Abstract
A survey on vaccinations was conducted among patients with chronic inflammatory diseases (CIDs), including inflammatory bowel disease, inflammatory rheumatological diseases, and dermatological diseases (DDs). The objective was to identify differences between established vaccination guidelines and actual practices in healthcare as experienced by patients. Among eligible responders (n = 1434), 57% were on immunomodulatory treatment, and 59% were treated in specialized care. The most recommended vaccines were COVID-19 (66%), influenza (63%), and pneumococcal (46%). Regarding common vaccination principles for patients with CID, 61% reported not receiving information on vaccinations before treatment initiation. Only 23% were advised to check their vaccination status before starting medications, and just 20% reported that their vaccination status is regularly assessed. Logistic regression revealed that the DD group was less likely, while patients over 65 years of age or on immunomodulatory treatment were more likely to be recommended and receive vaccinations. Ideally, all CID patients should be eligible for preventive immunization starting from the time of diagnosis. A targeted vaccination program with clear responsibilities across the healthcare system is strongly recommended.
Keywords
Introduction
Patients with chronic inflammatory diseases (CIDs), such as inflammatory rheumatic diseases (IRDs), dermatological diseases (DDs), and inflammatory bowel diseases (IBDs), are commonly treated with immunomodulatory drugs, which increased their susceptibility to infections.1–3 Preventive immunization is therefore critical, and specific guidelines have been issued by the European Alliance of Associations for Rheumatology (EULAR) and European Crohn's and Colitis Organization (ECCO) for IRD and IBD, repectively.1,2,4 Conversely, the European Academy for Dermatology and Venerology (EADV) has yet to release recommendations for preventive immunization in patients with DD. However, clinical reviews from the United States have offered guidance for individuals with psoriasis and atopic dermatitis.3,5
Nordic countries have well-established national vaccination programs for the general population. Local guidelines for inflammatory diseases6–9 are based on European vaccination recommendations.1,2,4,5 Nordic countries have similar healthcare systems, vaccination coverages, and reimbursement policies. However, preventive immunization in patients with CID has not been studied previously.
The successful immunization of patients with CID faces multiple challenges.2,10,11 Vaccine hesitance has been linked to a lack of awareness among patients and healthcare providers.12,13 This gap is partly attributed to insufficient knowledge, limited patient interaction time, and unclear responsibilities for identifying candidates for preventive immunization and ensuring timely vaccination.13–16
The objective of this survey was to identify gaps between existing European and Nordic vaccination recommendations and current healthcare practices experienced by patients in the Nordic countries (Denmark, Finland, Norway, and Sweden). Additionally, the survey assessed the patients’ understanding of immunization principles related to their disease and treatment.
Methods
Design and Setting
This quantitative, structured, and anonymous survey (Questback) for adults (≥ 18 years) with self-reported CID was conducted in the Nordic countries from June to November 2022. All participants were required to provide consent for the aggregated publication of their anonymous responses before proceeding with the survey. A survey was conducted in Sweden, Denmark, and Norway through Facebook. In Finland, it was distributed via newsletters and patient organization websites. With similar ethical standards across Nordic countries, ethics committee approval was not required for anonymous, survey-based research, as it did not involve identifiable human subjects, individual data, or biological materials.17–20 A data privacy impact assessment was conducted to ensure anonymity of responders and compliance with data privacy regulations.
