Abstract
Gaining an understanding of patient preferences regarding treatment administration can help inform approaches to patient care. A discrete choice experiment survey comprising of 7 attributes (treatment environment, convenience, travel times, waiting times, interactions with patients, interactions with healthcare providers, and concerns about complications) was used to assess the stated preferences of patients with chronic conditions, including congenital thrombotic thrombocytopenic purpura (cTTP), for receiving intravenous prophylactic infusions administered at home versus in an outpatient setting. The survey also contained a time trade-off exercise. In total, 307 survey responses were included in the analyses, of which 25 were from patients with cTTP. An overall preference for receiving infusions at home was reported by 137 patients (44.6%), and a high proportion of patients (n = 187, 60.9%) were willing to trade at least 12 months of their lives to receive treatment at home. Patients considered being comfortable with the treatment environment to be the most important attribute. These findings provide insights into the importance of the treatment setting preferences of patients with chronic conditions receiving prophylactic infusions.
Keywords
Key Points
A discrete choice experiment (DCE) survey was used to assess the stated preferences of patients with chronic conditions for receiving intravenous prophylactic infusions administered at home versus in an outpatient setting.
An overall preference for receiving infusions at home was reported by 137/307 (44.6%) patients.
A high proportion of patients (n = 187/307, 60.9%) were willing to trade at least 12 months of their lives to receive treatment at home.
Patients considered being comfortable with the treatment environment to be the most important attribute within the DCE survey.
Introduction
Congenital thrombotic thrombocytopenic purpura (cTTP) is an ultra-rare (estimated diagnosed prevalence of 0.5-2 cases per million) thrombotic microangiopathy.1–3 Congenital thrombotic thrombocytopenic purpura is caused by an inherited deficiency of the enzyme ADAMTS13 (a disintegrin and metalloproteinase with thrombospondin motifs 13), leading to the accumulation of ultra-large von Willebrand factor multimers with a high platelet-binding capacity.1,4 The resulting platelet-rich microthrombi and organ ischemia can lead to long-term organ damage.1,4
Current treatment approaches for cTTP involve ADAMTS13 replacement with intravenous (IV) infusions of plasma-based therapies. 5 Treatment guidelines suggest that patients with cTTP receive 10 to 15 mL/kg plasma infusions as maintenance therapy every 1 to 3 weeks, whereas patients who are symptomatic should receive daily infusions until symptoms resolve and the patient's platelet count returns to the normal range. 6 Plasma-based therapies are, however, associated with a high treatment burden, as they can only be administered in a clinical setting, necessitating patients to travel to receive treatment. 7 The large infusion volumes associated with plasma-based therapies can take several hours to administer, further adding to the treatment burden.5,7 Repeated vascular access and the risk of allergic reactions associated with plasma infusions also pose concerns.7,8
A recombinant ADAMTS13 (rADAMTS13; Takeda Pharmaceuticals USA, Inc.) enzyme was approved by the US Food and Drug Administration in 2023 and the European Medicines Agency in 2024 for prophylactic or on-demand ADAMTS13 replacement therapy in patients with cTTP.9,10 rADAMTS13 prophylactic IV infusions are shorter in duration and appear to have an improved safety profile compared with plasma-based therapies. 11
Gaining an understanding of patient preferences regarding treatment administration can help to inform approaches to patient care. The treatment preferences of patients with cTTP are not currently reported in the literature. An online discrete choice experiment (DCE) survey was developed to assess the preferences of patients with cTTP for receiving regular IV prophylaxis at home versus in an outpatient setting. Owing to the low diagnosed prevalence of cTTP, the study was extended to include patients with other chronic conditions that require similar treatment to cTTP in terms of dosing frequency and mode of administration, such as multiple sclerosis and rheumatoid arthritis.
Methods
Study Design
A targeted literature review was initially conducted to identify attributes for inclusion in the DCE. These attributes were subsequently validated by qualitative interviews with healthcare providers (HCPs) and patients with cTTP (unpublished data). Owing to the low diagnosed prevalence of cTTP, qualitative interviews were also conducted with patients with other chronic conditions that require regular prophylactic infusions: Gaucher disease, multiple sclerosis, myasthenia gravis, hemophilia, Fabry disease, and chronic inflammatory demyelinating polyneuropathy. The interviews indicated that patients with cTTP and those with other chronic conditions had similar opinions when describing the impact of the attributes. In addition, all attributes were valued at a similar level of importance by both patient groups. Based on these findings and the low diagnosed prevalence of cTTP, the study was expanded to include patients with other chronic conditions. Further information describing how the attributes and levels used in the DCE were identified is provided in Supplemental Methods, Supplemental Table 1, and Supplemental Table 2. Seven attributes with 26 associated attribute levels were identified for inclusion in the DCE (Supplemental Table 3).
