Abstract
Hemophilia is a bleeding disorder caused by a single absent/defective gene and characterized by a lack of functional clotting factors. People with hemophilia may experience joint damage, pain, and psychological impairments, all of which could contribute to reduced health-related quality of life (HRQoL). The current standard of care is clotting factor replacement, which is associated with regular infusions; therefore, alternative treatments such as gene therapy (GT) are in development. GT involves the delivery of a functional copy of the clotting factor 8/9 gene by a single infusion into the patient's cells, enabling them to produce their own clotting factor VIII/IX. The impact of treatment on patients’ HRQoL can be assessed using hemophilia-specific patient-reported outcome (PRO) measures. Since these measures were designed before the advent of GT, there is a need for updated individualized PRO measures. Patient groups and regulatory authorities emphasize the need for increased patient engagement when considering clinical trial design. Here, we provide patients’ perspective on undergoing GT and discuss how to capture the patient voice when measuring the therapy's transformative impact.
Keywords
Plain language summary
Blood clotting is impaired in people with hemophilia (PWH) due to reduced activity of one of the essential clotting factors. Depending on the type of hemophilia, a different clotting factor is missing. PWH may have spontaneous joint and muscle bleeds. When there is an injury or during surgeries, they bleed more than healthy people.
Hemophilia can be treated with a replacement of the missing or inactive clotting factor, which is given directly into the vein. Depending on the medication, frequent injections are necessary, approximately 180 times a year. PWH have a high treatment burden.
Gene therapy (GT) is a new treatment option. The faulty gene causing hemophilia can be replaced with a healthy version. Patients receive GT once only and after treatment should produce enough of their own clotting factors. Researchers can collect the views of PWH using patient-reported outcomes (PROs) tools, eg, questionnaires, to evaluate how the treatment impacts patients’ everyday life. The questions asked need to capture the big changes expected after GT.
A group of patients who have already received GT described their experiences. Feelings before GT were generally negative. Key highlighted issues were treatment burden, pain, and barriers to physical activity. However, a few positive sentiments included feeling in control of the health condition. Feelings after GT were mostly positive, such as improvements in physical activity, work, lifestyle, and pain. Negative sentiments included less control over treatment and anxiety about how long GT will work for.
The authors suggest the most important domains to be updated in PRO tools for hemophilia GT. These include the ability to participate in physical activities, future conditions, and future treatment. Pain was considered less important. New PRO tools will hopefully be more individualized to better capture the impact of GT in hemophilia. See Supplemental material for additional details.
Introduction
Background
Hemophilia is an X-linked recessive bleeding disorder that primarily affects males; it is characterized by a lack of functional blood-clotting factors. 1 Hemophilia A (missing or dysfunctional factor VIII [FVIII]) has a prevalence at birth reported as 24.6 per 100,000 males, while hemophilia B (missing or dysfunctional factor IX [FIX]) is less common (prevalence at birth 5.0 per 100,000 males). 2 The different severity levels of hemophilia are as follows: mild, 5% to 40% factor activity; moderate, 1% to 5% factor activity; and severe, ≤ 1% factor activity. 3
Patients with hemophilia can present with spontaneous bleeds, mainly in joints, or traumatic bleeds with excess bleeding after injury or surgery.1,4 These bleeding events have an adverse impact on patients’ health-related quality of life (HRQoL), which is increasingly recognized as an important outcome measure for hemophilia.5,6 Regular prophylactic replacement therapy is currently the evidence-based standard of care (SOC) for severe hemophilia, with hemostatic agents including clotting factor concentrates (CFCs; eg, purified FVIII/FIX) or nonfactor replacement (NFR) therapies. 3 Although SOC treatments have evolved over time, they still require regular intravenous (CFCs) or subcutaneous (NFR) injections and are associated with the risk of adverse events such as inhibitor formation (for CFCs) or thrombotic events (for NFRs).7–9 Therefore an unmet need remains for novel hemophilia therapy options, which may offer increased choice to suit patient preferences; one such option currently in development is GT.
