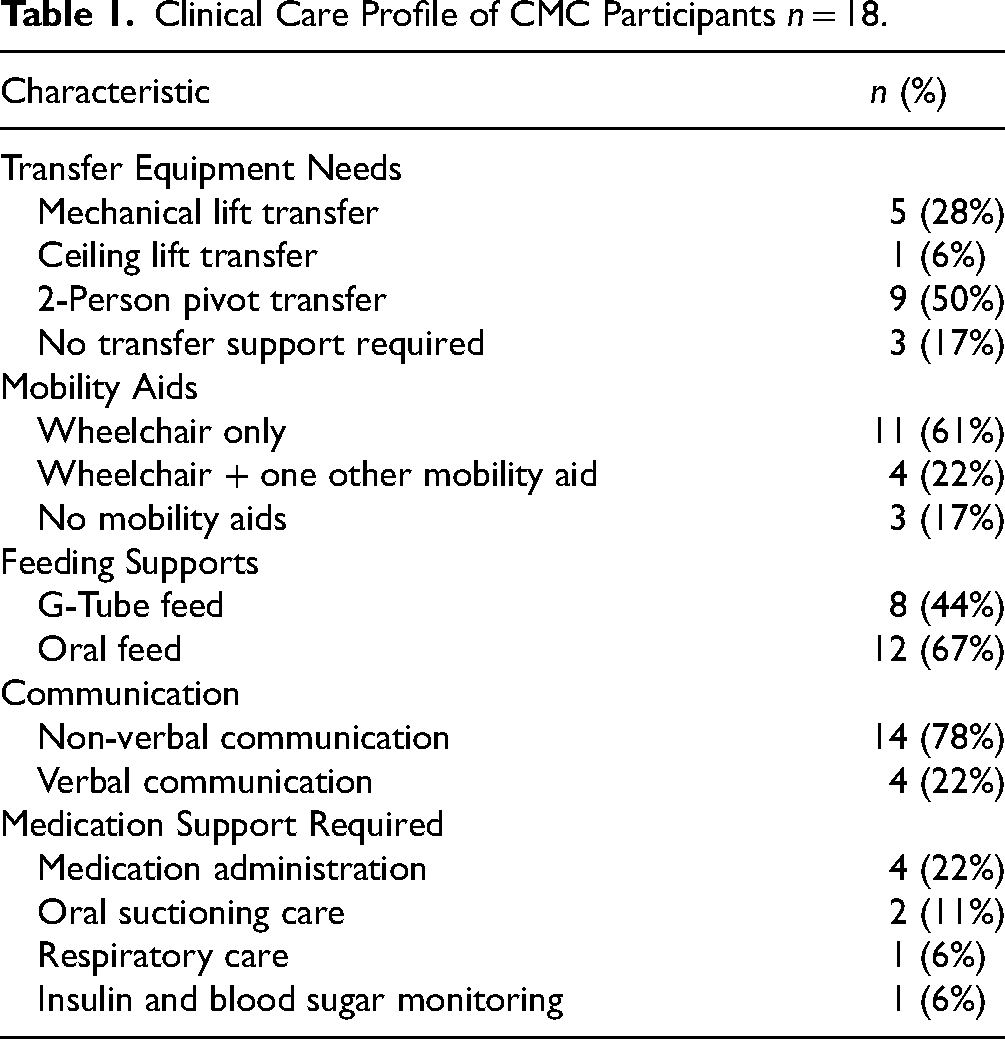
To understand the program's early impact, two overarching themes (a) improved quality-of-life; and (b) caregiver respite were derived from the pilot data. Table 1 highlights the medical complexity and clinical care needs within the playgroup.
Clinical Care Profile of CMC Participants
Improved Quality-of-Life
The PedsQL™ pre-post tests showed an increase across total scores (Table 2), which suggest that participants experienced an improvement in their overall quality-of-life functioning (27.4%, p = 0.006). There was significant improvement around the psychosocial domains (31.7%, p = 0.001) for participants in their emotional, social, and school functioning. Observations shared by parents bring context to these scores (Table 2); they described positive changes in disposition, motivation, and engagement in activities at home and school. Parents observed their child as happier at home, having a calm demeaner and sleeping better. Some noted their child/ren exhibiting signs of motivation, especially towards attending
Joint Data Display of Improved Quality-of-Life Theme.