Statistical Analysis
Descriptive statistics (Microsoft Excel®) in terms of counts (%) were used to describe the knowledge and awareness of vaccination among the 3 CIDs and across different demographic subgroups and vaccination types. Multivariate logistic regression models adjusted for demographics, confounding variables, and disease status (age, sex, ethnicity, CID status, and education) were used to estimate odds ratios for variables of interest between the study groups. The significance threshold for associations was set at 0.05. Logistic regression models using the binomial function were implemented in R (version 4.4.3), with the stats library, and model fit was evaluated using DHARMa 21 to ensure robust performance. The 4-scale awareness (fully unaware, quite unaware, somewhat aware, and fully aware) on EULAR principles of vaccination and recommendations for patients with CIDs were reported as count (%) for older adults (≥ 65 years) and immunomodulatory treatment, respectively. General vaccination principles are similar for patients with IRD, DD, and IBD treated with immunomodulatory treatments, including screening and disease management. A Numeric Rating Scale (NRS) from 0 to 10 was applied, that is, to measure patient satisfaction. Responses on an NRS of 7 to 10, 4 to 6, and 0 to 3 were predefined as satisfied, neutral, and unsatisfied, respectively.
Results
Study Population
A total of 2169 respondents answered the survey. After removing responses with > 20% missing values, 1748 respondents remained, including 1031 patients with IRD, 543 with IBD, and 563 with DD. The IRD group included individuals with rheumatoid arthritis (N = 408), psoriatic arthritis (N = 278), juvenile idiopathic arthritis (N = 9), ankylosing spondylitis (N = 74), and other conditions (N = 377). The DD group included patients with atopic dermatitis (N = 116), psoriasis (N = 357), and other conditions (N = 139), whereas the IBD group included patients with Crohn's disease (N = 218) and ulcerative colitis (N = 306). Respondents who reported their disease as “other” only, with no other condition in the IRD or DD group, were excluded from the analysis. This group of