Abstract
To understand how patients perceive their experiences leading up to, during, and after a clinical trial, and the relationship these experiences had with future willingness to participate, we conducted 3 focus groups with patients who had prior clinical trial involvement (n = 25). Discussion topics included clinical trial discovery, enrollment, communication, trust, patient-centricity, and future enrollment. Patient focus groups revealed a variety of motivations for enrolling in clinical trials (eg, altruism, efficacious treatment, curiosity, desperation, etc.). Patients learned about clinical trials through trusted sources (eg, primary care physicians, patient advocacy groups) and social media. Access and uncertainty about clinical trials were barriers to enrollment. Patient-centric communication and attention given to disease states and symptom severity were valued and made patients feel genuinely cared about. Post-trial follow up and being informed of trial results were inconsistently reported by patients. Critically, patients described frustration with an overall lack of patient experience measurement. Patients identified a need to measure experiences before, during, and after clinical trials and emphasized that doing so would facilitate patient trust and overall experience.
Key Points
Patient centricity is critical in facilitating trust and patient engagement during clinical trials.
Patients enrolled in clinical trials want to be asked about their experience.
Regular and frequent communication with patients should occur, even after trial completion.
Patient-centric resources, for example, user-friendly technology, transportation, and on-site support, can impact patient experience.
Introduction
Trust is a central component of patient-provider relationships at large, 1 and notably, a critical component of patient willingness to enroll in and remain in a clinical trial.2‐4 Uncertainties about the risks and benefits of participating in clinical trials, and the motives of care providers and organizations conducting the trials can undermine trust. 5 As such, there is a need to understand how patients experience trust throughout clinical trials, because clinical trials are in and of themselves, uncertain. Doing so may bolster enrollment and retention across all phases of clinical trials, which has been a longstanding issue.6,7 Unfortunately, measurement of patient experience and perceptions of clinical trial research is heavily under-researched. 8 As such, standardized measurement tools are needed to determine where site and sponsor quality improvement efforts should be focused.
The first step in understanding patient experience in clinical trials is identifying how it is currently measured and how potentially burdensome it may be for patients to have their experiences measured during a clinical trial. The goal of this research was to qualitatively examine how patient experience has been measured in previous clinical trials with a diverse set of patients who have been involved in clinical trial research (eg, sought to enroll, enrolled, participated in, or completed a trial). To do this, we conducted focus groups with former clinical trial patients and asked them if and how patient experience was measured, and about common facilitators and barriers to positive patient experience.
Method
Recruitment and Sample
Patient focus group participants were recruited by Savvy Cooperative® participant pool using prespecified inclusion/exclusion criteria. If participants answered “yes” to “Do you have any clinical trial experience?”, they were further screened. Participants having comorbid conditions were also included in the sample. Purposive sampling using patient demographics was employed to identify and ensure diversity of focus groups. Savvy Cooperative® then confirmed participant availability and scheduled focus groups with patients.
Data Collection
The research team created patient focus group discussion guides with topics derived from peer-reviewed literature and internal and external stakeholder input. Topics included the experience of clinical trial discovery and enrollment, communication across a trial, patient trust, patient centricity, patient needs, patient burden, and reasons for potentially participating in a clinical trial in the future.
Data Analysis
Focus groups were conducted over the course of 3 sessions, using the Forsta InterVu platform within Forsta Foundations. Average length of focus groups was 81.04 min (range: 78.58-85.27 min).
Results
Twenty-five clinical trial patients participated in one of the focus groups depending on condition type (eg, oncology, neurological, and other chronic). Seventeen identified as women (68%), 5 as men (20%), 1 as transgender male, and 2 as gender nonbinary. The mean age was 44.0. Most participants reported their general health as “good” (44%), however, none rated it as “excellent” (see Table 1).
Patient Characteristics.
