Abstract
A qualitative study of the experiences of patients who received autologous stem cell transplant (ASCT) for the treatment of multiple myeloma (MM) was conducted to better understand their MM disease journey, including first symptoms, diagnosis, ASCT, and recovery. Sixteen participants, including 12 patients with MM and 4 caregivers of patients with MM, were interviewed in focus group meetings. Pain, weakness, and bone pain were common first symptoms among patients. The MM diagnosis was often made by a hematologist or oncologist. Patients were referred to a specialized oncologist after diagnosis, who was the primary driver in making ASCT treatment decisions. Eight patients received their ASCT in the inpatient setting, with some having lengthy hospital stays; 4 received their ASCT in an outpatient setting with 3 eventually being hospitalized. The focus groups identified that patients and caregivers perceived various unmet needs and impacts on quality of life throughout their transplant journey. Educational resources and innovative therapies are needed to reduce the disease burden of MM and enhance the quality of life for both patients and their caregivers.
Key Points: Submission Must Include a Numbered List of 3–5 key Points or Findings (Appearing in the Abstract)
A qualitative study of patient experiences of autologous stem cell transplant (ASCT) for the treatment of multiple myeloma (MM) was conducted to better understand their MM disease journey, including first symptoms, diagnosis, ASCT, and recovery.
Oncologists were the primary drivers in making ASCT treatment decisions.
Post-ASCT symptoms and lengthy recovery process had a negative impact on patients’ and caregivers’ quality of life.
Findings from this study highlight unmet needs and burdens of patients with MM treated with ASCT.
Educational resources and innovative therapies are needed to reduce the disease burden of MM and enhance the quality of life for both patients and their caregivers.
Introduction
Multiple myeloma (MM) is a rare cancer of the bone marrow, with approximately 35,700 new cases expected to be diagnosed in the United States in 2023. 1 For patients newly diagnosed with MM and eligible for an autologous stem cell transplant (ASCT), the standard of care is induction chemotherapy with bortezomib, lenalidomide, and dexamethasone, or daratumumab, thalidomide plus bortezomib, and dexamethasone.2-4 Induction therapy is followed most commonly by an ASCT and maintenance therapy with lenalidomide.2-4 Treatment advances made in recent years have significantly extended the survival of patients living with MM.4,5 with overall 4-year survival rates ranging 80% to 84% among patients ≤65 years of age treated with induction regimens and ASCT in routine clinical practice. 5 However, similar to other cancer, a diagnosis of MM is life-changing, and patients with MM will transition through periods of treatment, remission, and relapse.
The disease journey of patients with MM can vary by disease stage at diagnosis, cytogenetic classification, symptoms, course of treatment, recovery experience and duration, and trajectory toward progression.6-8 The burden of MM also varies by patient characteristics, including age, general health, and comorbidity level, and social and financial determinants, in addition to patient emotional characteristics and caregivers' situations and well-being.8-10 An understanding of patient experiences of their MM disease journey, particularly related to the burden of ASCT and recovery process, may help to identify unmet patient needs in their quality of care. Therefore, in this qualitative study, patients who were diagnosed with MM and received an ASCT shared their experiences including first symptoms, diagnosis, ASCT, and recovery. Since caregivers of patients with MM play a crucial role in their disease journey, 10 they were also included in this study to share their perspectives of the MM disease journey.
Methods
Recruitment
Patient/caregiver recruitment was initiated with an online MM-specific screener posted to the Patient Engagement Research Council website in June of 2022. Study participants were required to have “disease awareness,” meaning that they had experience with ≥1 of the following: read about or participated in a clinical trial; read or contributed to medical journal articles; used online health information sources; had an understanding of how pharmaceutical companies advertise to people with certain health conditions; had training in nursing, medicine, or other health-related areas; had participation in patient advocacy; or had participation in patient assistance programs. This requirement of disease awareness was included to potentially stimulate valuable and actionable feedback. Additionally, participants had to be ≥18 years of age, diagnosed with MM, and had received ASCT within the past 5 years, with a preference for those who received a transplant within the past 2 years. A participant could also be a caregiver of a patient diagnosed with MM who received ASCT within the past 5 years.
