Abstract
Background:
Conventional injectable glucagon (IG) and nasal glucagon (NG), both having similar efficacy, are two options for the emergency treatment of severe hypoglycemia in Spain. This study elicited the effect of changes in key attributes on preferences for NG and IG medication profiles of people with diabetes and caregivers in Spain.
Methods:
The relative attribute importance (RAI) that participants placed on glucagon preparation, preparation and administration time, delivery method, recovery time, device size, storage temperature, and headache risk was estimated from an online discrete choice experiment. In addition, patients and caregivers were presented with NG and IG profiles that included rates of successful administration; the proportion of participants choosing each profile was summarized.
Results:
The analysis included 276 adults with diabetes (65% type 1) and 270 caregivers (49% type 1). Overall mean age was 40 years; 51% were female. The most important attributes were storage temperature (RAI [95% confidence interval] = 27.3% [22.9-32.2]) and delivery method (17.4% [13.1-21.9]). Headache risk (16.2% [11.8-20.7]), time to prepare and administer (14.5% [10.1-18.8]), glucagon preparation (11.4% [6.8-15.8]), recovery time (8.9% [4.3-13.3]), and device size (4.3% [0.3-8.8]) were also relevant. When comparing medication profiles, significantly more participants (78%) preferred NG over IG profiles (P < .001).
Conclusion:
Adults with diabetes and caregivers prefer a glucagon treatment with a higher rate of successful administration, wider storage temperature, and nasal delivery method, when efficacy is similar. Participants favored NG over conventional IG as a rescue medication for severe hypoglycemia. This information may help decision-making by payers and treatment discussions between health care professionals and patients.
Introduction
People with type 1 diabetes mellitus (T1DM) and insulin-treated type 2 diabetes mellitus (T2DM) in Spain reported experiencing severe hypoglycemia approximately once per year. 1 Previous estimates indicate the total annual cost of severe hypoglycemia to the Spanish health care system was more than six million Euros or approximately €1200 per event. 2 Although most events are managed by caregivers outside of the health care system, the burden to both the person with diabetes and their caregivers does impact their psychosocial health including feelings of unpreparedness and helplessness.3,4
Treatment guidelines recommend that glucagon be administered to people experiencing severe hypoglycemia who are unable or unwilling to consume oral carbohydrates.3,5-7 Conventional injectable glucagon (IG), which requires reconstitution using a multistep process, has been shown to be fraught with preparation errors that can lead to administration failure.8-10 To address this, there have been significant improvements in the technology of glucagon options in recent years. Advancements have led to the development of nasal glucagon (NG), a ready-to-use (ie, no reconstitution) medication for the treatment of severe hypoglycemia. 11 NG and IG have demonstrated similar efficacy in resolving insulin-induced hypoglycemia events with consistent safety profiles, aside from local side effects associated with nasal administration (eg, lacrimation) or with injection (eg, injection site reactions).12-16
With such similarities, several different studies, conducted for other purposes, have queried on preference, showing that NG was preferred over IG,9,10,16-18 but their limitation is that the drivers of participants’ overall preference remain unknown. In this study among a Spanish population, we examined how the preferences for glucagon treatments with similar efficacy are driven by preferences for key attributes.
Methods
Overall Study Design
Adults with diabetes and caregivers of people with diabetes in Spain completed an online survey, a discrete choice experiment (DCE), between June and August 2020. DCEs are designed to estimate the effects of important and relevant attributes on participants’ preferences and how they affect their treatment choices. This DCE was designed to elicit preferences for key attributes (ie, variables) of glucagon for the treatment of severe hypoglycemia. The attributes and levels for this study were selected to represent the real options that patients have available to them currently. The attributes which differentiate the current rescue medication options, and the levels which define these options therefore were key drivers of the study design. An overview of the study is provided in Figure 1.
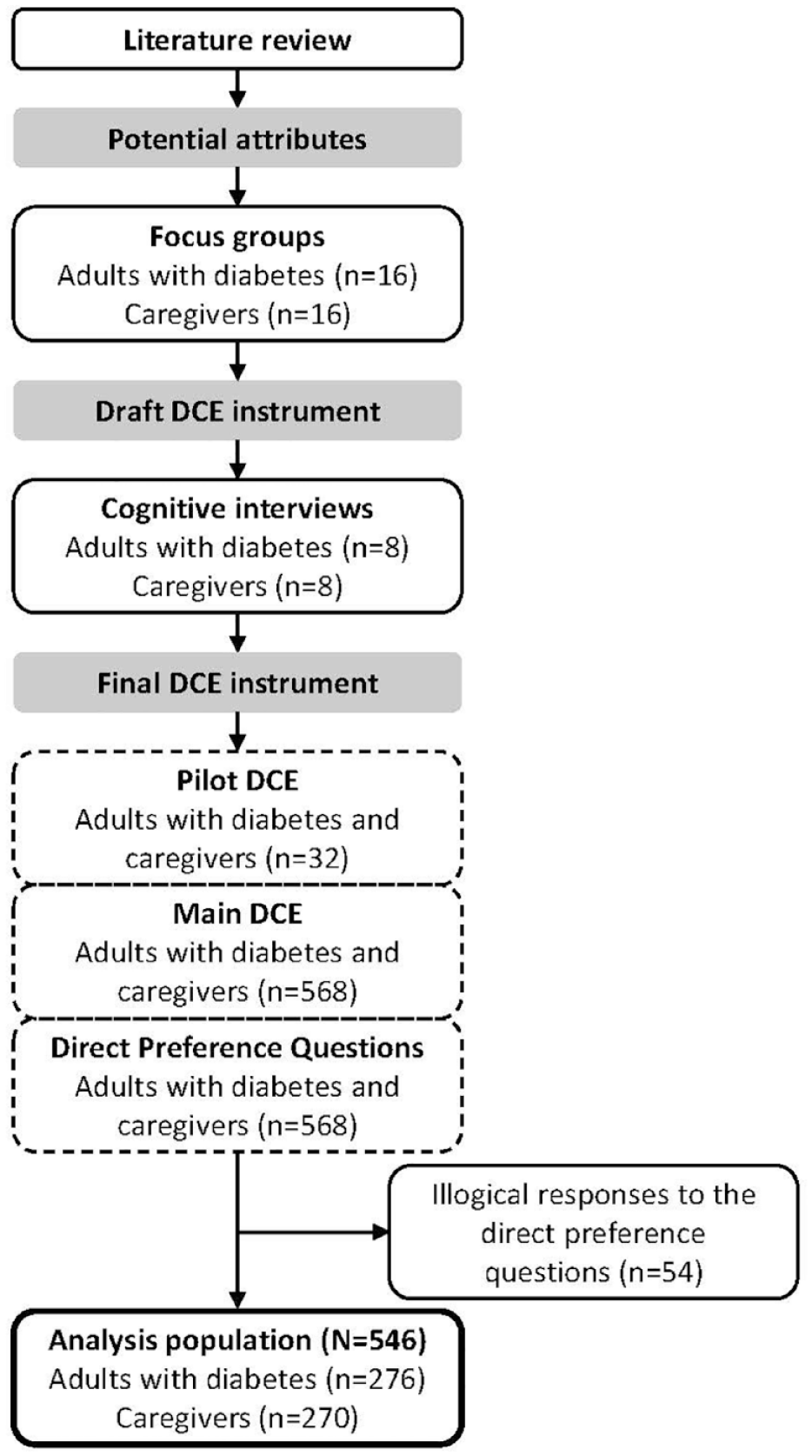
Overview of the study design.