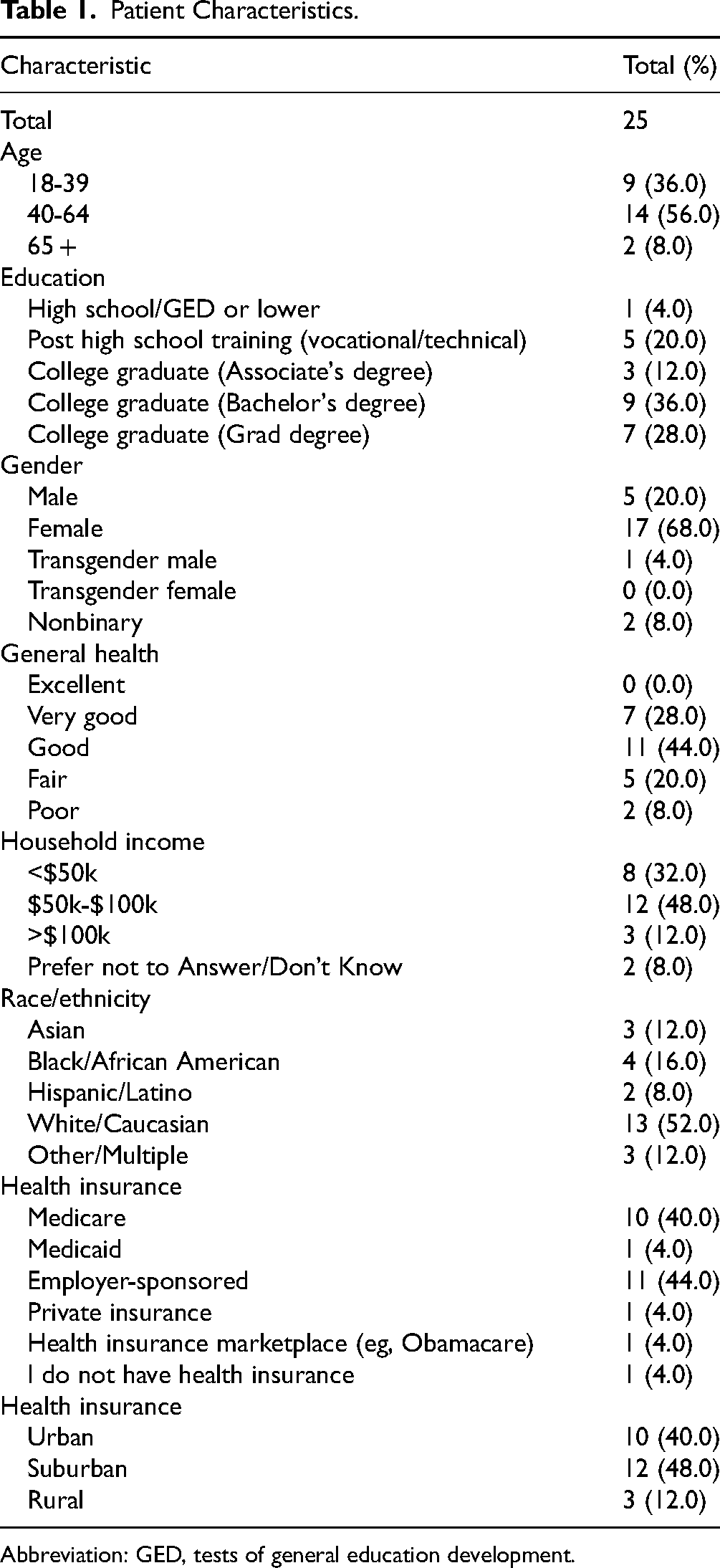
Abbreviation: GED, tests of general education development.
Learning About Clinical Trials
Patients described numerous barriers and facilitators related to clinical trial discovery and learning about the specifics of a clinical trial they were considering. Common methods for patients to learn about clinical trials were through patient advocacy groups and newsletters, primary care providers, and social media. Patients voiced frustrations and uncertainty about requirements of clinical trial enrollment and participation due to complexity of language about the clinical trial itself as well as information contained within the informed consent.
Reasons for Enrollment
Patients described a variety of reasons for enrolling in trials including reducing stigma, desperation and wanting to live longer, assisting patient communities, curiosity, and other altruistic purposes.
Patient Experience Measurement in Clinical Trials
Patients described if and how patient experience was measured in clinical trials, and how often. Patients described frustration over the lack of patient experience measurement. Clinical trial teams with a patient-centric focus that put the unique needs of each patient first was valued by patients. Regular communication, responsiveness, multiple contact methods, and genuine feelings of care increased trust and overall patient experience. Negative experiences during clinical trials centered around participant burden and not feeling genuinely cared about. Patients often voiced frustration with the time commitment required for participation, transportation obstacles, waiting around, and infrequent or impersonal communication with clinical trial teams. Gathering of requisite health history to participate in a trial was also a burden for patients.
