Abstract
Diabetes is the seventh leading cause of death in the US. Diabetes group visits (GVs), which include group education and individual medical visits have been shown to improve clinical outcomes. However, few studies have evaluated virtual GVs. We conducted a single-arm pilot study to test the impact of virtual diabetes GVs in Midwestern community health centers (CHCs). Adult patients with diabetes participated in monthly virtual GVs for 6 months. Surveys and chart abstraction were used to assess patient-reported and clinical outcomes. Five CHCs implemented virtual GVs with 34 patients attending at least one session. Virtual GVs show promise as evidenced by these findings: (1) Patients had a nonsignificant decrease in A1C. (2) In the subgroup of patients with baseline A1C ≥ 9%, there was a significant decrease in A1C. (3) Patients had significant increases in diabetes knowledge and support as well as a decrease in diabetes distress. Future studies with a larger sample size and a control comparison group are needed to assess the impact of virtual GVs on patient outcomes.
Introduction
Over 30 million Americans have a diagnosis of diabetes and glycemic control in people with diabetes has declined in recent years. 1 Patients with diabetes may need to make significant adjustments to their diets and adopt new medication regimens. In addition, many patients lack access to primary care providers (PCPs), and traditional one-on-one care with PCPs may not be sufficient for patients to gain the knowledge to effectively manage diabetes. 2 Group visits (GVs) that include group diabetes education and individual medical visits are a strategy to aid in patients’ self-management of disease. 3 GVs have been shown to improve glycemic control, diabetes knowledge, and quality of life.4–7
Though GVs can be beneficial to patients, the coronavirus disease 2019 (COVID-19) pandemic posed a new challenge. In-person visits presented increased health risk, thus many health systems shifted to virtual visits. Several studies have found that virtual GVs could improve health outcomes, lead to increased patient satisfaction, and increase accessibility to care given the flexibility in transportation and location it enables.8–11 However, certain groups may have limited access to the internet and low digital literacy skills. 8 No studies have evaluated the use of virtual GVs in community health centers (CHCs) settings or across multiple sites.
It is important to evaluate virtual options for activities that involve group gathering such as GVs, a model of care that has been shown to benefit patients when conducted in person. It is particularly vital to study virtual GVs in CHCs, which play a critical role in providing primary care to many vulnerable patients, including patients who are low income and from racial and ethnic minority populations. 12 Further, patients who receive care at CHCs have higher rates of diabetes and diabetes-related complications.13,14 We report findings from a 1-year pilot study implementing virtual GVs for adult patients with uncontrolled diabetes at Midwestern CHCs, including patient-reported outcomes on diabetes self-management and clinical outcomes such as A1C.
Methods
The virtual GV pilot was part of a larger study called Diabetes MESSAGES (clinicaltrials.gov #NCT03487692), which was a cluster randomized trial to test the impact of diabetes GVs on patients with type 2 diabetes in CHCs in the Midwest Clinicians’ Network (MWCN), an organization with more than 150 CHC members. Sixteen CHCs were recruited and randomized to intervention or control in a 1:1 ratio. A block randomization scheme with randomly permuted block sizes of 2 or 4 was conducted and the randomization was stratified by state and setting (rural vs urban). The intervention CHCs conducted in-person GVs for their patients, and patients were followed for 18 months. The control arm collected data during this time on patients via chart review. Five CHCs (2 in the intervention arm and 3 in the control arm) dropped out during the first 18 months due to administrative issues, staffing changes, and time and resource constraints. After 18 months, the remaining CHCs in the control arm received training on how to conduct in-person diabetes GVs for equity purposes. Due to the COVID-19 pandemic, the control CHCs had to switch GVs to a virtual format. In this paper, we report the results from the control arm's experience with virtual GVs through a single-arm prospective cohort study design. The University of Chicago Institutional Review Board approved the study procedures.
Health Center Recruitment and Training
To participate in the Diabetes MESSAGES trial, CHCs needed to assemble an organizing team of 3 to 4 staff with at least 1 medical provider. After the intervention group concluded GVs, the control arm CHC staff attended an in-person training session on the implementation of in-person GVs. Following this session but prior to recruitment of patients, the COVID-19 pandemic forced the control arm's GVs to adapt to a virtual format as described previously. 15 Briefly, CHCs received training on telehealth and implementing virtual GVs. Guest speakers with telehealth experience spoke with staff about the effective utilization of telehealth for group sessions and gave tips on facilitating and implementing virtual GVs.