Step 1. Literature review to identify potential attributes
Potential attributes to be included in the DCE were identified through a review of the published literature to ensure key attributes were included. In addition, the prescribing information and instructions for use for glucagon were reviewed. These included (1) glucagon preparation, (2) preparation and administration time, (3) delivery method, (4) recovery time (recover to stable blood sugar/glucose), (5) device size, (6) storage temperature, and (7) risk of headache.
Step 2. Focus groups to assess potential attributes
The importance, relevance, and completeness of these identified attributes were assessed through four inperson focus groups conducted in October 2019 at two sites (Madrid and Oviedo) in Spain. Two focus groups included adults with diabetes taking insulin (n = 16), and two included caregivers of people with diabetes taking insulin (n = 16). Participants described their experiences with severe hypoglycemic events and use of rescue medication. Characteristics of glucagon identified from the literature review were then introduced to the focus groups by the moderator, and each participant ranked the characteristics in order of importance. The initial list of treatment attributes was then revised based on the focus group discussions.
Step 3. Interviews to test attributes for relevance and understandability
The revised treatment attributes were used to create DCE choice tasks that asked participants to select between two hypothetical rescue medications and implicitly make trade-offs between the attributes of these treatment options. Levels (ie, attribute variations) were based on the actual medication profiles of NG and IG.10,19-21 The DCE was tested in interviews with adults with diabetes (n = 8) and caregivers (n = 8) to determine whether the attributes and levels were understandable and relevant. 22
Step 4. Pilot DCE to test online survey
After further refinements, the online DCE survey was piloted in 32 participants to confirm data quality. The final attributes included in the pilot DCE were glucagon preparation steps (need to mix versus ready-to-use), time to prepare and administer, delivery method (injection versus nasal administration), recovery time, device size, storage temperature, and headache risk (Table 1). No changes were made based on the results from the pilot survey.
Attributes and Levels Included in the Discrete Choice Experiment.
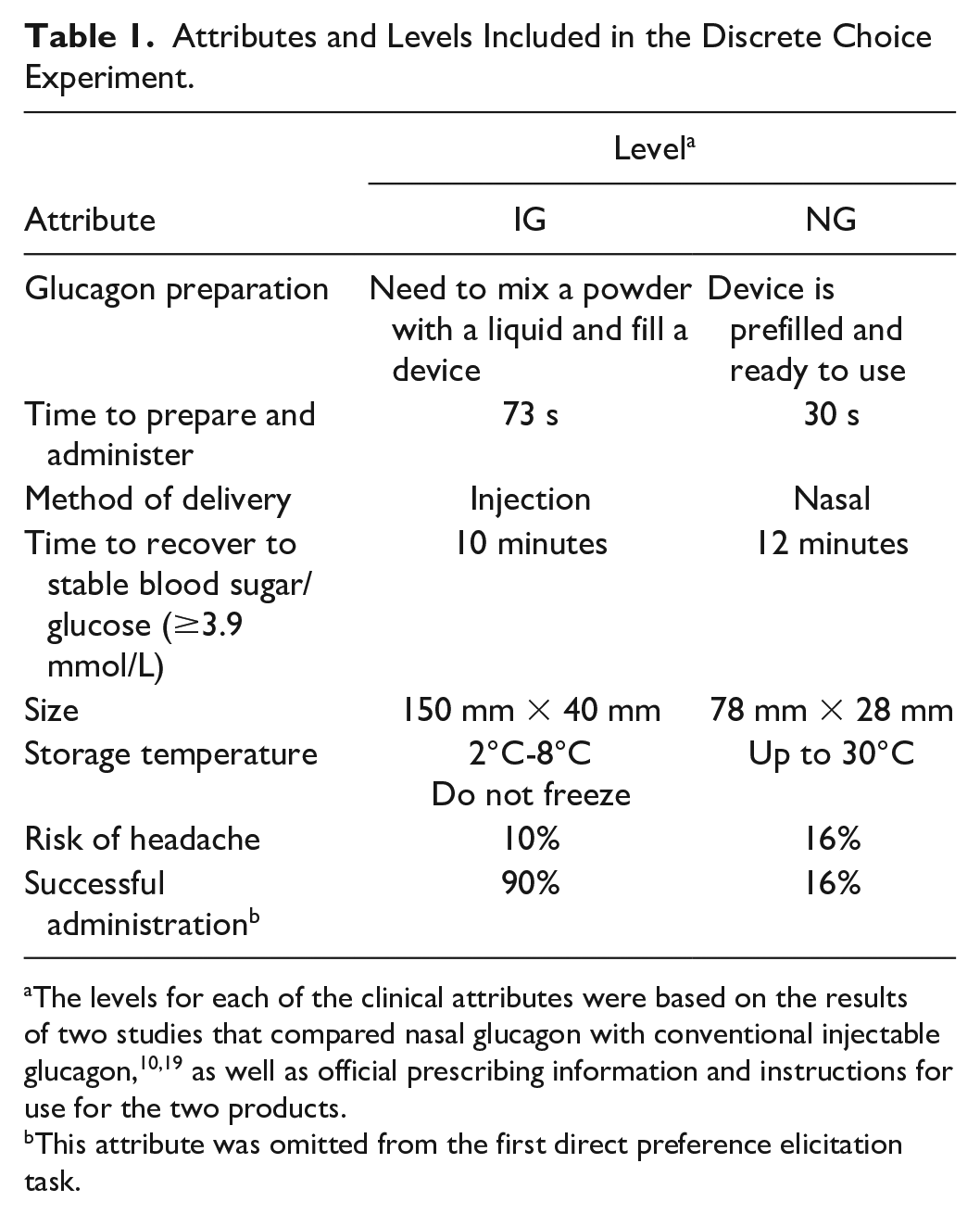
The levels for each of the clinical attributes were based on the results of two studies that compared nasal glucagon with conventional injectable glucagon,10,19 as well as official prescribing information and instructions for use for the two products.