Candidates voluntarily participated in a brief telephone interview. Candidate biographies were developed based on the online screening and interviews and presented to a core research team. Sociodemographic factors, including sex, age, race and ethnicity, and education level, were collected in the candidate biographies. The core research team reviewed and selected candidates to create a demographically, epidemiologically, and clinically diverse set of participants. Through a secure electronic document platform, each selected candidate was sent a formal invitation email with a consent form/contract, which communicated confidentiality and Health Insurance Portability and Accountability Act–compliant practices, to sign; patients and caregivers were deidentified until consent forms/contracts were signed.
Focus Groups
Participants completed a 15-min prework assignment that included questions related to diagnosis and symptoms of MM, treatment journey to ASCT, and recovery, prior to virtual focus group sessions (Supplementary Table 1). An initial 1-h virtual meeting with patients and caregivers was conducted on October 11, 2022. The agenda of the initial meeting was to introduce the core research team members and establish a relationship with the participating patients and caregivers. Two other virtual focus groups were conducted; one 2-h session on October 12, in which patients were asked to narrate their MM disease journey starting with first symptoms and diagnosis, then focusing on the ASCT and recovery period, and another 1-h session on October 13, in which caregivers of patients with MM were asked to provide their perspectives of the ASCT experience. The initial meeting and focus groups were facilitated by a research specialist with participation of the core research team. Virtual group sessions were conducted based on a research-informed discussion guide and were audio-recorded and transcribed verbatim. Thematic narrative analysis was utilized to identify concepts from the audio transcripts.
Results
Participant Sociodemographic Characteristics
A total of 16 selected candidates consented to being a study participant, including 12 patients with MM and 4 caregivers of patients with MM. Most participants were female, including 8 of the patients and 3 of the caregivers (Table 1). Seven patients were <60 years of age, while the other 5 were 60 years of age or older (Table 1). There was one caregiver in each of the age groups, 40 to 49, 50 to 59, 60 to 69, and 70 to 79 years (Table 1). Study participants were diverse in race/ethnicity and education level (Table 1).
Sociodemographic Characteristics of Study Participants.

First Symptoms
Some of the patients’ comments regarding their first symptoms are presented in Figure 1. Several patients remarked that their first symptom was pain; 4 patients specifically said back pain was an initial symptom. Others noted general pain or shoulder and leg pain as first symptoms. Weakness and tiredness were often stated. A few patients suffered broken bones, including a spinal fracture, a broken femur, and a broken hip.