Patient Recruitment and Enrollment
Up to 12 adult patients with type 2 diabetes who were at least 18 years of age, English or Spanish speaking, had recent A1C ≥ 8%, and attended ≥2 clinic visits in the past year and ≥1 in the past 6 months were recruited by each CHC. Patients who were pregnant or had an uncontrolled psychiatric problem, dementia, cognitive impairment, or hearing difficulties were excluded. CHC PCPs reviewed the list of eligible patients before enrollment and could exclude patients who were not appropriate for GVs. Consent was obtained via phone or video from all patients enrolled in the study.
Virtual GV Intervention
Six monthly 60-to-90-min virtual GVs were conducted. Each visit was led by trained clinic staff on a video conferencing platform available at the CHC. Trained CHC staff provided diabetes group education and patients participated in facilitator-led group discussions that enabled material review, peer support, and self-management goal setting. Patients were recommended to have an appointment with their PCP within 2 weeks of each virtual GV. CHC teams were provided with a sample curriculum and resources for patient education materials, but specific session content was not dictated by the research team. CHC teams were encouraged to tailor their GVs to the interests and needs of their patients and make use of the resources available within their organizations and communities. GV sessions covered a range of topics such as the “ABC's” of diabetes, nutrition, physical activity, medication, and mental health, and included interactive components to keep patients engaged. 15
Clinical Outcomes
For enrolled patients, staff completed retrospective chart abstraction for demographics including age, gender, and race/ethnicity. Clinical data including A1C, LDL, blood pressure, and weight were abstracted at baseline (the start of the intervention), 6 months (the end of the intervention period), and 12 months (6 months post intervention period). Dates of key healthcare services such as PCP visits and A1C tests were also abstracted.
Patient Surveys
Patients completed a baseline survey that included demographic information as well as anticipated barriers to participation such as GV timing, work schedule, and technology access. Surveys also included validated scales on diabetes support, 16 diabetes knowledge, 17 and diabetes distress. 18 These validated scales were readministered at six months. At 6 months, patients also completed an evaluation of the virtual GVs. No validated scales of patient satisfaction with virtual GVs existed, so surveys were created as per previous work done by this group, 19 a previous study on virtual GVs in young adults, and benefits and challenges identified in literature review of GVs. 8 Survey questions covered topics such as interaction with other patients (eg, “I felt like the other GV patients really understood my experiences with diabetes”), medical providers (eg, “I liked seeing nurses, dietitians, social workers during the visit, instead of only seeing my doctor”), diabetes self-management (eg, “The GVs helped me improve my diabetes self-management skills”). These questions were answered on a 5-point Likert scale. Survey items to assess overall satisfaction included, “Overall, how satisfied are you with virtual GVs” and “Would you recommend virtual GVs to a friend or family member”. Surveys were administered by telephone and responses were recorded in the online survey and database application REDCap.
Analysis
For this pilot study, descriptive statistics were assessed for all quantitative outcomes. For survey outcomes, linear mixed effect models (LMMs) were used to evaluate change in knowledge, distress, and support before and after GVs. For clinical outcomes, LMMs and generalized linear mixed models (GLMMs) were used to test the GV effect (ie time trend) for continuous outcomes (such as blood pressure) and binary outcomes (such as dichotomized A1C data), respectively. Cohort and subject were added as random effects in all LMMs and GLMMs to consider within-cohort and within-subject associations. No covariates were considered in the models due to the single-arm design and the limited sample size. Normality was checked for all continuous outcomes in LMMs. The values closest to each timepoint of interest within predefined windows were used for analysis: Baseline (12 months before baseline until 2 months after baseline), 6 months (3 to 9 months after baseline), and 12 months (9 to 15 months after baseline). All analyses were conducted in R studio (R version 4.2.0).