This attribute was omitted from the first direct preference elicitation task.
Step 5. Main study
The main DCE survey was then conducted with a planned enrollment of 300 people with diabetes and 300 caregivers. The survey provided detailed descriptions of severe hypoglycemia and its treatment with glucagon, followed by the different treatment attributes and levels (Table 1).
As a full combination of the attributes’ levels would have led to a very large number of choice tasks, Ngene software 23 was used to obtain a d-efficient design in 16 experimental tasks. These 16 tasks were divided into two blocks such that each participant completed eight experimental choice tasks. Each task offered a choice between two hypothetical treatment options (Figure 2 shows an example DCE choice task). Participants were also asked to complete two nonexperimental choice tasks, a repeat question and a dominance question in which one treatment option was superior, to assess whether participants were actively engaged in the survey. The order of the experimental choice tasks was randomized between participants to mitigate potential ordering effects. 24
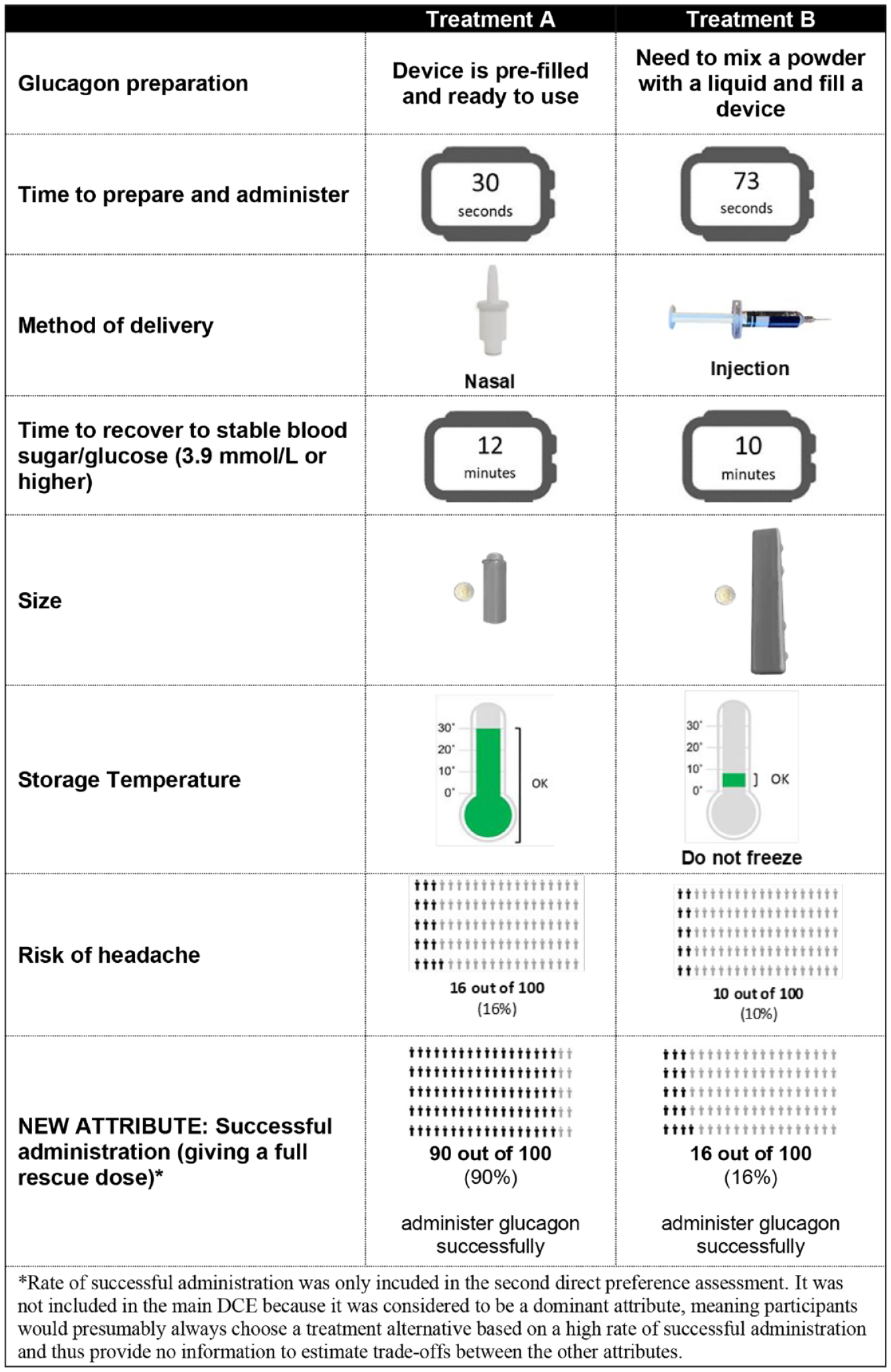
Example discrete choice experiment choice task.
Data from the nonexperimental tasks were not included in the data analysis.