respondents could potentially include patients with noninflammatory conditions, such as osteoarthritis, fibromyalgia, and keratosis, which are beyond the scope of this study. All subsequent analyses were based on responses from 1434 respondents who clearly identified themselves as belonging to the IRD, IBD, or DD groups (Supplemental Figure S1).
Demographic and Clinical Characteristics
The demographics of respondents and their respective disease groups are presented in Table 1. The time since the first diagnosis was > 5 years for 77% of the respondents (N = 1109), with 61% receiving their first diagnosis more than 10 years ago. At the time of the survey response, the proportion of patients taking medication, including oral steroids, immunomodulatory or immunosuppressive agents, biological treatment, Janus kinase inhibitors, topical treatment (only DD patients), and rectal suppositories (only IBD patients) was 87% for IRD, 80% for IBD, and 74% for DD. Among the IRD and IBD respondents, immunomodulatory treatments (oral steroids, conventional or targeted immunosuppressive treatment, and biological treatment options) were the most common medications (Table 1), whereas topical treatment was the most common among DD respondents.
Demographic Characteristics of the Participants (N = 1434), n (%).
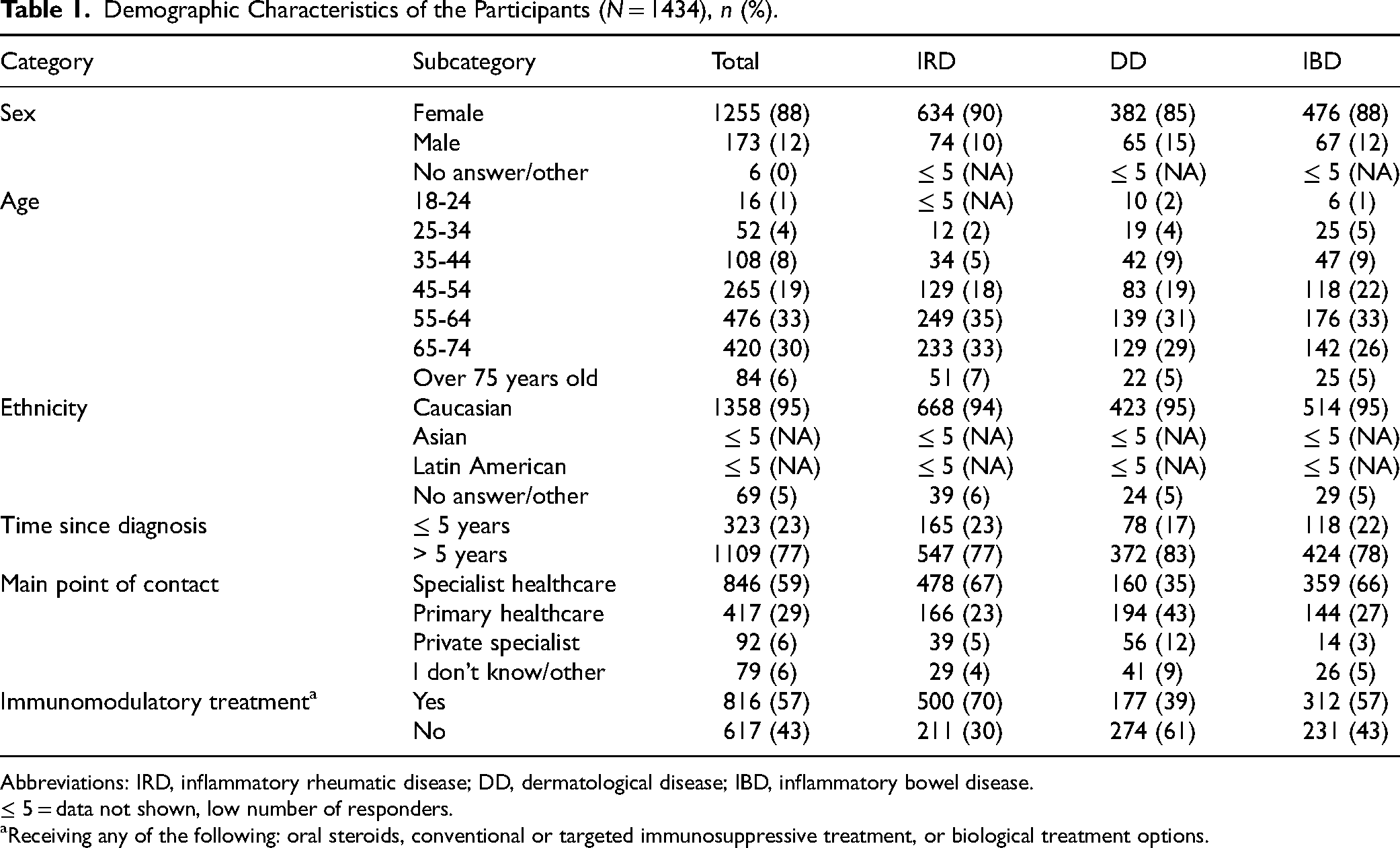
Abbreviations: IRD, inflammatory rheumatic disease; DD, dermatological disease; IBD, inflammatory bowel disease.
≤ 5 = data not shown, low number of responders.
Receiving any of the following: oral steroids, conventional or targeted immunosuppressive treatment, or biological treatment options.
Vaccines Recommended and Received
The survey aimed to investigate the vaccines recommended by healthcare professionals and those reported as received by patients either prior to or during ongoing treatment, or before the initiation of treatment. The vaccinations most recommended by healthcare professionals were COVID-19 (66%), influenza (63%), and pneumococcal (46%). Correspondingly, 80% of the respondents received the COVID-19 vaccine, 72% the influenza vaccine, and 50% the pneumococcal vaccine, either prior to or during treatment.