Results
Five MWCN-affiliated CHCs participated in the virtual GVs. Two were in urban settings, 2 were in suburban settings, and one was in a rural setting. A total of 251 patients were spoken to about the study and 91 agreed to participate. Most patients who declined to participate did so because of schedule conflicts while a smaller number were not interested in additional diabetes education or were not comfortable with the group setting. Of those who agreed to participate, 43 were not enrolled, largely because they were lost to follow-up from the clinic. Forty-eight patients were enrolled in the study. Thirty-eight filled out a baseline survey. Demographic information is summarized in the Table 1. Among the 48 enrolled, the average age was 55 (SD = 12) and the average duration of diabetes was 11 years (SD = 9). Participants were 67% female, 67% Black/African American, 27% non-Hispanic white, and 6% Hispanic/Latino. Thirty-four patients attended at least one virtual GV and the mean number of GVs attended was 2.1 (SD = 2.2) sessions across sites. Twenty-one participants completed the 6-month survey. The mean number of GVs attended among those who completed the 6-month survey was 3.8 (SD = 2.2).
Patient Demographics (N = 48).

No patients were American Indian, Native American, Alaskan native, Asian, or Pacific Islanders.
These items were retrieved from patient baseline surveys.
The characteristics of the patients with low attendance (0-1, n = 26) were compared to patients with high attendance (2+, n = 22). There was no significant difference observed in age, gender, race, A1C, years with diabetes, or employment. A significant difference was observed in education between the 2 groups (P = .02). Among the high attendance group, 67% completed at least some college compared to 24% in the low attendance group.
Clinical Outcomes
A1C decreased from 9.84 ± 1.78% at baseline to 8.98 ± 1.84% at 6 months (p = .11) and 9.06 ± 2.04% at 12 months (P = .10) (Figure 1a). Among patients with A1C ≥ 9 at baseline (N = 31), there was a significant decrease in A1C from 10.70 ± 1.65% at baseline to 9.36 ± 2.07% at 6 months (P = .04) and 9.23 ± 2.35% at 12 months (P = .01) (Figure 1b). The percentage of patients with A1C < 9% increased from 35% at baseline to 56% at 6 months (P = .055). There was no significant change in blood pressure, LDL, or