Procedure and Study Population
The study was conducted from February to October 2022 in 5 countries: France, Germany, Spain, the United Kingdom, and the United States. Patients who completed the survey qualified for the study if they were ≥18 years old and receiving prophylactic IV infusions for one of the following: cTTP, multiple sclerosis, rheumatoid arthritis, chronic liver disease, cirrhosis, aplastic anemia, chronic graft versus host disease, chronic lymphocytic leukemia, congestive heart failure, Crohn's disease, von Willebrand disease, Gaucher disease, amyotrophic lateral sclerosis, or other thrombocytopenia (not thrombotic). Patients must have received prophylactic IV infusions for ≥3 months (frequency ranging from once a week to once every 3 weeks) in either an outpatient setting or a hybrid outpatient and at-home setting. Patients with a condition that would interfere with their ability to understand or interpret the questions in the survey were not eligible for inclusion.
Potential participants were identified using a variety of direct-to-patient approaches, including local social media outreach, patient advocacy groups, and local patient panels, in addition to a digital recruitment approach through a third-party vendor. Direct-to-patient approaches were used to help identify as many patients as possible, particularly those with rare or ultra-rare conditions. All patients interested in participating completed an online screening to determine eligibility. After completing the screening, those who were considered eligible were notified and asked to provide their consent online before being redirected to the survey hosted on the QualtricsXM online platform. A pilot survey involving 15 patients from each country was run to test the feasibility, ease of use, and understanding of the survey. The pilot survey took approximately 45 min to complete, and each participant received an honorarium for their time. Modifications to the survey were made based on the pilot testing feedback.
The final survey was composed of 6 sections (
The multinational study was approved by an independent review board, WCG. Patients provided online consent to a data protection notice before accessing the survey. By agreeing to the data protection notice, patients consented to their data being used in the study, unless they voluntarily withdrew permission.
Sample Size
The sample size was determined based on information reported in the literature (see
Statistical Analysis
Continuous variables were described using means and standard deviations. Categorical variables were described using frequency and percentages. Data were analyzed using SAS version 9.20. A multinomial logit regression (MNL) model was used to analyze patient preference weights for each attribute and associated attribute levels. The patient's utility (the dependent variable) for a specific treatment was described by a non-explainable random component and an explainable component (see
The TTO exercise was used to estimate the utility and disutility of the attributes associated with the stated preferences in the DCE. A one-way error components random effect model, considering the variation within and between respondents, represented the different health states shown to each participant. The predicted utility values were the average value for a given health state identified in the DCE survey for a given TTO time horizon estimated directly on the health utility scale. A conditional logit model provided the estimates of the utilities derived from the DCE on a latent utility scale.
Results
Participants
A total of 307 (France, n = 18; Germany, n = 95; Spain, n = 96; United States, n = 91; United Kingdom, n = 7) of 1047 completed survey responses were considered legitimate. Median (range) age was 44.0 (20.0-78.0) years, and 165 (53.7%) patients were male at birth (Table 1). The 2 most common health conditions were rheumatoid arthritis (24.1%) and Crohn's disease (19.5%). There were 25 patients (8.1%) with cTTP.
Patient Demographics and Clinical Characteristics (N = 307).