Step 6. Glucagon medication profiles comparison
In addition to the DCE, participants completed two direct preference questions asking them to choose between the medication profiles for NG and IG (Table 1). The first direct preference question was presented in the same way as the experimental choice tasks and was indistinguishable from the other DCE questions. The second direct preference question included the rate of successful administration of a full dose as an additional attribute, with the following levels: 90% for NG and 16% for IG. 10 This attribute was not included in the main DCE because it was considered to be a dominant attribute, meaning participants would presumably always choose a treatment alternative based on a high rate of successful administration and thus provide no information to estimate trade-offs between the other attributes.
Step 7. Participant characteristics assessed
All participants also completed three validated questions to assess health literacy (each scored on a 5-point scale from 0 to 4) 25 and five validated questions testing numeracy skills. 26 Participants were considered to have adequate health literacy if their average literacy score was above 2 and adequate numeracy if they correctly answered three or more of the numeracy questions. In addition, people with diabetes completed questionnaires collecting information on sociodemographics, clinical characteristics, and experience with diabetes and its treatment. Caregivers completed questionnaires about the person they cared for and their experience witnessing and treating severe hypoglycemic events.
Participants
Participants were adults (aged ≥18 years) who either had a self-reported diagnosis of T1DM or T2DM and were taking insulin or who reported being a caregiver of a person with T1DM or T2DM (aged ≥4 years) taking insulin. Caregivers had to be living with or be the primary medical emergency contact for the person with diabetes. Potential participants were excluded if they or the person they cared for had participated in an earlier phase of the study.
Participants in the focus groups and interviews were recruited through clinical referrals, flyers, advertisements, and patient associations. Participants in the online DCE survey were recruited by a third-party vendor’s patient database and patient associations. Potential participants were sent an email and invited to complete an online screening questionnaire. Eligible participants who provided consent were redirected to the survey. Participants received non-coercive remuneration for their time.
All participants provided informed consent. The study was conducted in accordance with the Declaration of Helsinki, International Council for Harmonisation Good Clinical Practice, national laws, and the European Union General Data Protection Regulation. The study was submitted to the ethics committee of Gregorio Marañón Hospital (Madrid, Spain), and the ethics committee judged that no approval was required.
Statistical Analysis
Statistical analyses were performed using R version 3.6.1 (R Foundation for Statistical Computing, Vienna). Data from the pilot and main DCE surveys were pooled because the surveys were identical.
For descriptive sample characteristics, categorical variables (eg, gender) were summarized by frequency statistics (N [%]). Continuous variables (eg, age) were summarized using means and standard deviations (SD).