weight. There was a decrease in the number of patients who had an A1C test within the last 6 months from baseline (90%) to 6 months (70%) and 12 months (76%). There were also decreases in the number of patients who had routine health maintenance such as foot exam, eye exam, and lipid panel from baseline to 6 and 12 months. In the 12 months prior to GV intervention, patients attended a mean of 4.83 PCP visits compared to a mean of 5.37 PCP visits during the 12 months that included the 6 months of intervention and the 6 months following (P = .46).
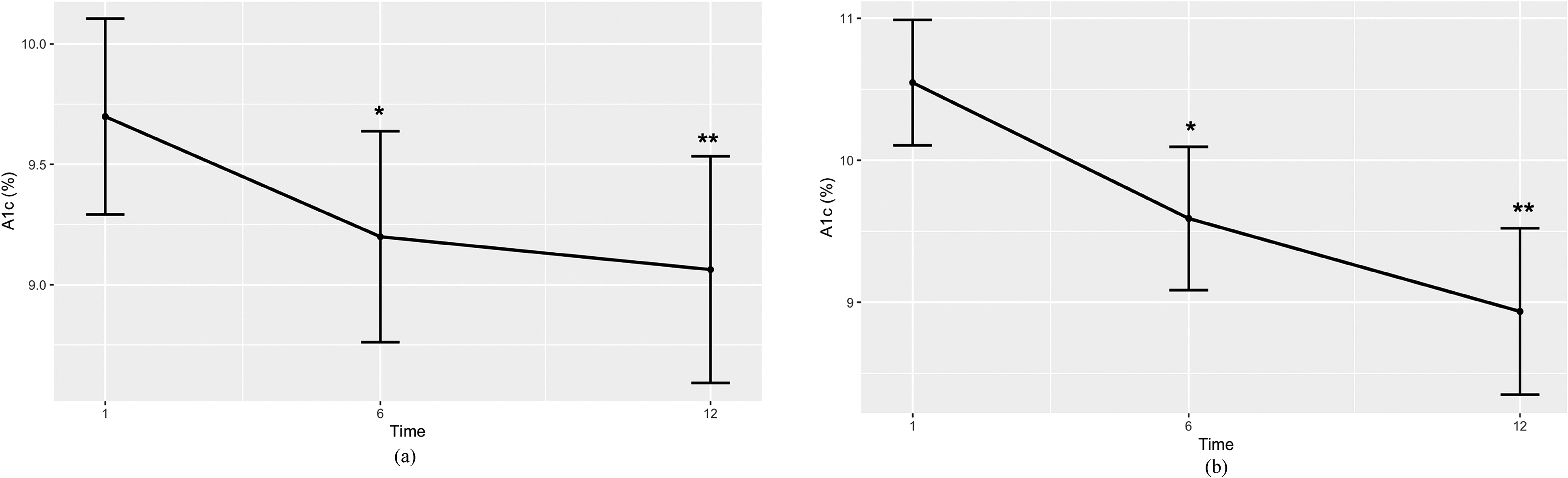
Adjusted mean A1C over time for (a) all participants and (b) participants with baseline A1C
Patient Reported Outcomes
Patients had an increase in their diabetes knowledge (mean (SD): 3.2/5 (0.9) to 3.6/5 (0.7), P = .02) and diabetes support (3.5/5 (0.64) to 4.1/5 (0.7), P < .001) as well as decreased diabetes distress (2.9/6 (1.5) to 1.2/6 (0.5), P = .03) from baseline to 6 months (Figure 2). There were no significant changes in diabetes self-care including foot self-exams, blood sugar testing, or exercise, but patients did have more days of healthy eating in the past week at 6 months compared to baseline (4.5 (2.3) versus 3.2 (2.7), P = .02).
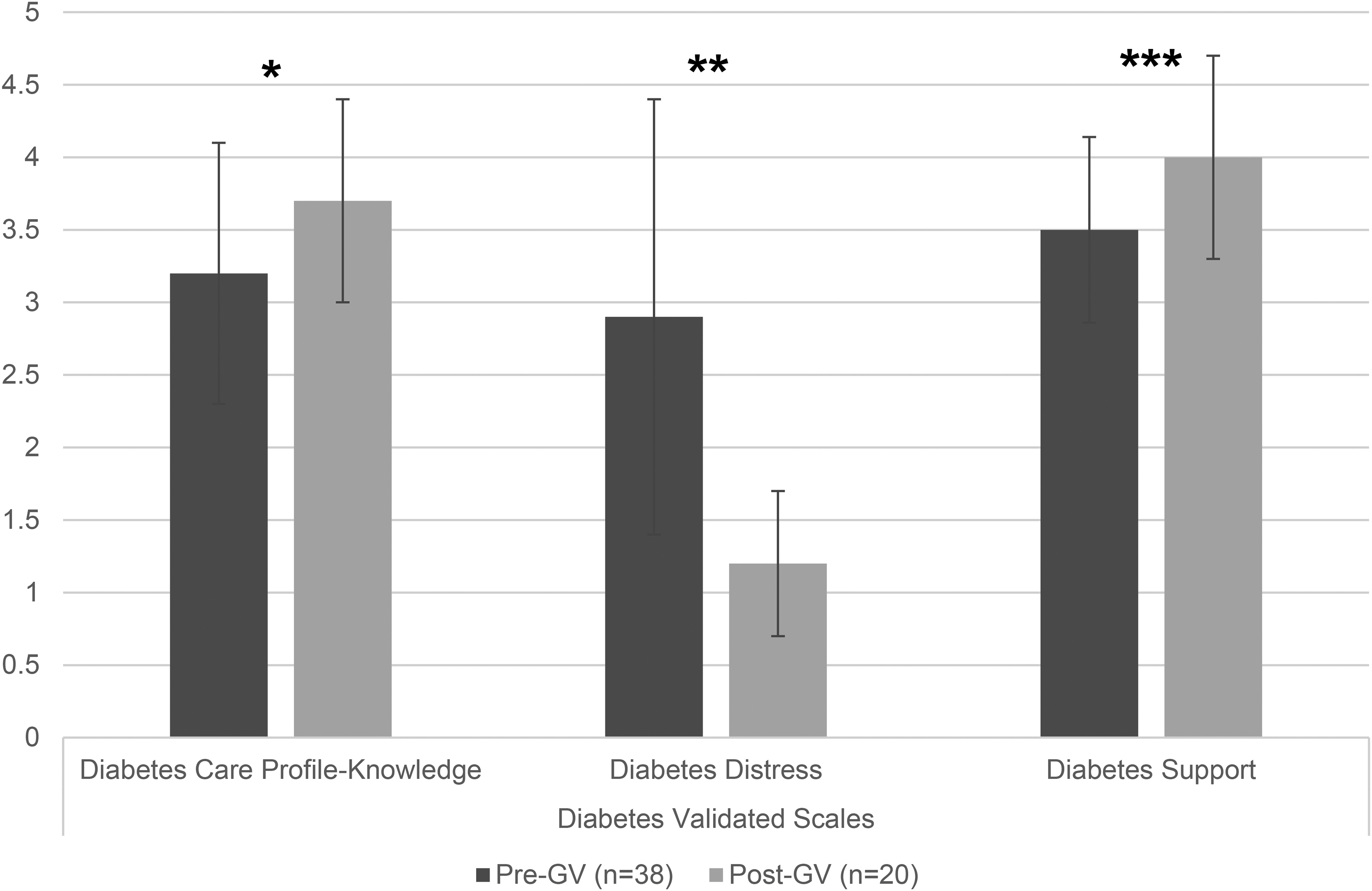
Mean scores from pre and postvirtual GV surveys on diabetes knowledge, distress, and support. *P = .02 compared to baseline. **P = .03 compared to baseline. ***P < .001 compared to baseline.
Patient Satisfaction
Overall satisfaction with the virtual GVs was high with 18/20 (90%) participants stating they were very satisfied. 19/21 (90%) said they would recommend the virtual GVs to others and 20/21 participants (95%) said they would attend GVs in the future. Most participants agreed that GVs taught them diabetes self-management skills (14/18, 78%), motivated them to achieve health goals (16/18, 89%), and introduced them to others living with diabetes (14/18, 78%) (Figure 3).

Patient evaluation of virtual group visits. N = 18.
Patient Barriers
Though participants were satisfied with the program, there were barriers to participating. In total, 18% (4/22) of patients noted each of the following as barriers to participating: timing of the GVs, access to a computer, tablet, or phone, and internet connection. Only 5% (2/37) of patients anticipated that these obstacles would hinder participation in the pre-GV baseline survey.
