Abstract
Bereavement, the experience of losing a loved one, significantly affects emotional, psychological, and physical wellbeing. Assessing the quality of end-of-life care and the bereavement experiences of family members is crucial for improving healthcare services. The CANHELP Lite Bereavement Questionnaire, a validated tool for evaluating end-of-life care from bereaved family members’ perspectives, needs cultural adaptation for non-English-speaking contexts. This study aimed to adapt and validate the Arabic version of the CANHELP Lite Bereavement Questionnaire for Arabic-speaking populations, considering their unique cultural, social, and religious contexts. The adaptation process involved systematic translation, back-translation, and expert evaluation to ensure cultural sensitivity and linguistic appropriateness. The psychometric properties of the adapted questionnaire were tested among 269 bereaved Saudi family members, revealing a Cronbach's alpha of 0.950 for internal consistency reliability. Face and content validity assessments confirmed item relevance, and concurrent validity was established with a Pearson correlation coefficient of 0.886. The study concluded that the Arabic version is a valid and reliable instrument for assessing satisfaction with end-of-life care, aiding in identifying areas for improvement in bereavement support within Arabic-speaking populations and enhancing healthcare services in culturally diverse settings.
Introduction
Bereavement, the experience of losing a loved one, is a profound and multifaceted process that significantly impacts an individual's emotional, psychological, and physical wellbeing. 1 Assessing the quality of end-of-life care and the subsequent bereavement experiences of family members is crucial for improving healthcare services. 2 The Canadian Health Care Evaluation Project (CANHELP) Lite Bereavement Questionnaire is a validated tool designed to measure the quality of end-of-life care from the perspective of bereaved family members. It provides valuable insights into areas requiring enhancement to better support those facing the loss of a loved one.3,4
However, the CANHELP Lite Bereavement Questionnaire was originally developed and validated in English-speaking populations. To ensure its applicability and accuracy in different cultural contexts, it is essential to adapt and validate the instrument for use in other languages and cultures. 5 Cultural adaptation involves not only translating the questionnaire but also modifying it to reflect cultural nuances, beliefs, and practices related to end-of-life care and bereavement. This process helps in maintaining the validity and reliability of the instrument in the new cultural setting. 6
The Arab world, with its unique cultural, social, and religious contexts, necessitates the adaptation of the CANHELP Lite Bereavement Questionnaire to Arabic. Arabic-speaking populations may have distinct perspectives on death, dying, and bereavement, influenced by Islamic teachings and cultural practices. 7 Ensuring that the questionnaire is culturally sensitive and linguistically appropriate for Arabic speakers is essential for accurately capturing their bereavement experiences and evaluating the quality of end-of-life care they received. 8
This study aims to culturally adapt and validate the Arabic version of the CANHELP Lite Bereavement Questionnaire. The process involves a systematic translation, back-translation, and evaluation by a panel of experts to ensure cultural relevance and comprehensibility. 9 Additionally, the study will test the psychometric properties of the adapted questionnaire, including its reliability and validity, within Arabic-speaking populations. 10 The psychometric properties refer to the characteristics and qualities of psychological tests and assessments that determine their quality, reliability, and validity. By achieving this, healthcare providers can obtain a reliable tool to assess and improve end-of-life care and bereavement support for Arabic-speaking families, ultimately enhancing the quality of healthcare services in culturally diverse settings. 11
Method
Study Design
This was a methodological study including translation, semantic, and cultural adaptation, and the evaluation of the psychometric properties, according to the guidelines by Beaton et al. 12
Translation, Cross-Cultural Adaptation, and Validation Process
The process of cultural adaptation looks at both language (translation) and cultural adaptation issues, while preparing a questionnaire for use in another setting. The following stages composed the translation and semantic and cultural adaptation of the instrument.
12
Linguistic translation of the instrument into Arabic language by 3 translators. Two translators were unaware of the concept under study, but the third was expert in the palliative care filed. Synthesis and review of the first translation. Back-translation of the reviewed version by 2 other translators, both of them fluent in English and their mother tongue is Arabic language. They did the semantic analysis and had an agreement result of 100%. Review of all translations by a panel of experts (1 expert in research and methodological studies, 2 experts in palliative care, and 1 expert in bereavement, in addition to all translators and back-translators). The original developers of the questionnaire were in close contact with the panel of experts during this part of the process. Assessment of face and content validity through a pretest on a sample of 25 bereaved family members, which aimed to verify the understanding of the items, evaluate the ease of using the response set, and estimate the response time. The instrument was easily understood, and the bereaved family members answered when requested. The average answer time was 10 min for properties were examined. The literature proposes the inclusion 5 to 10 participants per item for the sample (Pais-Ribeiro 2010). The inclusion of 10 participants per item was estimated and the final sample was composed of 269 bereaved family members.