Responses to the main DCE were analyzed using a multinomial logit (MNL) model. 27 The attributes’ levels entered the MNL model as dummy-coded variables such that the model estimated the effect of discrete changes in the treatment (eg, effect of decreasing headache risk from 16% to 10%). The model estimates were then used to calculate relative attribute importance (RAI) or the importance of each attribute relative to the others 28 and participant’s predicted choice probabilities. Participants’ predicted choice probabilities, the probabilities of hypothetical treatments being accepted by participants, were computed for their preferred treatment and estimated part-worth utilities from the MNL model. More details about the statistical analysis are provided in the Supplemental Methods. The choice data were separately analyzed for the patient and caregiver samples.
Percentages of participants choosing NG and IG in response to the direct preference questions were analyzed with a chi-square test.
Results
Participants
Between June and August 2020, 2,944 adults with diabetes or caregivers of people with diabetes were screened for eligibility, 643 were eligible and provided consent, and 600 (301 people with diabetes and 299 caregivers) completed the online DCE survey. Twenty-five adults with diabetes and 29 caregivers were excluded because they provided inconsistent responses to the direct preference questions, indicating participants were making illogical selections. The main analysis population included 546 participants (276 adults with diabetes and 270 caregivers).
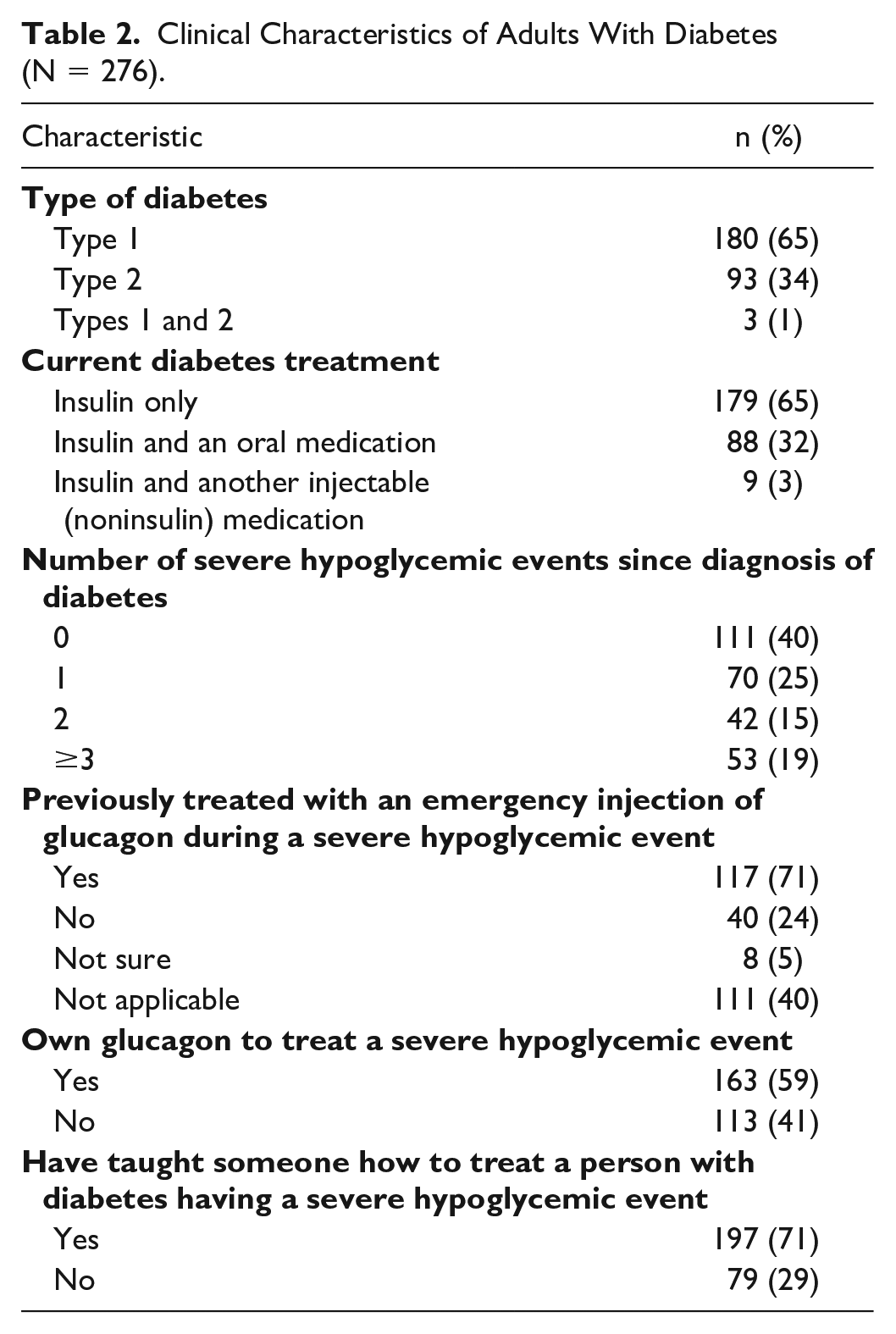
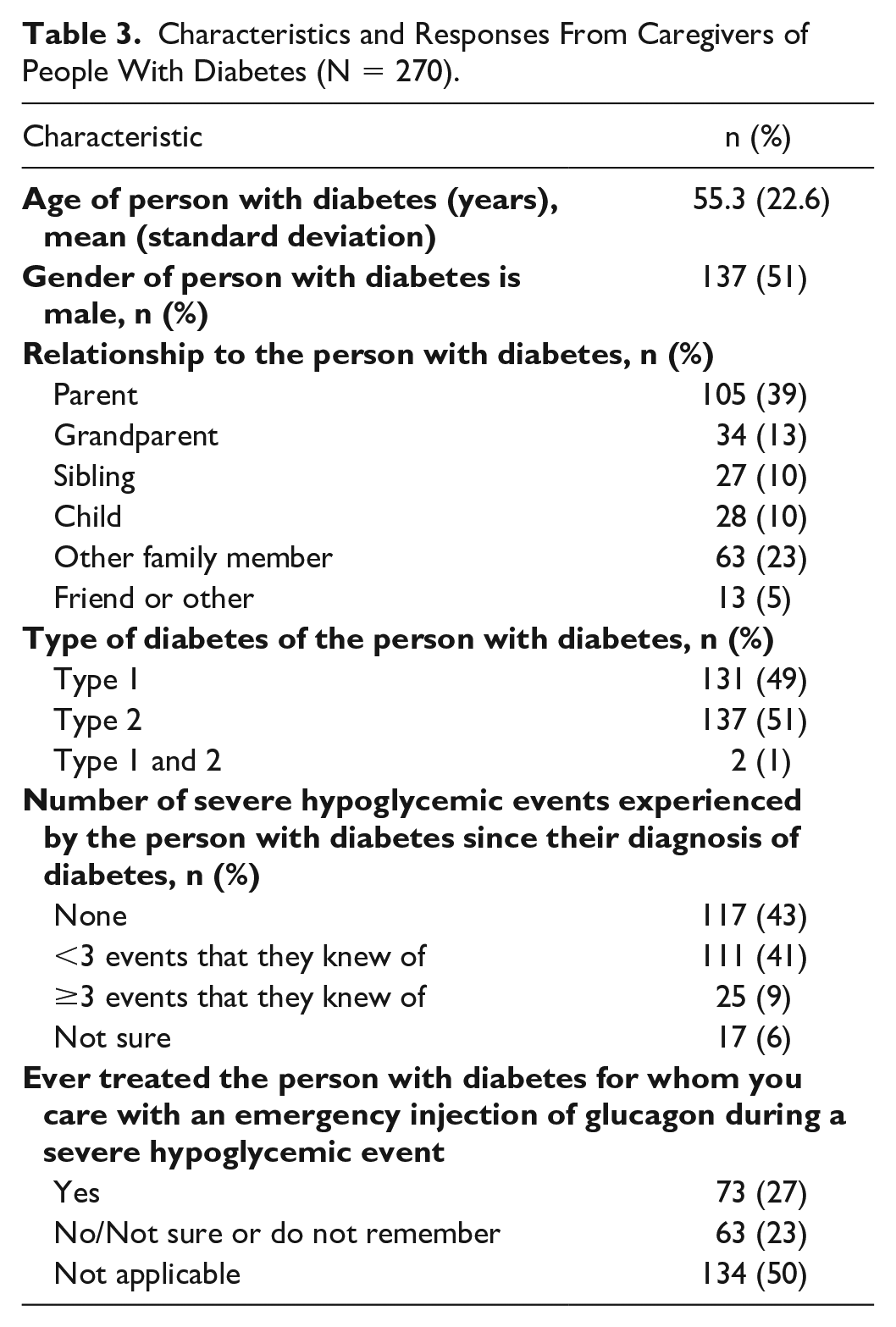
The mean age of participants was 40.3 years, 51% were female, and 58% had completed a university education (Supplemental Table 1). Most (65%) of the adults with diabetes had T1DM (Table 2), whereas caregivers reported caring for approximately equal proportions of people with T1DM (49%) and T2DM (51%) (Table 3). Sixty percent of adults with diabetes and 50% of caregivers reported at least one severe hypoglycemic event since diagnosis of diabetes; 19% of adults with diabetes and 9% of caregivers reported greater than or equal to three severe hypoglycemic events since diagnosis. Of the adults with diabetes who reported experiencing a severe hypoglycemic event in the past, 71% reported receiving an emergency injection of glucagon as a result. Fifty-nine percent of caregivers reported ever treating someone with diabetes with an emergency injection of glucagon during a severe hypoglycemic event.
Clinical Characteristics of Adults With Diabetes (N = 276).
Characteristics and Responses From Caregivers of People With Diabetes (N = 270).
Data Quality of the DCE
Overall, 83% of participants had adequate health literacy and 89% had adequate numeracy. Most participants chose the same answers to the repeat question (79%), passed the dominance test (77%), and did not make decisions based solely on a single attribute (65%). The dominance pass rate is similar to other published literature. 29 Only 2% of participants always chose treatment A or always chose treatment B. These data indicate that most participants understood the survey content.