Discussion
We conducted a prospective single-arm pilot study assessing virtual GVs at CHCs across the Midwest and evaluated clinical and patient-reported outcomes. While there were no significant changes in clinical outcomes overall, among patients with poor glycemic control at baseline (A1C ≥ 9%), A1C decreased significantly. Patients also had significant improvements in diabetes distress, diabetes knowledge, and diabetes support. Overall, patients who attended the virtual GVs were satisfied with the intervention and would recommend GVs to others. Patients also largely agreed that the virtual GVs helped them stay on track with personal health goals and improved diabetes self-management. However, there were barriers to the virtual GVs including low attendance and technological barriers.
Virtual GVs show promise for improving A1C among patients with poor glycemic control in CHCs. Studies have shown that mean A1C in patients with type 2 diabetes increased during the COVID-19 pandemic compared to a non-COVID cohort. 20 In addition, low-income patients with diabetes were the most impacted by the pandemic. 21 With this context, the trend towards a decrease in A1C among patients in our study suggests that virtual GVs could have been an effective means of providing diabetes care during the pandemic. In addition, there was a significant decrease in A1C in patients with baseline levels greater than or equal to 9%. This is important as A1C ≥ 9% is associated with increased complications and decreasing A1C below this level is a public health goal and evaluation measure for CHCs. 22
There were no significant changes in other clinical measures such as LDL, blood pressure, and weight, although patients had good control of these measures at baseline. We found that patients had decreased routine health maintenance such as A1C tests, foot, dental, and eye exams as well as a nonsignificant change in PCP visits. This finding is in line with previous studies that have shown patients received less routine health maintenance during the early months of the pandemic.23–25 However, this does bring into question how to ensure patients in virtual GV programs and those who attend telehealth PCP appointments still attend in-person health maintenance including labs and physical exams.