Participants, Setting, and Procedure
The participants were bereaved family members knowledgeable about the healthcare provided during the past 3 months of life to an adult decedent (aged 18 years and older). To identify potential participants, the death certificates of all adults (aged 18 years and older) that were available in the national electronic death certificate registration were analyzed. Records of decedents where the cause of death codes were associated with external or accidental causes, suicides, pregnancy complications, or medical and surgical complications, as well as death certificates that had uncertain cause of death were excluded.
In Saudi Arabia, death certificates do not have information regarding family members and thus, it was necessary to cross information and to search for a phone number on the health system national registration using the health information registration number provided on the death certificate. A first phone call was made to inform the participants about the study's aims and the voluntary nature of their participation. They were also given guarantees of data confidentiality and anonymity. Informed consent was obtained from each bereaved family member. After that, a phone interview was scheduled at a date of the bereaved family members’ preference. Subsequently, the instrument was applied through a phone interview conducted by trained interviews. All interviews were conducted 3 to 12 months after death. As the period of bereavement is recognized as a sensitive time, this interval was decided to respect the family bereavement period but also to guarantee that the memory of the experience was recent enough to be considered reliable.
Data Analysis
Descriptive analysis was performed through means and standard deviation (SD), or numbers and percentages of the demographic and clinical variables. The Kolmogorov-Smirnov test was performed to check the distribution of the variables enrolled. A bivariate correlation by Pearson was used.
The assumptions used in the original version were followed and the internal consistency of the scale was obtained using Cronbach's alpha. Cronbach's alpha if the item deleted and corrected item-total correlation were also obtained. According to the literature, alpha values under 0.50 are unacceptable, from 0.50 to 0.60 are questionable, from 0.60 to 0.70 are acceptable, from 0.70 to 0.80 are good, from 0.80 to 0.90 are very good, and over 0.90 are considered excellent.13–15
Criterion-related validity was determined through the concurrent use of a single-item, global rating of caregivers’ general satisfaction with the care received in the past 3 months of life (scored between 1 and 5, meaning that 1 was the worst level of satisfaction possible and 5 the best level of satisfaction possible). This option is justified by the fact that there isn't a validated tool in Arabic to assess bereaved family members’ satisfaction with the care received in the last months of life regardless of the existing pathology or setting of care. This was performed using the Pearson correlation coefficient.
Convergent-discriminant validity was performed using an exploratory factorial analysis considering the principal component method with orthogonal rotation (varimax method with Kaiser normalization). The exploratory factor analysis is a method to explore the underlying structure of a set of observed variables, and is a crucial step in the scale development process.
It was not possible to perform test-retest reliability, as the purpose of the instrument was to assess the bereaved families’ satisfaction with the care received in the past 3 months of life. As this might be considered a sensitive matter, we believe that respecting human dignity should have priority over the interests of the research.
The data were analyzed using SPSS, version 23, for Windows. A level of significance of 0.05 was assumed.
Ethical Procedures
A research protocol was designed. It was then submitted, analyzed, and approved by the Health Data Protection Authority and Research Ethics Committee in Saudi Ministry of Health (approval no. 5274/1401).
Result
Study Population
All 269 participants were bereaved Saudi family members from the center region recruited through the identification provided in the health system national registration after analysis of the death certificates. The majority of the participants were female (78.4%) and married (59.5%), and were most often daughters of the deceased (47.6%). The average age was 58.2 years (SD = 13.3 years; minimum = 18 years and maximum = 91 years). The interview was conducted, on average, 295.93 days after the death of the relative (SD = 76.6 days; minimum = 93 days and maximum = 364 days).
Reliability
Considering the 24 items of the scale, the analysis of internal consistency reliability revealed an alpha of 0.950. The global alpha when removing items were also analyzed (Table 1). Although Cronbach's alpha would increase if items 14, 16, and 23 were deleted, it was decided to retain them, as the alpha obtained is already considered excellent.
Descriptive Statistics and Internal Consistency for the Items.