Relative Attributes of Importance and Part Worth Utilities
Storage temperature was the overall highest ranked attribute (RAI [95% confidence interval, CI] = 27.3% [22.9-32.2], followed by delivery method (17.4% [13.1-21.9]), and then headache risk (16.2% [11.8-20.7]) (Figure 3). Device size was the least important attribute overall (4.3% [0.3-8.8]). While storage temperature was the highest ranked attribute for both adults with diabetes and caregivers, the second highest attribute was delivery method for adults with diabetes and headache risk for caregivers (Supplemental Table 2).
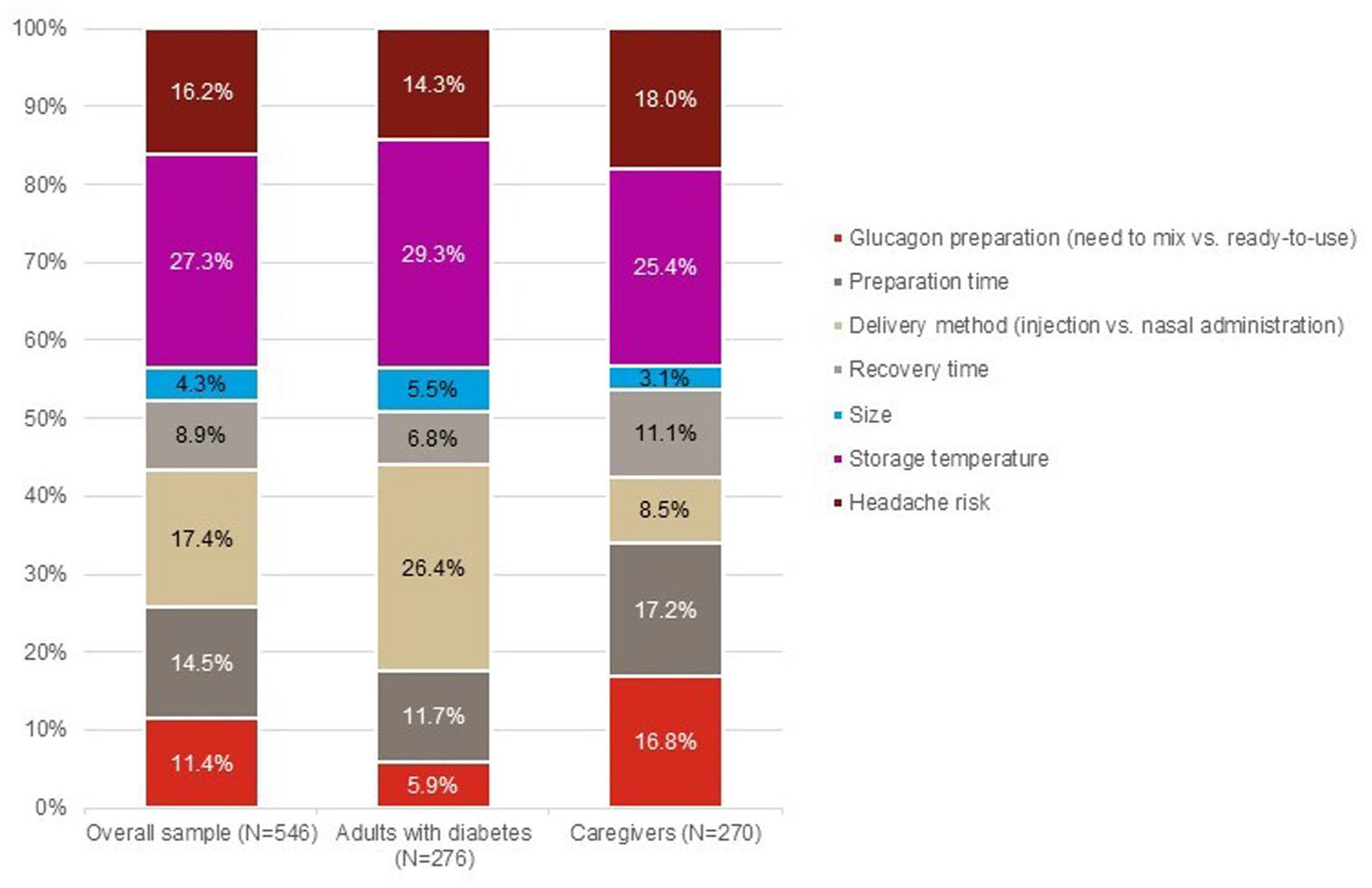
Relative attribute importance in the main sample, adults with diabetes, and caregivers.
Based on part-worth utilities, overall, participants significantly preferred a wider storage temperature range, a lower risk of headache, shorter preparation time, a ready-to-use glucagon, shorter recovery time, and a nasal delivery (all P < .001; Figure 4).