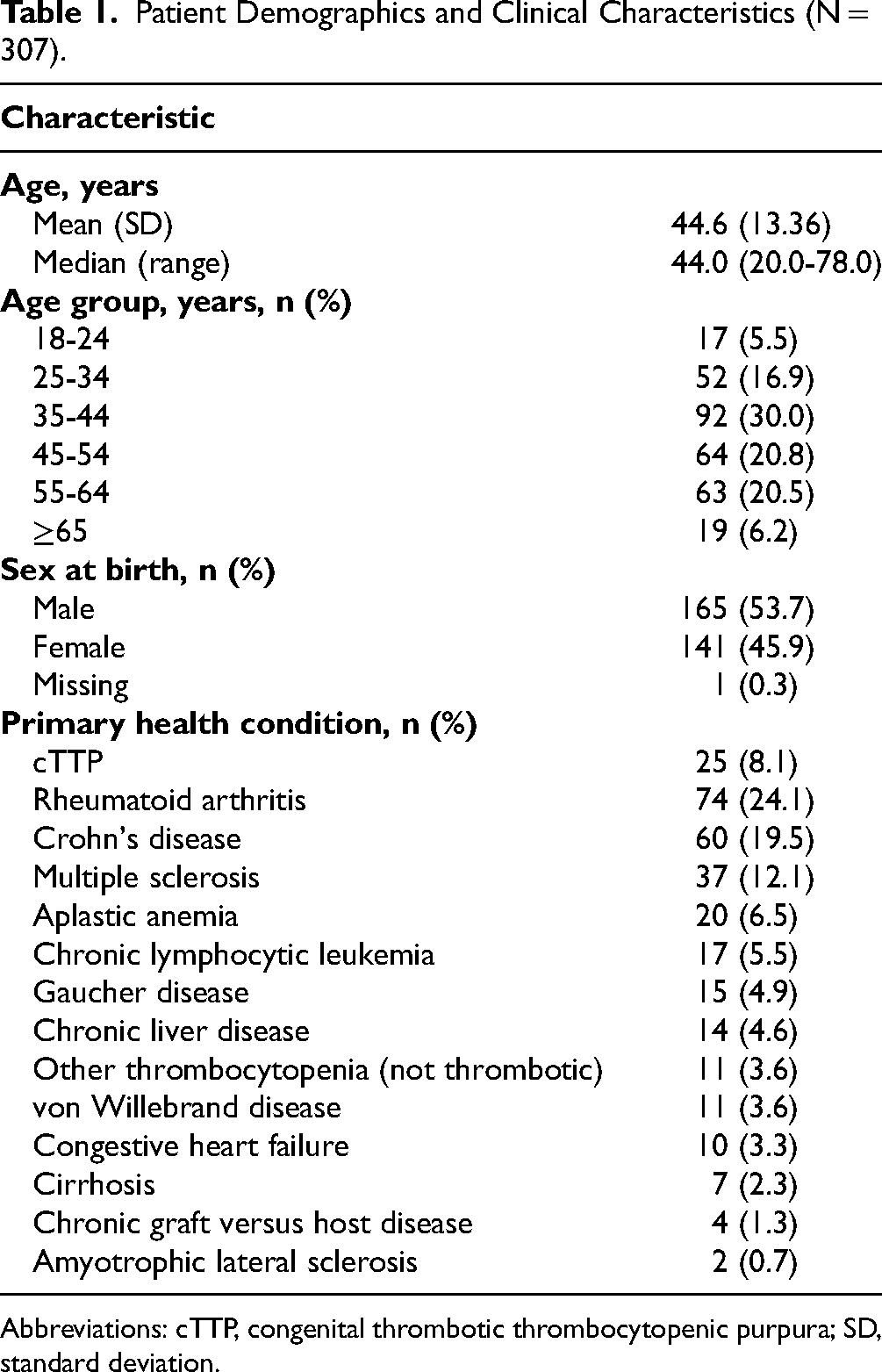
Abbreviations: cTTP, congenital thrombotic thrombocytopenic purpura; SD, standard deviation.
Overall Patient Preferences
A total of 137 patients (44.6%) reported an overall preference for receiving infusions at home, 85 (27.7%) for receiving treatment in a hospital/clinic, and 85 (27.7%) for receiving treatment in a hybrid outpatient and at-home setting (Figure 1A). Treatment administered at home by either a doctor or a nurse was reported as the most preferred option by 125 (40.7%) patients (Figure 1B and C). For patients with cTTP, 11 (44.0%) reported a preference for receiving infusions at home, 4 (16.0%) in a hospital/clinic, and 10 (40.0%) in a hybrid outpatient and at-home setting (Supplemental Table 4). Treatment administered at home by either a doctor or a nurse was the most preferred option for 11 (44.0%) patients with cTTP.

Patient preferences for the administration of hypothetical prophylactic intravenous (IV) infusions either at home or in an outpatient setting (A) overall treatment preference, (B) most preferred option, and (C) least preferred option.