Construct Validity
During the translation and cultural adaptation phase, it was observed that the scale was answered quickly and that there were no suggestions in items or terms considered inadequate or unnecessary, which shows a good understanding and acceptance of the items by the participants.
Concurrent validity was determined using the Pearson correlation coefficient, and a value of 0.886 (P < .001) was obtained.
Regarding convergent—discriminant validity, an exploratory factorial analysis considering the principal components method with orthogonal rotation (varimax method with Kaiser normalization) was performed. The initial exploratory factor analysis of our study proposed 4 factors (Table 2).
Exploratory Factorial Analysis Considering the Principal Components Method With Orthogonal Rotation (Varimax Method With Kaiser Normalization).
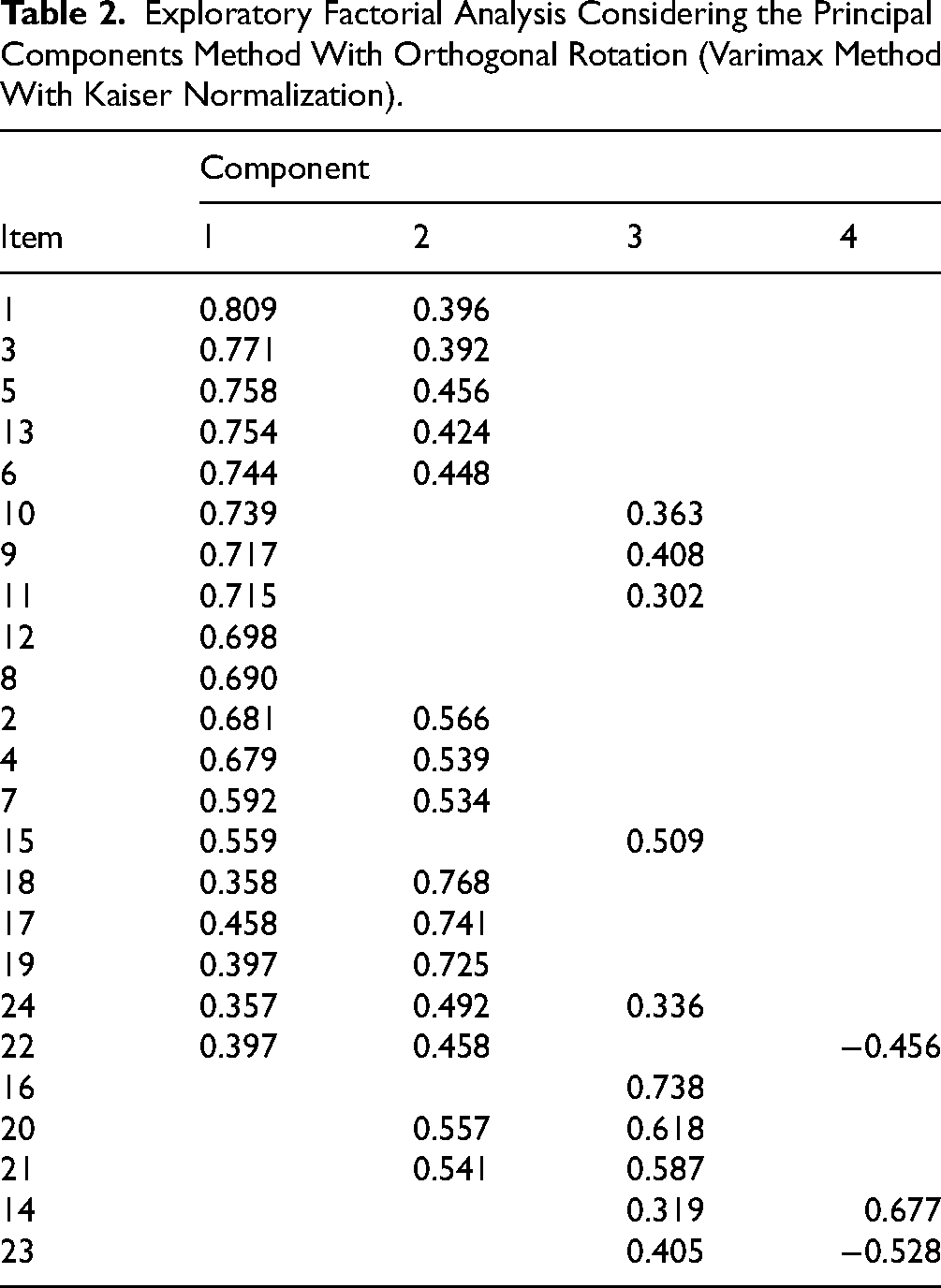
The CANHELP Life Bereavement Questionnaire proposes the evaluation of 5 quality subscales, namely relationship with the doctors, characteristics of doctors and nurses, illness management, communication and decision making, and your involvement. Despite these considerations, the instrument remains unidimensional as the first factor explains 53.061% of the total variance of the items and the second factor explains 6.545%, indicating unidimensional adequacy.