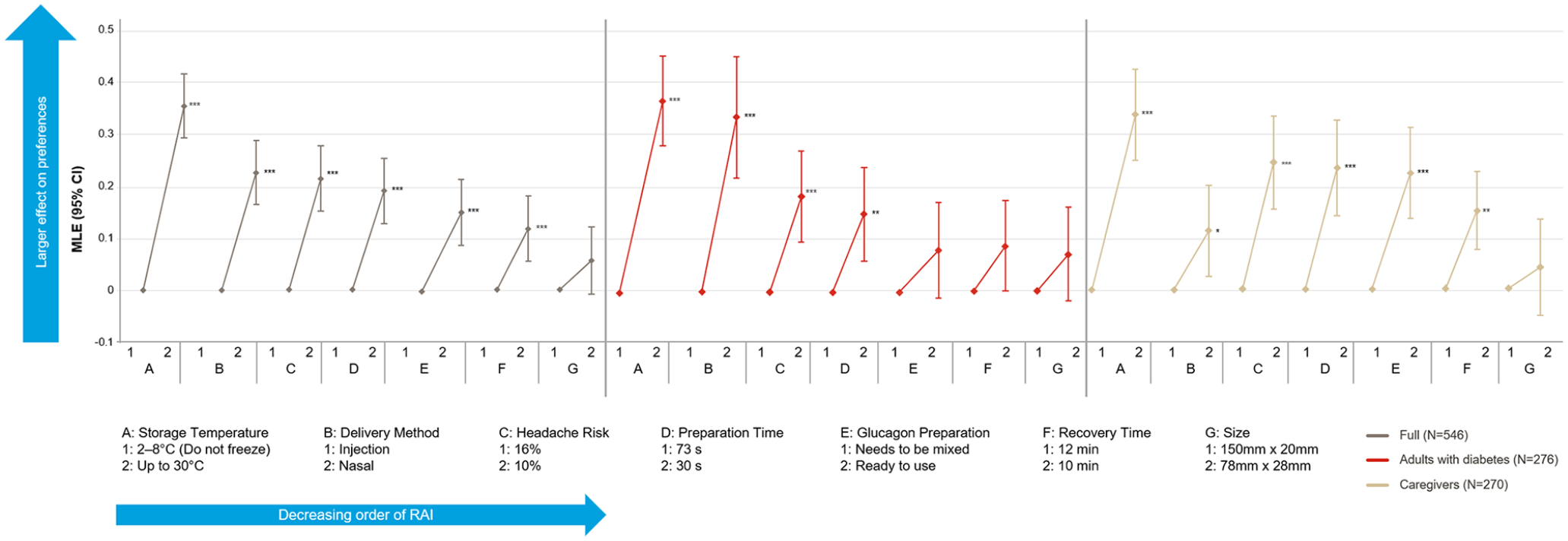
Part-worth utilities (N = 546).
Predicted Choice Probability
A nasal rescue medication was predicted to be preferred over an injectable rescue medication by adults with diabetes (probability [95% CI] = 67.6% [62.1-72.8]) and caregivers (63.5% [57.6-68.8]).
Preferences for Medication Profiles of Nasal versus IG
When directly comparing the NG or IG medication profiles including the rate of successful administration, 78.0% of participants (79.3% adults with diabetes and 76.7% of caregivers) preferred NG and 22.0% preferred IG (P < .001) (Figure 5). When the rate of successful administration was not considered, NG was still preferred over IG by 62.6% of participants (64.9% of adults with diabetes and 60.4% of caregivers) (P < .001).
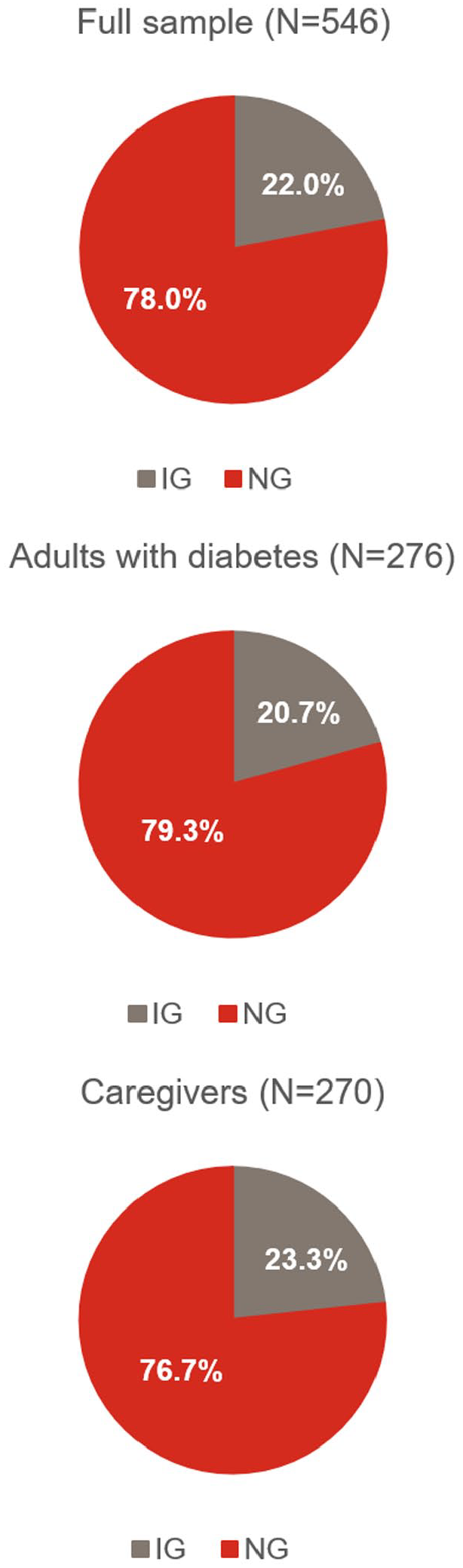
Direct preferences for treatment profiles of injectable and nasal glucagon.
Discussion
As the first DCE between NG and IG, the study provides insight into the preferences of adults with diabetes and caregivers of people with diabetes. Overall, storage temperature and delivery method were the most important attributes influencing preferences, although, delivery method was more important to adults with diabetes than to caregivers. Based on the preferences elicited in the DCE, approximately two-thirds of adults with diabetes and caregivers were predicted to prefer NG to conventional IG due to the wider storage temperature range, a preference for nasal delivery, and the faster preparation and administration time. Similarly, direct preference questions indicated a preference for NG over IG when participants directly compared the two medication profiles.
Our study is the first to assess glucagon treatment attributes of utmost importance, as defined by adults with diabetes and caregivers, using the discrete choice methodology, which is widely accepted to assess preference in the scientific community. The results are consistent with findings from previous literature, which suggest that participants prefer NG.9,10,16 Importantly, the current work provides information about the relative importance of included attributes, informing the drivers of overall preference. The agreement between the predicted choice probabilities from the DCE and preferences from the glucagon medication profiles supports the conclusion that most adults with diabetes and caregivers prefer NG over IG to treat severe hypoglycemia. According to the clinical guidelines, providers should make decisions “collaboratively with patients based on individual preferences, prognoses, and comorbidities.”30,31 The patient voice heard through DCEs can be complementary to safety and efficacy data for informed decision-making by patients, providers, and payers.