Patient-Centric Resources
Patients described numerous ways that clinical trials could become more patient-centric to decrease patient burden. Common patient-centric solutions included being able to schedule appointments as opposed to being given appointment times, the ability to schedule appointments far in advance, more user-friendly communication tools, and decentralized trials for more severe disease states that made travel difficult.
Follow Up and Trial Results
Patients described frustration with communication about clinical trial results as well as follow up about results after the conclusion of a clinical trial. They also described how the lack of communication about trials results and follow up by clinical trial teams made them feel like they were “datapoints” as opposed to individuals (Table 2).
Patient Quotes.

Discussion
We conducted focus groups with patients possessing various disease states (eg, oncological, neurological, and other chronic) to examine if and how patient experience had been measured in clinical trials they had participated in. Overall, patient experience measurement was viewed positively by patients when conducted frequently, consistently, and importantly, in a genuine manner. In fact, many patients expressed frustration that patient experience was not being measured more. Although sponsors of clinical trials have begun to distribute patient experience surveys at the conclusion of trials, this lends credence to the fact that it should be measured more consistently
Patients discussed different ways of learning about clinical trials such as through social media, primary care providers, and patient advocacy groups and newsletters. The desire to help others and give back to patient communities were primary reasons for clinical trial participation, and for some, the ability to give a voice to historically marginalized communities. Patients voiced concerns over stigma associated with clinical trial participation, being another “datapoint.” Patients also cited the ability to attain care that was superior to standard care, desperation, compensation, and curiosity as reasons for participating.
Many patients identified multiple barriers to enrolling in a clinical trial, including uncertainty about clinical trial requirements, preexisting symptom severity, long distances required for travel, time commitment, family obligations, and gathering of extensive requisite health information. Prior research supports these findings, demonstrating how extended screening processes and symptom severity undermine enrollment. 10 Patients also described wanting more “plain language,” prior to enrollment, about what the trial would entail, and during the informed consent process, corroborating research describing how information about a trial can be overwhelming. 11 Our research supports how informed consent processes and understanding of risks involved in participating in clinical trial research continues to be an issue, 12 and that ultimately, trial participation and remaining in a trial is driven by patient/provider trust.11,13,14
High symptom severity made travel difficult for some patients and they recommended more support services to compensate for such burdens. Past studies have found that symptom severity is a strong predictor of patient drop-out, 15 and identifying ways to compensate for this can decrease attrition (ie, providing transportation and/or decentralized trials). Recent research has attempted to decrease the burden of in-person visits through digital solutions that can replace such visits, 16 however, more work in this area still needs to be done. Patients also expressed a desire to communicate with clinical trial research teams via means that catered to their unique needs and preferences, and frustrations with usability of communication platforms. Sensitivity to patient sociodemographics (eg, urban/rural divide and education) should also be used to inform patient-centered approaches to care to decrease burden.
At the conclusion of a clinical trial, patients described a need for more follow up and information about the results of their clinical trial, which has been described in previous research. 17 However, some patients described wanting to learn more about research findings beyond it “being in the journal.”
Limitations
Many clinical trials exclude patients with chronic or comorbid conditions (eg, vaccination trials), however, these focus groups did not. Additionally, a minority of focus group participants were only exposed to clinical trial participation through enrollment, having no on trial experience. We view these potential limitations as strengths, as these participants aided in understanding the clinical trial enrollment experience and inclusivity of trial design. Caregivers were excluded from participating so that focus group discussion was more fluid, but caregiver experience should be examined in future studies.
Conclusion
Frequent and consistent patient experience measurement was identified as facilitating trust between patients and clinical trial care teams during a clinical trial. Patients valued being asked about their experience, but only when it was done in a genuine manner. Patients described a willingness to answer patient experience measures and expressed deep frustration that they had not been asked about their experiences more. Efforts should be made to develop and validate a standardized clinical trial patient experience instruments across a variety of clinical trials. Doing so will facilitate patient-centric quality improvement efforts to decrease patient burden and increase patient retention.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for this study was obtained from Savvy Cooperative®.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with Savvy Cooperative®.
Statement of Informed Consent
Written informed consent was obtained from the patient(s) by Savvy Cooperative® for their anonymized information to be published in this article.