The English and Arabic item description of the CANHELP Lite Bereavement Questionnaire is available in Appendix A.
Discussion
The current study provides robust evidence for the reliability and validity of the Arabic version of the CANHELP Lite Bereavement Questionnaire. To our knowledge, this is the first cross-cultural adaptation and psychometric analysis of this instrument conducted in Saudi Arabia and other Arab countries. We employed a rigorous methodology for translation, cross-cultural adaptation, and validation, adhering to recommended guidelines, which strengthens the study's credibility.
The psychometric testing yielded satisfactory results. Notably, the Arabic version demonstrated strong face and content validity, confirming that all items are relevant for measuring the intended construct. 16 The internal consistency was exceptional, with a Cronbach's alpha of 0.950, indicating excellent homogeneity among items. These values align closely with those reported in original studies, reinforcing the instrument's reliability.17,18
Moreover, we observed adequate concurrent criterion-related validity, as evidenced by a significant correlation with caregivers’ global satisfaction ratings regarding the care received in the past 3 months of life. This correlation suggests that the Arabic version of the CANHELP Lite Bereavement Questionnaire effectively measures aspects of caregiver satisfaction during critical end-of-life stages.
The implications of these findings are profound. By enabling after-death assessments of caregiver satisfaction, the questionnaire can help identify and evaluate interventions aimed at enhancing care quality at the end of life. Such assessments are vital, as improving caregiver satisfaction not only benefits the caregivers themselves but can also indirectly elevate the overall quality of care received by patients.
Given the cultural and contextual nuances in Arab populations, the translation and validation of the CANHELP Lite Bereavement Questionnaire are particularly significant. This instrument will provide healthcare professionals and researchers in Saudi Arabia and other Arabic-speaking countries with a reliable tool to assess bereavement experiences and caregiver satisfaction. Ultimately, this can guide targeted interventions that improve end-of-life care and contribute to a broader understanding of bereavement in diverse cultural contexts.
Limitation
This study followed a rigorous methodology and was developed across care settings, which allow us to be confident regarding the generalizability of the results produced here. Nevertheless, there are some limitations. Effects related to memory might be considered a limitation of this study, as it is a retrospective evaluation by caregivers. In this context, the evaluation of satisfaction with care could be affected by the amount of time between the death and the evaluation. Proxy reports after end-of-life care can be obtained at varying time intervals post-death. Regarding this, it was decided to conduct the interview 3 to 12 months after the patient's death. This is a conventional time frame for evaluating this construct, as similar periods of time were used in other studies.19,20 However, we recognize that other intervals may provide more accurate data.
Future research should explore the optimal timing for the collection of data from bereaved caregivers. Also, the test-retest reliability was not performed, which is the reason why the instruments’ sensitivity to change was not evaluated.
Moreover, future research should explore the applicability of this instrument in various clinical settings and among different populations to further establish its utility and refine its use. Additionally, longitudinal studies could enhance our understanding of how caregiver satisfaction evolves over time and how it correlates with overall care quality.
Conclusions
The Arabic version of the CANHELP Lite Bereavement Questionnaire is a valid and reliable instrument for the assessment of satisfaction with end-of-life care and can be used in both clinical and research settings.
This is the first instrument available in Arabic to assess bereaved family members’ satisfaction with end-of-life care and it could be very important as it might be a valuable contribution to improve the Saudi healthcare system.
In summary, the Arabic version of the CANHELP Lite Bereavement Questionnaire represents a significant advancement in the measurement of caregiver satisfaction and bereavement in Arabic-speaking populations. Its adoption could pave the way for improved end-of-life care practices, ultimately benefiting patients and families alike.
Footnotes
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Health Data Protection Authority and Research Ethics Committee in Saudi Ministry of Health (approval no. 5274/1401) on July 15, 2023.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Verbal informed consent was obtained from the bereaved family members for their anonymized information to be published in this article.
Appendix A
Item description of the CANHELP Lite Bereavement Questionnaire