After adjusting for CID status, type of treatment, main point of contact (primary or specialized care), education, ethnicity, age, and sex, patients aged ≥ 65 years had a significantly higher probability of receiving a recommendation for pneumococcal (OR: 2.29, P < .001) and influenza (OR: 1.65, P < .001) vaccines when compared to patients aged < 65 years. However, age was not a significant predictor of COVID-19 vaccination recommendations. Patients with IRD had significantly higher odds of receiving recommendations for pneumococcal (OR: 2.00, P < .001), influenza (OR: 1.61, P = .004), and COVID-19 (OR: 1.78, P = .001) vaccines. Conversely, patients with DD were significantly less likely to receive recommendations for pneumococcal (OR: 0.64, P = .005) and influenza (OR: 0.63, P = .004) vaccines. IBD status was not associated with a significant difference in vaccine recommendation probability. Immunomodulatory treatment for CID was associated with significantly higher odds of receiving recommendations for pneumococcal (OR: 2.07, P < .001), influenza (OR: 1.98, P < .001), and COVID-19 (OR: 1.56, P = .001) vaccines. Additionally, patients managed in specialized healthcare settings were significantly more likely to receive vaccine recommendations (OR: 1.72, P < .001) than those managed in primary healthcare settings (Supplemental Table S1).
When adjusting for sex, age, ethnicity, education, type of treatment, main point of contact, and CID status, logistic regression models indicated that patients aged ≥ 65 years had significantly higher odds of receiving pneumococcal (OR: 2.78, P < .001) and influenza (OR: 2.35, P < .001) vaccines either before or during treatment than younger patients. Additionally, patients of non-Caucasian ethnicity exhibited significantly lower odds of receiving the influenza vaccine (OR: 0.57, P = .046). Patients with IRD had increased odds of receiving a pneumococcal vaccine (OR: 1.56, P = .009). Conversely, patients with DD had significantly reduced odds of receiving pneumococcal (OR: 0.64, P = .005) and influenza (OR: 0.58, P = .002) vaccines before or during treatment. Immunomodulatory treatment for CID significantly increased the likelihood of receiving pneumococcal (OR: 1.65, P < .001), influenza (OR: 2.07, P < .001), and COVID-19 (OR: 1.91, P < .001) vaccines before or during treatment. Additionally, patients managed by specialized healthcare (OR: 1.65, P < .001) had increased odds of receiving the vaccine compared to those managed in primary healthcare settings (Supplemental Table S1).
Logistic regression models indicated that, after adjusting for sex, age, ethnicity, education, type of treatment, main point of contact, and CID status patients aged ≥ 65 years had significantly higher odds of receiving pneumococcal (OR: 1.88, P < .001), influenza (OR: 1.72, P < .001), and COVID-19 (OR: 1.62, P < .001) vaccines before initiating treatment compared to patients aged < 65 years. The probability of receiving the COVID-19 vaccine before treatment was significantly higher among males (OR: 1.62, P = .009) than among females and individuals from non-Caucasian ethnic backgrounds (OR: 2.07, P = .006). Patients receiving immunomodulatory treatment had a significantly higher probability of receiving pneumococcal vaccine (OR: 1.69, P = .006), but a significantly lower probability of receiving COVID-19 vaccine (OR: 0.60, P < .001).
Vaccination Recommendations, Information, and Satisfaction
Among patients aged ≥ 65 years, awareness of vaccination recommendations, including the importance of informing healthcare providers about their disease and medication, annual flu vaccine, and pneumococcal vaccine, was generally high (Figure 1a). Conversely, recommendations for yellow fever, hepatitis, and herpes zoster vaccinations are known to a much lesser extent. Similar unawareness was observed regarding recommendations for family member vaccinations, annual assessments of vaccinations, and the preferred administration of vaccines prior to the planned treatment (Figure 1a). Figure 1b shows similar outcomes among respondents who reported receiving immunomodulatory treatments for their disease.

(a) Proportion of patients with chronic inflammatory diseases aged ≥ 65 years reporting awareness of vaccination principles. The numbers in the boxes indicate the percentage of patients who were fully aware, somewhat aware, quite unaware, or fully unaware in responses to each question listed on the left. (b) Proportion of patients with chronic inflammatory diseases on immunomodulatory treatment reporting awareness of vaccination principles. The numbers in the boxes indicate the percentage of patients who were fully aware, somewhat aware, quite unaware, and fully unaware in responses to each question listed on the left.