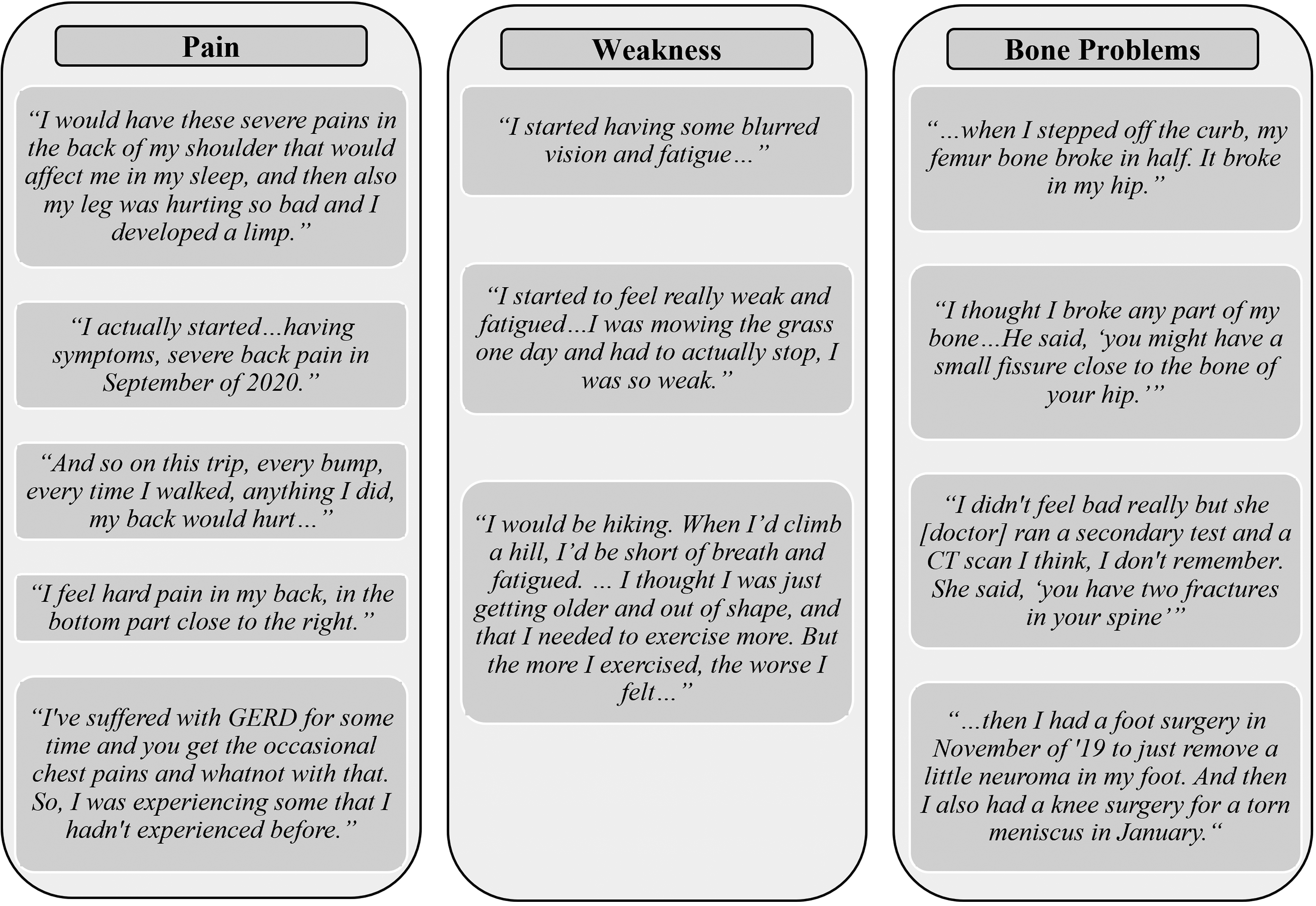
First symptoms of patients.
Other symptoms mentioned by patients included high blood pressure, blurred vision, more frequent illnesses, a limp, and anemia. Two patients said they had no symptoms, and their disease was discovered as a result of routine bloodwork or while donating blood. Two other patients said they were initially being treated for something else prior to their MM diagnosis; one thought they had kidney problems, and another was being treated for gastroesophageal reflux disease.
Diagnosis
Among patients diagnosed with MM, there was a mix of diagnostic settings and timelines. Several had an initial concern for MM found by their primary care physician, often as part of a longer process to understand health issues they had been experiencing. For many, it took a few months (up to 6) to receive a diagnosis from their hematologist or oncologist; after diagnosis, they were referred to a specialized oncologist. Others had initial lab work or tests that suggested cancer and were immediately referred to an oncologist who made the diagnosis: “I used to donate plasma at a local plasma center, and one day I came in to donate and they just told me I could no longer donate. They basically referred me to my local cancer center; they said I needed to find an oncologist.” Others were diagnosed in an urgent or emergency care setting over a span of days or weeks: “I went to my primary care physician to make sure that I was OK [after a bone break]. I didn’t feel bad, really, but she ran a secondary test and a CT scan. … She said, ‘You have two fractures in your spine.’ That was in May. … By the end of June, I got the diagnosis that I have multiple myeloma. … After that, the doctor transferred me to the cancer center.” Another patient said, “I went to the emergency room. From the emergency room, I was admitted into ICU. That's where I learned about my diagnosis.”