Discrete Choice Experiment Preference Weights for Each Attribute
Patients placed the greatest utility on being comfortable with the treatment environment (estimate: 0.891; 95% CI: 0.714-1.062; Table 2). Although greater value was placed on interacting with other patients compared with having no interactions (estimate: 0.120; 95% CI: −0.020, 0.263), this difference was not statistically significant.
DCE Multinomial Logistic Regression Analysis.a
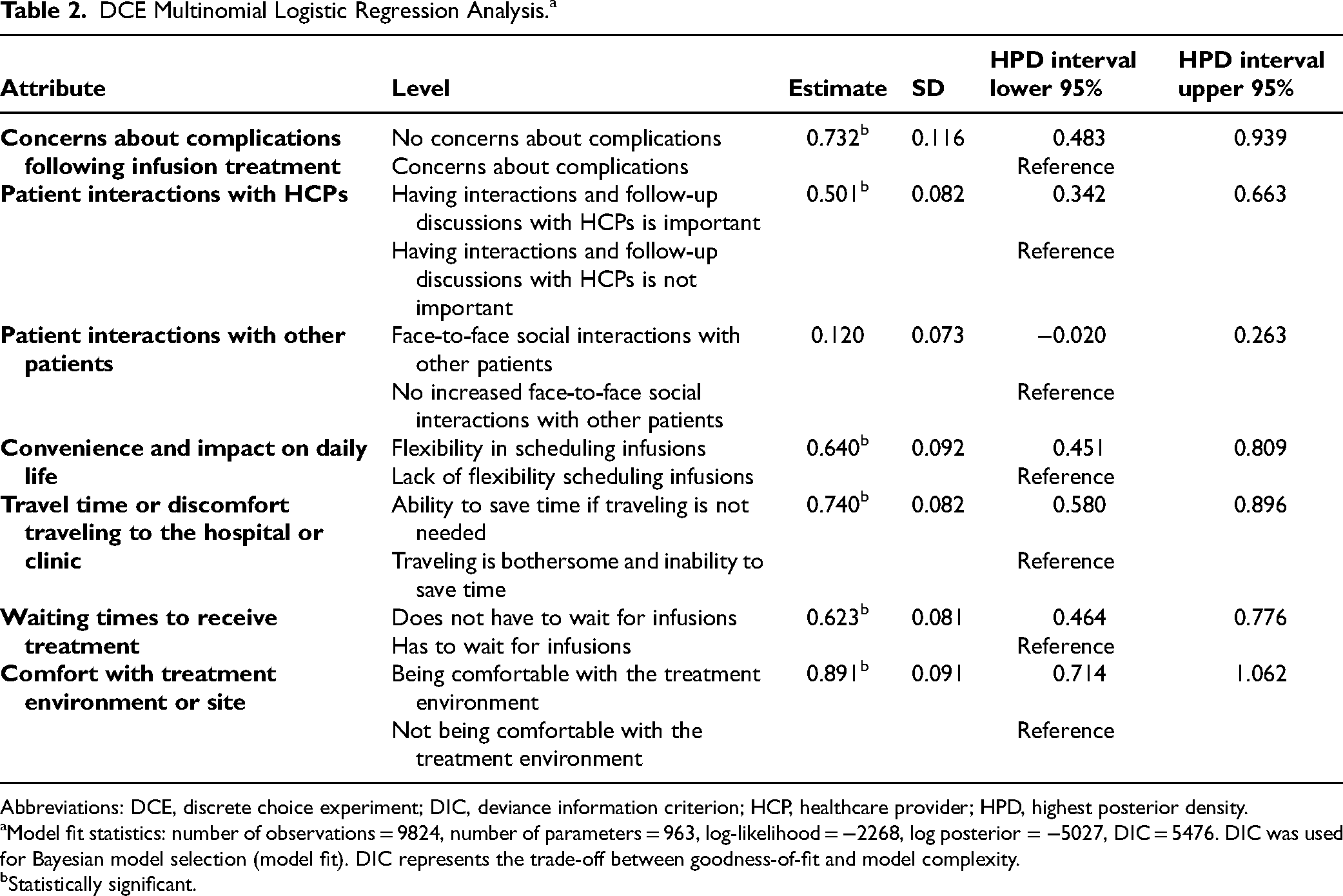
Abbreviations: DCE, discrete choice experiment; DIC, deviance information criterion; HCP, healthcare provider; HPD, highest posterior density.