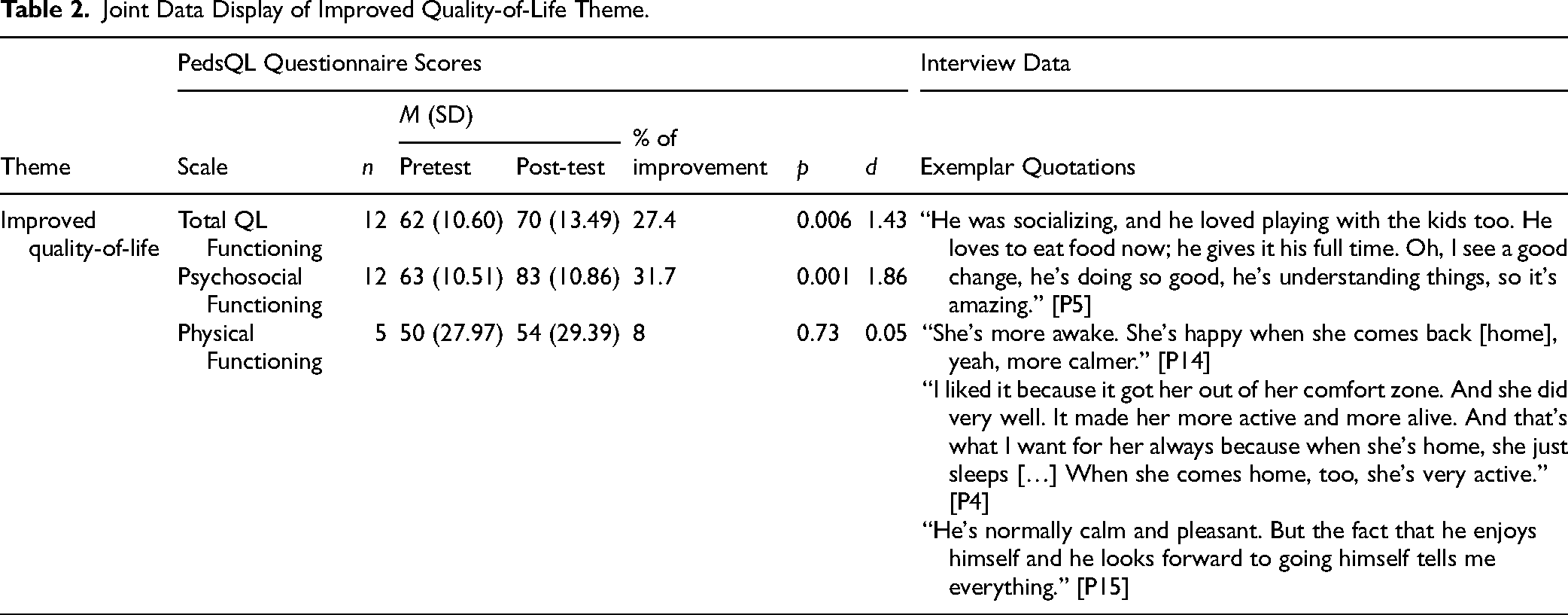
Conversely, while a small improvement was assessed in the physical functioning domain (8%, p = 0.73), most parents did not prioritize observing changes in this area considering their child's chronic conditions.
Caregiver Respite
All parents expressed satisfaction with the program, describing their experience as meaningful, and acknowledged the respite related benefits it afforded. Parents emphasized the value of “ “A couple of times it was me and my daughter going to the mall, and that was excellent because going during the week is not possible […] Or even hanging out with my parents, visiting my brother. I cannot do that with the [twins]. It's way too much disturbance for them.” [P1]
Notably, respite was possible only when trust with the program and staff was established. Trust and comfort were built through observation and engagement with staff. “I make sure that when I get a break like this it's first of all safe, good for her […] So just the level of care was so good, that the nurse was around. The whole team was around. So, I was completely comfortable, and I was able to do a few extra things that were getting piled up.” [P9]
Discussion
This pilot study provides better understanding of how programs that are designed for CMC can positively impact their quality-of-life, addressing an important social opportunity. Our early and exploratory findings indicate that participation in inclusive programming was particularly impactful in improving psychosocial domains rather than physical functioning, as demonstrated through the PedsQL questionnaire scores which parents validated through their insights. Most CMC participating in the pilot had chronic functional limitations and their mobility was supported by equipment or staff. Unlike other programming for children with disabilities evidenced in the literature,7,10
Another important outcome of our pilot findings were the extended benefits experienced by parents, whose stress and emotional exhaustion is well-documented.4,15,16 Parents of CMC have described challenges in addressing their personal needs due to fear for their child's safety in their absence.
15
At
Limitations
While our early findings are promising, due to the small sample size, statistical power and generalizability are limited. The exclusion of demographic characteristics related to CMC participants such as age, gender, and medical condition also limits the generalizability of the findings. We recommend future researchers evaluating recreational programming for CMC to consider a larger sample size and the addition of details related to diagnosis and abilities as they may be useful in identifying particular segments of the CMC population which may be best suited for play programming. Selection bias may have also elicited perspectives from participants with the strongest feelings towards the study.
Conclusion
The early insights from this pilot study support the need for inclusive play programs like
Footnotes
Acknowledgments
The authors would like to extend thanks and gratitude to all parents who graciously made time to participate in interviews. They also thank Emilia Cotter, Holly Opara, and Gary Grewal for their analytical support.
Data Availability Statement
The datasets generated and/or analyzed during the current study are not publicly available due to research ethics requirements ensuring participant anonymity and compliance with research ethics board regulations.
Declaration of Conflicting Interests
Authors are researchers employed by the organization at which the research was conducted.
Ethical Approval
All study procedures and materials received ethical approval from the University of Toronto Health Sciences Research Ethics Board REB Protocol #37615.
Funding
This research did not receive any funding from agencies in the public, commercial, or not-for-profit sectors.
Informed Consent
Written informed consent was obtained from participants before data collection.