The current study found that wider storage temperature range was the most important attribute for both adults with diabetes and caregivers. This suggests that both groups value a rescue medication with a less restrictive temperature range, enabling them to carry it with them as part of day-to-day life, so it is available for use in case of an emergency. According to the product labels in Spain, NG can be stored up to 30°C, 11 and IG should be stored at 2°C to 8°C (in a refrigerator; do not freeze), which may impact the portability of these two products. 32
Delivery method (nasal versus injectable) was also an important attribute influencing the preferences of adults with diabetes more so than compared with caregivers, which may reflect patient concerns about the ability of caregivers to properly administer an injection in an emergency.9,33,34
DCE studies have inherent limitations, which warrant attention. First, the number of attributes was limited to reduce the cognitive burden on study participants but did include the attributes of utmost importance from the literature and focus groups. Furthermore, while treatment costs may play an important role in the participants’ preferences, it did not come up in the focus groups as most important and the final cost for NG in Spain was unknown at the time of the DCE. Therefore, cost was not included as an attribute in this study. In addition, the most common side effects of glucagon treatment are nausea and vomiting. However, as the incidence of nausea and vomiting is similar between NG and IG, 14 these attributes would not have led to trade-offs or informed preference. Therefore, they were not included in the study. Also, adults with diabetes (71%) and caregivers (59%) in this study reported a higher rate of past experience with glucagon compared with Spanish participants from a survey in which only 29% of people with insulin-treated diabetes and 40% of caregivers of people treated with insulin reported treating their most recent severe hypoglycemic event (ie, within the past 3 years) with IG. 1 However, because this study focused specifically on glucagon rather than generally on severe hypoglycemia, people with diabetes who have experienced severe hypoglycemia and used glucagon previously have a personal interest in glucagon treatment and may have self-selected to participate. Therefore, the generalizability of these results to the general Spanish population may merit further investigation. Furthermore, these results may not be generalizable outside Spain. Moreover, while the present study examined the preferences of glucagon treatments that were available at the time the study was conducted, future studies are warranted to compare the preferences of other recently available ready-to-use glucagon options. It would be of particular interest to examine how the storage temperature restrictions of these treatments will be regarded by the end users.35,36
Conclusions
This study offers important insight into the preferences of both adults with diabetes and caregivers for features of rescue medications for severe hypoglycemia. These results demonstrate that when efficacy is similar, participants conveyed preferences for a rescue medication with a wider storage temperature, nasal delivery, and high likelihood of successful administration, informing the drivers of overall patient preference. Temperature storage conditions are important as they may impact the effectiveness of the medication and the likelihood of a person taking glucagon with them throughout their day-to-day activities. These results may help in treatment discussions between health care professionals and their patients, focusing on an individual’s specific situation, needs, and preferences as they consider available glucagon treatment options.
Supplemental Material
sj-docx-1-dst-10.1177_19322968221095882 – Supplemental material for People With Diabetes and Caregivers Prefer Rescue Glucagon Treatment With a Wider Storage Temperature Range and a Nasal Administration, When Efficacy is Similar: A Discrete Choice Experiment in Spain
Supplemental material, sj-docx-1-dst-10.1177_19322968221095882 for People With Diabetes and Caregivers Prefer Rescue Glucagon Treatment With a Wider Storage Temperature Range and a Nasal Administration, When Efficacy is Similar: A Discrete Choice Experiment in Spain by Beth D. Mitchell, Anne M. Rentz, Shannon Kummer, Yu Yan, Sebastian Heidenreich, Nicolas Krucien, Esther Artime, Beatrice Osumili, Miriam Rubio and Heather L. Gelhorn in Journal of Diabetes Science and Technology
Footnotes
Acknowledgements
The authors would like to thank Gin Nie Chua, Andrea Schulz, David Mott, Marissa Walsh, Melissa Garcia, Melissa Ross, Myrto Trapali, Robyn Cyr, and Rodolfo Matos for their assistance in executing the study, and Fritz Hamme and Emily Sargent for providing editing and production services in the development of the manuscript (all employed by Evidera at the time of the study). This work was conducted by Evidera, an independent research organization and received research study support from Eli Lilly. Medical writing was provided by Stephen Gilliver, PhD and Phillip S. Leventhal, PhD (Evidera) and Kristen Syring, PhD (Eli Lilly and Company) and was paid for by Eli Lilly and Company.
Author Contributions
BDM and YY were involved in the conception and design of the project, data interpretation, as well as the critical review, revision, and critic of the manuscript. AMR and SK were involved in the conception, design, and execution of the project, as well as the critical review, revision, and critic of the manuscript, and data analysis design and interpretation. AMR drafted the manuscript. SH, EA, BO, MR, and HLG were involved in the design of the project as well as the review and critic of the manuscript. SH and HLG were involved in the interpretation of the data analysis. NK was involved in the design, execution, and interpretation of the data analysis and review and critic of the manuscript.
Abbreviations
DCE, discrete choice experiment; IG, injectable glucagon; MNL, multinomial logit; NG, nasal glucagon; RAI, relative attribute importance; T1DM, type 1 diabetes mellitus; T2DM, type 2 diabetes mellitus.
Consent for Publication
There were no details, images, or videos relating to individual participants which were included in the article. All data were de-identified before data analysis and reporting.
Data Availability
Data are not publicly available but may be provided upon request.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The study sponsor was involved in the design of the study and analysis, interpretation of the data, and review and approval of the manuscript. Anne M. Rentz, Sebastian Heidenreich, Nicolas Krucien, and Heather L. Gelhorn are employed by Evidera, which provides consulting and other research services to pharmaceutical, device, government, and nongovernment organizations. As Evidera employees, they work with a variety of companies and organizations and are expressly prohibited from receiving any payment or honoraria directly from these organizations for services rendered.
Shannon Kummer was an employee of Evidera at the time of the study.
Beth Mitchell, Yu Yan, Esther Artime, Beatrice Osumili, and Miriam Rubio are employed by and own stock in Eli Lilly and Company.
The authors report no other conflicts of interest in this work.
All aspects of the study design, interpretation, and decision to submit for publication were determined by the authors.
Ethics Approval and Informed Consent
The study was submitted to the ethics committee of Gregorio Marañón Hospital (Madrid, Spain), which judged that it did not need their approval. Participants in the focus groups provided written informed consent; participants in the cognitive interviews and pilot and main DCE survey provided online consent.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Eli Lilly and Company.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