Most respondents (61%) reported not receiving any information regarding the recommended vaccinations when they first initiated treatment (IRD: 53%, IBD: 61%, and DD: 74%). Overall, 47% of the respondents reported finding information on vaccine recommendations specific to their disease or treatment somewhat or very difficult, while 74% expressed a desire to receive more information. After adjusting for age, sex, ethnicity, education, type of treatment, main point of contact, and CID status, the likelihood of being informed was significantly higher in male than in female patients (OR: 1.68, P = .01), in those with IRD (OR: 1.70, P = .008), and among individuals managed by specialized care compared with primary care (OR: 2.17, P < .001) (Figure 2). Patients receiving immunomodulatory treatment had a significantly higher chance of being informed (OR: 2.90, P < .001). Patients aged ≥65 years had a significantly lower likelihood of being informed compared to their younger counterparts (OR: 0.56, P < .001). Respondents with DD had a significantly lower odds of being informed (OR: 0.61, P = .006) (Figure 2).

Patient awareness and satisfaction with current immunization practices in the healthcare system (* P < .05).
Approximately one-fifth of the respondents reported being advised to check their vaccination status prior to initiating treatment, and a similar proportion confirmed undergoing regular assessments of their vaccination status (Table 2). Most respondents had not received a vaccination plan from their healthcare providers. These proportions were consistently low across the 3 therapeutic groups (Table 2). Overall, 43% of the respondents were unsatisfied with the current guidelines on vaccination provided by healthcare professionals. After controlling for sex, age, education, ethnicity, type of treatment, main point of contact, and CID status, individuals aged ≥ 65 years (OR: 1.38, P = .015) and those receiving immunomodulatory treatments (OR: 1.46, P = .01) had significantly higher odds of being very satisfied with the vaccine guidance provided by healthcare professionals. Conversely, individuals with DD had significantly lower odds of being very satisfied (OR: 0.57, P = .001) (Figure 2).
Proportion of Patients (%) With Positive Response to Vaccination Checks Prior to Treatment, Plan Received, and Regular Assessment (N = 1434).

Abbreviations: CID, chronic inflammatory diseases; IRD, inflammatory rheumatic diseases; DD, dermatological diseases; IBD, inflammatory bowel diseases
Overall, 61% of the respondents indicated that the COVID-19 pandemic increased their awareness of vaccination, particularly among those aged > 65 years (67%). Additionally, 46% of the respondents noted that the pandemic had enhanced their awareness of immunizations related to their medical conditions or ongoing treatments.
A significant proportion (48%) of respondents considered themselves responsible for their vaccinations. When asked, “Who should be responsible for your vaccinations related to your disease and treatment?,” opinions were divided, with 21% responding that patients themselves, 27% general practitioners, 26% specialists, and 20% indicating that responsibility depended on the type of vaccine.
Discussion
The findings of this survey highlight significant gaps between vaccination guidelines1,2,6–9 and real-world experiences of patients with CID. These discrepancies are evident in patients’ self-reported experiences, expectations, and lack of clear responsibility concerning vaccination recommendations and administration in practice. Patients with CID may benefit from more structured vaccination plans. Ideally, this plan should be established and thoroughly communicated shortly after diagnosis, and at least before the initiation of immunomodulatory treatment. Although the guidelines recommend that vaccines be administered prior to planned immunosuppression and assessed regularly, they are intended only as guidance for treating specialists to inform patients. Currently, no vaccination programs specifically designed for patients with CIDs clearly define the responsibilities of the healthcare system. National health authorities should manage the vaccination framework, including the guidelines, reimbursement criteria, and communication strategies. Specialist healthcare professionals should develop and communicate vaccination plans tailored to individual needs. Primary care centers should ensure timely vaccination. Moreover, patient education is a collective responsibility across all healthcare levels.