Model fit statistics: number of observations = 9824, number of parameters = 963, log-likelihood = −2268, log posterior = −5027, DIC = 5476. DIC was used for Bayesian model selection (model fit). DIC represents the trade-off between goodness-of-fit and model complexity.
Statistically significant.
Impact of Patient Characteristics on DCE Attributes
The results from the MNL model to assess the impact of patient factors on preference weights within the DCE are presented in Supplemental Table 5. For the “complications following treatment” attribute, the selection of having “no concerns about complications” was influenced by patient age and whether the patient had cTTP. Older patients placed greater value on having no concerns about complications compared with younger patients (estimate: 0.023; 95% CI: 0.005-0.041). Patients with cTTP also placed greater value on having no concerns about complications compared with patients with other chronic conditions (estimate: 0.896; 95% CI: 0.004-1.808). However, there was a large difference between the number of patients with cTTP (n = 25) versus other chronic conditions (n = 282).
For the “patient interactions with HCPs” attribute, patients in France placed less value on having interactions with HCPs compared with patients in the United States (estimate: −0.678; 95% CI: −1.379, −0.035). Location also had an impact on the “travel time or discomfort traveling to clinic” attribute as patients in the UK placed less value on being able to save time traveling compared with patients in the United States (estimate: −0.998; 95% CI: −2.014, −0.038). It should be noted that the number of patients in France (n = 18) and the UK (n = 7) were low.
For the “convenience and impact on daily life” attribute, the selection of having “flexibility in scheduling infusions” was influenced by patient age and when the patient had been diagnosed. Older patients placed greater value on flexibility compared with younger patients (estimate: 0.019; 95% CI: 0.007-0.032). Similarly, patients who had been diagnosed >5 years ago placed greater value on flexibility compared with those diagnosed <1 year ago (estimate: 0.744; 95% CI: 0.255-1.269).
No statistically significant impact was observed for any patient characteristics on the “patient interactions with other patients,” “waiting times for treatment,” and “comfort with treatment environment” attributes.
Ranking of Attributes in the DCE
The “concerns about complications following infusion treatment” attribute was ranked as the most important by 152 (49.5%) patients (Supplemental Table 6). “Patient interactions with other patients” was ranked as the least important by 141 (45.9%) patients. The “concerns about complications following infusion treatment” attribute had an overall ranking weight of 5.79, indicating that patients considered this to be the most important attribute, whereas “patient interactions with other patients” had the lowest overall ranking weight of 2.37.
Participant Comprehension of the DCE
Overall, 230 (74.9%) patients reported a full understanding of the scenarios included in the DCE, and 304 (99.0%) patients reported understanding all attributes (Supplemental Table 7).
Time Trade-off Exercise
A high proportion of patients (n = 187, 60.9%) were willing to trade off 12 months of their lives to receive treatment at home (Table 3). In addition, 181 (59.0%) patients were willing to trade off 12 months so that complications did not need to be managed in a hospital setting, and 173 (56.4%) were willing to trade off 12 months to avoid traveling to receive treatment. The attributes associated with the highest utility estimates (compared with the reference level) were patients being able to receive treatment at home (average utility estimate: 0.0148), hospitalization not being needed to manage complications (estimate: 0.0142), flexible scheduling (estimate: 0.0139), and not needing to travel to a hospital/clinic for treatment (estimate: 0.0130; Supplemental Table 8).
Descriptive Statistics From the Time Trade-off Exercise.

Abbreviation: SD, standard deviation.
Discussion
Gaining an understanding of patient preferences regarding treatment administration can help to inform approaches to patient care. The initial intention had been to develop a DCE survey to assess the preferences of patients with cTTP for receiving regular IV prophylaxis at home versus in an outpatient setting; however, owing to the low diagnosed prevalence of this condition, 3 it was not feasible to recruit enough patients with cTTP to meet DCE sample sizes recommended in the literature.16,17 Consequently, this study was extended to include patients with other chronic conditions that require similar treatment to cTTP in terms of dosing frequency and mode of administration. The decision to extend the study was supported by findings from the qualitative interviews used to validate the attributes used in the DCE. The interviews were conducted with patients with cTTP as well as with patients with other chronic conditions that require regular prophylactic infusions: Gaucher disease, multiple sclerosis, myasthenia gravis, hemophilia, Fabry disease, and chronic inflammatory demyelinating polyneuropathy. The interviews indicated that patients with cTTP and those with other chronic conditions had similar opinions when describing the impact of the attributes. In addition, all attributes were valued at a similar level of importance by both patient groups. Based on these findings, patients with one of the following conditions who had been receiving prophylactic IV infusions for ≥3 months were recruited to complete the survey: cTTP, multiple sclerosis, rheumatoid arthritis, chronic liver disease, cirrhosis, aplastic anemia, chronic graft versus host disease, chronic lymphocytic leukemia, congestive heart failure, Crohn's disease, von Willebrand disease, Gaucher disease, amyotrophic lateral sclerosis, or other thrombocytopenia (not thrombotic).