Decision-Making for ASCT
Oncologists were the primary drivers in making ASCT treatment decisions. All patients said their oncologist strongly recommended ASCT. Most patients were receptive to this recommendation and readily agreed with their oncologist's recommendation. One patient was somewhat hesitant due to COVID-19 and concerns about having a weakened immune system. Some caregivers were significantly involved in treatment decision-making. This included researching options and helping the patient to make treatment decisions.
Patients described their initial reactions to learning about ASCT in a variety of ways:
“When I was diagnosed, I was in a fog. … I focused on the word ‘cancer.’ I don’t know about anybody else, but for me, cancer meant death, and so that's what I was focusing on. It was good that I had my daughter with me. … She did the bulk of the research.” “They said stem cell transplant was my best chance at the longest remission.” “I tried to read as much about it as possible. … Having a stem cell transplant, that seemed to be the gold standard.”
About half of the patients stated ASCT was their first recommended line of treatment. Most patients said they required some initial treatment so that they were stable enough for ASCT; some achieved this more quickly, while others took months. One patient expressed some confusion about why ASCT was recommended once she was stable. A few had to delay their ASCTs, one patient had COVID-19, and another had to wait for her husband to be eligible for family medical leave.
Patient ASCT Experience
Among patient participants, 8 received their ASCT in the inpatient setting, with several having hospital stays that were 2 weeks or longer (Table 2). Of the 4 patients who received their ASCT in the outpatient setting, only one remained an outpatient the entire time; the other 3 patients were admitted to the hospital, with one having a stay of 17 days (Table 2). Thus, almost all patients spent a lengthy duration in the hospital.
ASCT Patient Experience.

Abbreviation: ASCT, autologous stem cell transplant.
Some remarks of patients’ ASCT experiences included the following:
“I just got so weak, I didn’t want to eat anything, didn't want to drink anything, I couldn’t walk.”—Inpatient “There was no way I would have been able to do outpatient. … I was so weak. It was tough.”—Inpatient “They told me, ‘You’re going to bottom out after you get that high dose of chemo.’ … I had a fever and it was like 103, and once that fever broke, then my counts started climbing.”—Inpatient “The day after I got my stem cells, I developed nausea, and my fever went up to about 104, and I had a fever for about 4 days. I literally was delirious.”—Outpatient
Patient ASCT Recovery
Figure 2 presents patients’ comments on their recovery from ASCT. After patients received their ASCT, initial recovery time ranged from 2 weeks to 4 months. Three patients felt they were initially recovered in under a month, 4 patients said it took about a month to recover, another 4 patients said it took 2 to 3 months, and one patient said it took 4 months. Several patients indicated the initial recovery period was quite challenging. The most frequently mentioned symptoms were nausea, loss of appetite, weakness, fatigue, and pain.

Patient autologous stem cell transplant recovery experience.
Among the patient participants, complete recovery took from 30 days to “never.” One patient recovered very quickly and was working again 1 week after arriving home from the hospital, 4 patients said they returned to full activity after 3 to 4 months, 2 patients said it took 6 months to recover, and one patient said it took a full year to recover. Two patients noted that they still have not fully recovered. Two of the patients had largely recovered but left the workforce, one to continue treatment and the other to protect from further injury. Patients who had not fully recovered expressed frustration and disappointment at their inability to fully resume their prior activities. Thus, most patients took a significant amount of time to recover from their ASCT.