The survey revealed that both the percentage of patients recommended for vaccination and actual vaccination rates before treatment initiation were suboptimal, particularly among younger patients and those in the DD group. Furthermore, most respondents were unfamiliar with the key principles of vaccination, such as the importance of a vaccination check before treatment initiation and regular assessment of vaccination status (Figure 1 and Table 2). Vaccination rates and awareness of common vaccinations, such as Influenza, COVID-19, and pneumococcal infections, were generally high across different disease groups. This is consistent with the well-functioning healthcare system and preventive immunization practices in Nordic countries. However, the awareness of less common vaccines and important principles relevant to patients with CID remains low (Figure 1). This highlights a significant gap between clinical guidelines and their implementation in clinical practice, which cannot be solely attributed to recall bias. Comparative studies, such as a US survey of patients with IBD, reported higher influenza vaccination rates (82%) but similar suboptimal rates for pneumococcal (43%), hepatitis A (34%), and hepatitis B (48%) vaccines. 22 This issue is compounded by several factors identified in previous research, including inadequate knowledge among patients and healthcare professionals, time constraints during patient consultations, and unclear responsibility for vaccine management.22–26 Our findings reflect similar challenges and highlight the need for improved communication and education, clearly defined accountability, and comprehensive reimbursement strategies.
Healthcare providers often fail to communicate key principles, such as advising patients to check their vaccination status before initiating treatment, providing a vaccination plan, and conducting regular reviews (Table 2). Patients with IRD and IBD tended to have slightly higher vaccination rates than those with DD, a difference that diminished among respondents aged ≥ 65 years, likely due to heightened public health campaigns targeting influenza and COVID-19 in older populations. For patients with DD, who are often managed in primary care with topical treatments, vaccination awareness and uptake are particularly low, especially among those aged < 65 years. This may reflect a limited engagement by primary care providers or a lack of disease-specific vaccination guidelines for DD, unlike those for IRD and IBD.
Patients aged > 65 years are better informed and have higher vaccination coverage. This is consistent with the general population, where the same age groups are generally well-informed and have better vaccination rates than their younger counterparts. However, in patients with CID, advanced age or the use of immunomodulatory drugs should not solely determine vaccine recommendations or improve preventive measures. All patients with CID are eligible for preventive immunization because of the increased risk of infections associated with autoimmune, inflammatory disease.27–29 Over time, a substantial number of patients diagnosed with CID require treatment with immunomodulatory drugs, which increase the risk of infections.1,2 The overarching principles for vaccination in adult patients, as recommended by the EULAR, suggest immunization preferably during periods of stable disease and ideally before the planned initiation of immunosuppressive treatment. 1 The ECCO guidelines recommend that vaccination history should ideally be obtained at diagnosis, and any outstanding vaccinations should be administered. If clinically safe to delay immunosuppressive therapy, outstanding live vaccinations should be considered before initiating immunosuppression, as per the guidelines. 2 Our results highlight the need for better compliance with immunization principles among healthcare providers. The involvement of multiple healthcare providers across primary and specialist care often creates uncertainty about who is responsible for preventive immunization. A dedicated vaccination program could help assign responsibility within specific layers of the healthcare system, while the final decision to receive vaccination remains with the patient.