Almost half of all patients (44.6%) who completed the survey reported an overall preference for receiving infusions at home, whereas 27.7% reported an overall preference for receiving treatment in a hybrid outpatient and at-home setting. Receiving treatment at home administered by either a doctor or a nurse was the most preferred option by the majority of patients (40.7%), followed by self-administration at home (32.2%). A preference for receiving treatment administered by an HCP suggests that some patients might be anxious about self-administration. Concerns about the self-administration of treatment at home has been previously reported by patients with rheumatoid arthritis, with some stating that they would prefer treatment to be administered by an HCP. 18
Patients placed the greatest utility on being comfortable with the treatment environment, followed by being able to save time if travel was not needed, and not having any concerns following treatment. These findings are consistent with those from previous studies in which a preference for receiving treatment administered at home rather than in a clinical setting was reported among some patients with chronic conditions including rheumatoid arthritis and multiple sclerosis, with convenience, comfort of the at-home setting, and the ability to avoid traveling to a hospital given as reasons for this preference.18–20 The study identified a few instances in which certain patient factors impacted attribute preferences; however, some factors had very small patient numbers, notably the number of patients with cTTP and patients who enrolled from France and the UK; therefore, findings associated with these factors should be treated with caution.
Over half of all patients were willing to trade off the maximum allowed time of 12 months to receive treatment at home and to avoid traveling to receive treatment. These findings suggest that most patients would be willing to give up a considerable portion of their lives to receive treatment in a nonclinical environment and avoid the burden associated with traveling to a hospital/clinic. A high proportion of patients were also willing to trade off 12 months to avoid complications being managed in a hospital setting.
Current treatment approaches for cTTP involve ADAMTS13 replacement with IV infusions of plasma-based therapies. 5 These therapies are associated with a high treatment burden that includes large infusion volumes and the need for administration in a clinical setting.5,7 Receiving treatment at home administered by either a doctor or a nurse was the most preferred option for 44.0% of patients with cTTP. Unlike plasma-based therapies, the at home or self-administration of rADAMTS13, under the supervision of an HCP, may be considered for patients with cTTP who are tolerant of their injections, 10 thus offering more treatment options for patient with cTTP.
The indication that many patients with chronic conditions requiring regular IV prophylaxis would prefer to receive treatment at home presents several considerations in terms of clinical practice and chronic disease management. Home-based treatment could make it easier for patients to receive care, especially for those who live in remote areas or those who have mobility issues or complex needs. Home-based infusions would also enable treatment flexibility, allowing patients to better incorporate their treatment into their everyday lives. In addition, home-based treatment may allow for more personalized care as it could help HCPs tailor treatments to the specific preferences and needs of each individual patient. This could in turn lead to better treatment adherence and improved health outcomes.
Home-based treatment may, however, make it more difficult to monitor patient treatment regimens. Responses to adverse events or complications could also be delayed for patients receiving treatment at home. Guidelines would need to be developed to support both HCPs and patients in the safe administration of infusions and the identification and management of adverse events or complications should at-home treatment become available. It should also be acknowledged that not every patient will have access to the resources necessary for home-based treatment.
The administration of treatment at home would reduce the need for frequent hospital visits which could lead to reduced healthcare costs. Home-based treatment for patients with multiple sclerosis has been shown to be less expensive than treatment administered in a hospital outpatient setting, suggesting that supporting at-home treatment may help to reduce the financial burden on healthcare systems. 21 Policies for providing wider access and insurance coverage for patients, particularly in the United States, might also be affected by the possibility of at-home treatment.