Role of Caregivers in Patient ASCT Recovery
Patients said their caregivers supported them in a variety of ways during their recovery, including staying with them during their recovery period, driving them to medical appointments, and taking time off work. Several patients noted that the caregiver role also had financial implications, largely related to missing work.
Patients noted the following about their caregivers:
“My wife was my caregiver and she would drive me. After the stem cell transplant, I had to go in daily for 5 days and that was one of the toughest parts.” “My husband would take me to the local cancer center, drop me off, and then he would pick me up in the afternoon.” “I have two adult daughters. So, they’ve helped me out tremendously. They’ve been there for me. They drive me to my appointments and whatnot.”
Caregiver Perceptions of Patient ASCT Recovery
Caregivers expressed significant burden associated with the patient ASCT recovery experience, including financial burden, social/emotional toll, and lack of preparedness to provide a high level of care when needed.
Some perspectives of caregivers on the recovery period following ASCT included the following:
“I was not prepared for how she responded when she was at her lowest point. That was a significant burden to me, not being a professional. And never having done this before, I really feared frankly for her being able to survive it.” “My normal day was wake up, make sure my daughter was up, getting ready for school, take her to school, drop her off, go to the hospital, and then sit with [wife] until it's time to leave to go pick up my daughter.” “Once the transplant was scheduled…we put together a team that would cover her support, physically being with her for a period of about 6 weeks.” “It was very hard for her. She did have times where she got very sick,… The most difficult thing … would be the doctor's appointments. There were so many. Sometimes we were going, I believe, every day.”
Discussion
In this qualitative study of the experiences of patients with MM who received ASCT and perspectives of caregivers, we learned that patients had a very high reliance on their oncologists to make treatment decisions, especially for having the ASCT. Many expressed they had limited personal involvement in the treatment decision-making process. Currently, ASCT remains the standard of care for patients diagnosed with MM and eligible for transplant. 3 Several clinical trials, including those conducted in the last decade, comparing ASCT to more novel therapies for MM, have demonstrated improved progression-free survival and overall survival in patients with MM who received ASCT.11,12 Furthermore, multiple facets of the ASCT procedure (eg, use and enhancement of the collection process of patients’ peripheral blood stem cells) have improved over time providing better management of patient safety and an easier treatment experience. 11 However, therapy selection should be centered around a well-informed shared decision-making process that includes medical professionals, patients, and their caregivers all involved in the pro/con assessment of ASCT.
A recent review of several scientific studies and social media posts of patients with MM found that for both younger and older patients, it is important to them to be involved in the treatment decision-making process. 13 In another qualitative study of patients with MM and caregivers, they reported a substantial burden related to the intensive research needed to understand the evolving MM treatment landscape in order to participate in personalized treatment decisions. 14 Such intensive personal involvement impacted patient health and well-being, in addition to the treatment decision-making process. 14 These findings support this study in that patients with MM may value more patient-oriented educational resources that incorporate changing treatment paradigms with meaningful patient-centered decision aids. Such educational resources should also integrate the physical and emotional well-being of patients to deliver more patient-centered care.