The strength of this study was its broad reach across 4 Nordic countries with similar healthcare policies, capturing the perspectives of a diverse group of patients. However, this study's reliance on self-reported data is both a strength and a limitation. While this provides a direct view of patient experiences, it also introduces potential biases, including recall bias, inability to verify medical histories or specific diagnoses, and inclusion of patients more engaged and aware of the disease. Additionally, the online nature of the survey may have led to underrepresentation of certain demographic groups, such as older adults (≥ 75 years), who are less familiar with digital platforms. 30 In future research on vaccination practices, partnering with elderly care facilities could be a more inclusive approach. Furthermore, this study excluded a significant number of patients with unclear or non-specific diagnoses, which may limit the generalizability of the findings to other autoimmune and inflammatory diseases. The survey was conducted after the COVID-19 pandemic disrupted health services. This likely affected how patients with CID received and processed information from healthcare providers, authorities, and media. While it may have increased awareness and boosted vaccination rates, continued efforts are needed to further improve preventive immunization in this vulnerable patient group. 10
Conclusions
This survey provided critical insights into the vaccination experiences of Nordic patients with CID and revealed substantial barriers to the effective implementation of vaccination recommendations. A gap exists between theoretical recommendations and real-world practice, driven by unclear responsibilities among healthcare providers, inadequate patient education, and inconsistent vaccination plans. Accordingly, a comprehensive authority-driven vaccination strategy is urgently needed for this patient group. One that integrates a clear vaccination plan at diagnosis, assigns explicit accountability for regular status checks across primary and specialist care, and ensures a structured approach. Enhanced collaboration between healthcare providers, coupled with robust patient education initiatives, could empower patients with CID and elevate vaccination rates, ultimately reducing the burden of vaccine-preventable diseases in this vulnerable population.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735251360485 - Supplemental material for Vaccine Knowledge and Awareness Among Patients With Chronic Inflammatory Diseases: Results From a Nordic Survey
Supplemental material, sj-docx-1-jpx-10.1177_23743735251360485 for Vaccine Knowledge and Awareness Among Patients With Chronic Inflammatory Diseases: Results From a Nordic Survey by Meliha Kapetanovic, Randeep Mandla, Susanne Thiesen Gren, Maria Seddighzadeh, Dan Henrohn, Maaria Palmroth, Anna-Maria Hiltunen, Jussi Ranta, Anna-Kaisa Asikainen, Anne Grete Frøstrup and Veli-Jukka Anttila in Journal of Patient Experience
Footnotes
Acknowledgments
We would like to thank all the respondents who generously shared their time and experience with us. We also thank Outi Isomeri and Petri Vänni from Nordic Healthcare Group, Finland, and Maria Lajer, Pfizer Denmark, for statistical and editorial support of this manuscript.
Data Availability Statement
The data supporting this study's findings are available from the corresponding author, RM, upon reasonable request.
Declaration of Conflicting Interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors AGF, RM, STG, DH and MS are employees/former employees of Pfizer and own stocks in the company. MP is an employee of MedEngine Oy, AMH, JR and AKA are employees of Nordic Healthcare Group, both contractors of Pfizer. The remaining authors declare no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by funding from Pfizer AB, Sweden, Pfizer OY, Finland, Pfizer Denmark, Ballerup, and Pfizer AS, Norway.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki. No ethical approval was required as the anonymous health survey-based research did not concern any identifiable human subjects, their individual data, or biological material. Approval from the Danish ethical committee is not required for survey-based studies in Denmark (komitélovens § 14, stk. 2, Komitéloven § 14 (danskelove.dk)). Approval from the Swedish ethical committee is not required as long as information collected for research purposes cannot be linked to personal information via any means. Vanliga frågor - Etikprövningsmyndigheten (etikprovningsmyndigheten.se). According to Norwegian Ethical Committee (REK) guidelines, no approval is required if the collected data are anonymous, and individuals are not identifiable based on the data Home – Insights (rekportalen.no). The Finnish Medical Research Act is not applicable to surveys and questionnaires as the law does not concern such kind of research as medical research. For nonmedical research involving human participants, the Finnish National Board on Research Integrity, TENK, has issued a set of guidelines on the ethical principles that were followed: The ethical principles of research with human participants and ethical review in the human sciences in Finland by the Finnish National Board on Research Integrity, TENK. Data were handled confidentially and in accordance with data protection legislation and GDPR, with the anonymity of respondents preserved throughout.
Statement of Human and Animal Rights
Not applicable.
Statement of Informed Consent
Informed consent for publication of results at the aggregated level was obtained from all participants before answering the survey, with anonymity of respondents preserved throughout.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