Limitations
The ability to assess the stated preferences of patients with cTTP was limited by the low diagnosed prevalence of this condition. This was minimized by extending the study to patients with other chronic conditions that require similar treatment in terms of dosing frequency and mode of administration. Future studies should aim for a larger and more targeted patient sample to enhance the generalizability of the study findings.
The study recruited fewer patients than anticipated, which may have impacted the internal and external validity of the patient subgroup findings. Many fraudulent responses were received during initial study recruitment, which may also have impacted the results; however, fraudulent responses were removed once they were identified, and several rounds of quality control reviews were conducted to check the clean data before analyses were undertaken.
The use of an anonymized online survey was intended to reduce information bias; however, self-reported patient preferences may differ from observed or actual preferences for reasons that could not be controlled for in the study. The study may also have been affected by selection bias.
There may be other treatment attributes that are important to patients that were not identified and included in the DCE survey. There is also the possibility that patients may not have related to or understood certain attributes, although this risk was minimized by using qualitative patient interviews in the development of the survey. Patients were also asked to assess the extent to which they understood the survey, with the majority reporting that they had a full understanding of the scenarios included in the DCE.
Given that this survey was focused on patients with a chronic health condition, the maximum amount of time that patients could trade off in the TTO exercise was limited to 12 months. The fact that the median time that patients were willing to trade off was 12 months for most attributes would suggest that there was a ceiling effect on the current utility estimates and that patients may have been willing to trade off more time in some cases.
Conclusion
Despite the limitations, the findings from this study provide insights into the importance of the treatment setting preferences of patients with chronic conditions receiving prophylaxis and the different trade-offs that patients would be willing to make to receive treatment at home.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735251342135 - Supplemental material for Preferences of Patients With cTTP and Other Chronic Conditions for At-Home Prophylactic Infusions: A Discrete Choice Experiment
Supplemental material, sj-docx-1-jpx-10.1177_23743735251342135 for Preferences of Patients With cTTP and Other Chronic Conditions for At-Home Prophylactic Infusions: A Discrete Choice Experiment by Laurie Batchelder, Montserrat Casamayor, Ana María Rodríguez, Spiros Tzivelekis and Shawn X. Sun in Journal of Patient Experience
Footnotes
Authors’ Note
Ethics Approval and Consent to Participate: The study was approved by the WCG independent review board. In accordance with local regulations and the ethical principles that have their origin in the principles of the Declaration of Helsinki, patients had to provide their online consent to a data protection notice before gaining access to the DCE survey. By agreeing to the data protection notice, patients consented to their data being used in the study unless they voluntarily withdrew permission. Availability of Data and Materials: The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. Sections of this manuscript were presented at the ASH 2023 Congress as a poster presentation. The ASH 2023 Congress abstract was published in Blood and is available online: ![]() . Tables 1 and 2 have been reprinted from Blood, Volume 142 (Supplement 1), Laurie Batchelder, Shawn X. Sun, Montserrat Casamayor, Ana María Rodríguez, Preferences of Patients with cTTP and Other Chronic Conditions for In-Home Prophylactic Infusions, 2366, Copyright © 2023 The American Society of Hematology with permission from Elsevier.
. Tables 1 and 2 have been reprinted from Blood, Volume 142 (Supplement 1), Laurie Batchelder, Shawn X. Sun, Montserrat Casamayor, Ana María Rodríguez, Preferences of Patients with cTTP and Other Chronic Conditions for In-Home Prophylactic Infusions, 2366, Copyright © 2023 The American Society of Hematology with permission from Elsevier.
Acknowledgments
The authors would like to thank Giulio Flore and Aleksei Komarov (both former employees at IQVIA) for their statistical support analyzing the survey data. Under the direction of the authors, medical writing support was provided by Sarah Morgan, PhD, employee of Excel Scientific Solutions, Inc. (Fairfield, CT, USA) and was funded by Takeda Development Center Americas, Inc., Cambridge, MA, USA.
Authors’ Contribution
Study conception and design: LB, AMR
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: LB, MC, and AMR are employees of IQVIA (formerly QuintilesIMS), a global provider of healthcare information and contract research services. ST is an employee of Takeda Development Center Americas Inc., and a Takeda stockholder. SXS is an employee of Takeda Development Center Americas Inc., and a Takeda stockholder.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Takeda Development Center Americas, Inc., Cambridge, MA, USA.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