Patients who participated in virtual GVs noted more social support and less diabetes distress from baseline to 6 months. This is encouraging as studies have shown that patients with diabetes were more vulnerable to psychological distress 26 and pandemic social distancing measures. 27 In addition, loneliness and social isolation were large problems facing individuals during the pandemic, and both have been shown to lead to increased all-cause mortality.28,29 Participants’ comfort with sharing and connecting with other patients in virtual GVs is also promising, especially given that studies have shown that peer support is associated with improved health outcomes and less distress in patients with diabetes.30,31 Through discussing their experiences with diabetes and sharing their stories, participants can avoid the negative impact of isolation and loneliness while decreasing diabetes distress. This is an important aspect of diabetes care as studies have shown that as many as one-third of patients with type 2 diabetes have distress from their disease, and those with diabetes distress are less likely to adhere to recommendations on smoking cessation, diet, physical activity, and glucose monitoring. 32
Patients were satisfied with the virtual GVs and would recommend them to others. This finding is in line with previous work that showed similar overall satisfaction with in-person GVs. 19 This is encouraging as patients could be drawn to this convenient way of learning and interacting, which could lead to further engagement between patients and CHCs. Studies show that being connected to a primary care practice is associated with fewer hospitalizations and better glycemic control. 33 CHCs offering a virtual GV program could retain and attract old and new patients who are interested in this model of diabetes care.
There were several challenges in implementing virtual GVs. Recruitment and attendance were difficult as patients were often unfamiliar with the virtual platforms used for the visits. Previous work on in-person GVs has also noted high drop-out rates among participants, though optimizing the meeting cadence could improve attrition rates. 34 Patients also listed technology challenges as a barrier to attendance. Though virtual GVs can improve some patients’ ability to receive care, studies have shown that difficulty in technology access and literacy are barriers to effective utilization of virtual platforms in healthcare, particularly for those who are older, disabled, low income, and Black or Hispanic. 35 Finally, patients stated inconvenient timing of the meetings was a reason why they could not attend GVs. From this, it appears virtual GVs may not be an option for patients with long work hours, family obligations, or difficulty with technology access. These patients may benefit more from standard medical appointments.
Limitations
Our study was conducted in Midwestern CHCs and enrolled 48 participants, the majority of whom were female and Black/African American. Because of this, our results may not be generalizable to all populations or settings. In addition, although the patients who participated had a generally positive experience, many patients who were enrolled did not attend any GVs or fill out surveys. Our study did not have a control group; thus, we were unable to compare our findings to temporal trends in change in A1C. Virtual GVs were held during a pandemic which could have impacted patient attendance and increased staff workload.
Conclusions
In summary, participants in the virtual GVs with suboptimal glycemic control saw a decrease in A1C. In addition, patients had a significant increase in diabetes knowledge and support and a decrease in distress. Coupled with patient's high satisfaction with virtual GVs, this model of care shows promise in the CHC setting. Overall, while the study suggests that virtual GVs have a positive impact on patients with diabetes, further research with a larger sample size and a longer follow-up period is needed to confirm these findings. Future work should focus on addressing barriers to patient participation in virtual visits and testing virtual GVs with a larger patient population and a comparison group.36–38
Footnotes
Acknowledgements
The authors would like to acknowledge the FQHCs that collaborated with us in this research. We would also like to thank Chasity Kasir, Sara Siddiqui, Mahima Akula, and Viridiana Luna, for their contributions to this project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Institute of Diabetes and Digestive and Kidney Diseases Chicago Center for Diabetes Translation Research (P30 DK092949) and the U.S. Department of Health and Human Services Office of Minority Health (1 CPIMP171145-01-00). Study data were collected using REDCap, hosted by the University of Chicago Center for Research Informatics (NIH CTSA UL1 TR000430). Baig is supported by a NIDDK Mentored Patient-Oriented Career Development Award (K23 DK087903-01A1).
Ethical Approval
All procedures in this study were conducted in accordance with the University of Chicago Institutional Review Board (IRB17-1385).
Informed Consent
Verbal informed consent was obtained from the patients for their anonymized information to be published in this article.