Another prominent area of unmet need identified in this study was the unanticipated challenge of recovery following the ASCT procedure. Many patients experienced extended hospital stays and lengthy recoveries. Both patients and caregivers emphasized they felt unprepared for the difficulties they faced during recovery from the ASCT. These study findings underscore the need for greater advances in the treatments for patients with MM to alleviate the disease burden on both patients and caregivers. Since MM is chronic and unpredictable in its course, supporting patients through recovery from frontline ASCT treatment is exceptionally important. In fact, a study at the Mayo Clinic found that worse outcomes are associated with the absence of caregivers among patients with MM who have ASCT. 15 The findings of a study by Gertz et al 15 emphasize the significance of caregivers to patients with MM and are suggestive of the substantial strain they likely endure often without supportive services. 10 Other qualitative studies of patients with MM have found that healthcare services post treatment are an unmet need, and improving care in this setting may enhance patient experiences.16-19 Outpatient multidisciplinary team-led care, holistic care plans that also address psychological and emotional needs, and support groups/networks are all suggested improvements for the survivorship care of patients with MM and the psychosocial support of caregivers.16-19
Limitations
This study included a small population of participants and the individual-level perceptions of disease journeys may not be generalizable at the population level. Additionally, patients with MM who chose to delay or forego ASCT therapy were not included, and thus perspectives regarding the decision-making process and treatment journey in this subpopulation were not captured. Future studies are needed to capture the in-depth MM disease journey. Also, men and those >70 years of age were underrepresented in this study. For this study, participants were required to have exhibited some aspects of disease awareness and were to some extent engaged in the management of their MM. Such participant inclusion criteria likely impacted the study findings on the patient and caregiver perspectives throughout the disease journey, which may not be reflective of the experiences of all patients with MM and their caregivers.
Conclusions
This qualitative study of the experiences of patients with MM treated with an ASCT and their caregivers highlights the unmet needs and burdens during the MM disease journey. Patients who had an ASCT were most affected by post-ASCT symptoms that had a negative impact on their quality of life. Recovery from ASCT is lengthy and challenging for patients and strong caregiver support is needed. More educational resources, innovative therapies, and holistic care plans may improve the quality of care for patients with newly diagnosed MM to reduce the burden of disease and enhance the quality of life for both patients and their caregivers.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735231202733 - Supplemental material for Experience of Autologous Stem Cell Transplant in Multiple Myeloma: The Patient and Caregiver Perspective
Supplemental material, sj-docx-1-jpx-10.1177_23743735231202733 for Experience of Autologous Stem Cell Transplant in Multiple Myeloma: The Patient and Caregiver Perspective by Doris K Hansen, Todd Bixby, Karen Fixler, Lisa Shea, Christine Brittle, Kimberly Brunisholz, Yi-Hsuan Liu and Jinhai Stephen Huo in Journal of Patient Experience
Footnotes
Authors’ Note
Writing Assistance: Editorial and writing assistance for this article were provided by Jay Lin and Melissa Lingohr-Smith of Novosys Health, which was funded by Janssen Pharmaceuticals. Todd Bixby, Karen Fixler, Lisa Shea, Kimberly Brunisholz, Yi-Hsuan Liu, and Jinhai Stephen Huo are employees of Janssen Pharmaceuticals and may own stock in the company. Christine Brittle is an employee of CorEvitas and consultant for Janssen Pharmaceuticals. Participants were informed that no treatments would be provided and they could withdraw at any time. The purpose of this research study was to collect patients’ and caregivers’ perspectives and qualitative insights. All participants signed a consent and release form which communicated confidentiality and Health Insurance Portability and Accountability Act (HIPAA)-compliant practices. As all data collected were deidentified, no ethics board review was required. This study was conducted in accordance with the Helsinki Declaration of 1964 and its later amendments. Written informed consent was obtained from patients and caregivers for their anonymized information to be published in this article.
Acknowledgments
The authors would like to thank the patients and caregivers who participated in Janssen's Patient Engagement Research Council (PERC) activities for their engagement and insightful feedback.
Declaration of Conflicting Interests
DKH has consulted with BMS, Karyopharm, Janssen, and Pfizer; received research funding from BMS, the International Myeloma Society, Karyopharm, and the Pentecost Family Myeloma Research Center; received honoraria from OncLive and Survivorship; and served on BMS IMW Ide-celacademic advisory board, BMS MM ASH Steering Committee, and MM Pfizer Advisory Board. TB, KF, LS, KB, Y-HL, and SH are employees of Janssen Scientific Affairs, LLC, and hold Johnson & Johnson stock. CB works for CorEvitas, which provided analytical services funded by Janssen Pharmaceuticals.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was funded by Janssen Scientific Affairs, LLC and Legend Biotech USA, Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