GT as hemophilia treatment—initial considerations
GT for hemophilia is a new therapeutic approach where an absent or defective gene is restored with a functional copy, leading to endogenous production of functional FVIII or FIX.8,9 An increase of plasma clotting factor levels up to ≥12% after GT was achieved in clinical phase 1/2 studies and was effective in controlling spontaneous joint bleeding. 10 GT, therefore, has the potential to significantly improve a patient's life (eg, by reducing treatment burden) but is not necessarily a simple process to undergo. Currently, only one hemophilia A GT product (valoctocogene roxaparvovec) is available, marketed since 2022 in the European Union. 11 The GT patient journey has recently been described based on roundtable meetings between patients and healthcare professionals (HCPs) and can be broadly split into 5 stages: (1) pre-GT information seeking, (2) decision-making, (3) GT initiation, (4) short-term post-treatment follow-up, and (5) long-term post-treatment follow-up. 12 This journey extends through all life stages and involves an evaluation of the various outcomes and goals that are important to the patient (eg, restrictions on physical activity/wish to socially fit in), both before and after GT. 12 Anticipated functional treatment outcomes (eg, reduced bleeding and improved joint health) may include the resolution of hemophilia from severe to mild, and patients also hope that treatment will be effective for several years; however, these outcomes are not guaranteed. 13 In addition, patients should also be aware of the emotional demands of treatment, including the possibility of not meeting all eligibility criteria, a need to adjust to their new routine (due to differences in how treatment is managed), and a lack of feasible retreatment in case of reduced GT efficacy over time.8,13
The GT patient decision-making process may vary based on existing treatment evidence. Data for GT in hemophilia B have shown reduced bleeding and sustained FIX levels up to 8 years, 14 and results for GT in hemophilia A are also encouraging. 15 These trials have predominantly used recombinant adeno-associated viral vectors as a delivery system for the functional gene, but alternative approaches such as gene editing or different viral vectors are also being investigated. 16 While these findings support the potential utility of GT in reducing treatment burden, there is still limited information available on HRQoL outcomes.17,18
PROs in Clinical Research
Definitions
A PRO is defined as any report of the status of a patient's health condition that comes directly from the patient, without interpretation of the patient's response by a clinician or anyone else. 19 PROs are one of the clinical outcome assessments reported in clinical trials and nonrandomized studies and are designed to capture patients’ perspectives regarding treatment benefits, harms, symptoms, and HRQoL; these perspectives are assessed via interviews, diaries, or validated questionnaires.19,20 These questionnaires can be distinguished between generic and disease- or condition-specific measures; recently, treatment-specific PROs have been developed. 21
Regulatory agencies such as the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA) have also been steadily increasing their level of engagement with patients over the past decade through patient involvement in public meetings, workshops, scientific advisory groups, ad hoc expert meetings, and committee consultations. 22 The real-life experiences of patients collected in this process have subsequently been incorporated into regulatory output, demonstrating the effectiveness of patient engagement in improving disease management and helping to deliver meaningful treatment outcomes.22,23
PROs in Hemophilia Treatment: Successes and Limitations
There is a growing understanding in the medical community that PWH have a unique perspective on their treatment (ie, the “patient voice”) and may view issues differently from physicians and other stakeholders associated with drug development or approval. 24 Therefore, a greater focus on patient-centered research has emerged in recent years, and PRO measures are now considered an essential component of hemophilia clinical trials.20,25 In addition, while annualized bleeding rate (ABR) is considered a key outcome in clinical trials investigating new hemophilia treatments,25,26 it has known limitations; these include the variability of “bleed” and “observation period” definitions, 26 the subjective patient reports of bleeding/pain, and the fact that ABR is not an ideal surrogate measure of joint damage as subclinical bleeds are not captured. 25 Finally, generic HRQoL instruments are not readily applicable to hemophilia, since they may not appropriately capture the real-life situation of PWH. It has been noted that a “disability paradox” could be demonstrated using such generic tools, whereby patients with hemophilia report better HRQoL than the general population. 27
Disease-specific PROs are used in hemophilia trials to capture different aspects of the patient experience. They cover a wide variety of patient impacts by including hemophilia-specific questionnaires that measure HRQoL,28–31 physical functioning,32,33 treatment adherence,33–35 and treatment satisfaction. 36 Current PRO measures for adults with hemophilia (and the aspects they encompass) are summarized in Table 1.
Main Aspects Captured by Existing Hemophilia-Specific PRO Measures.
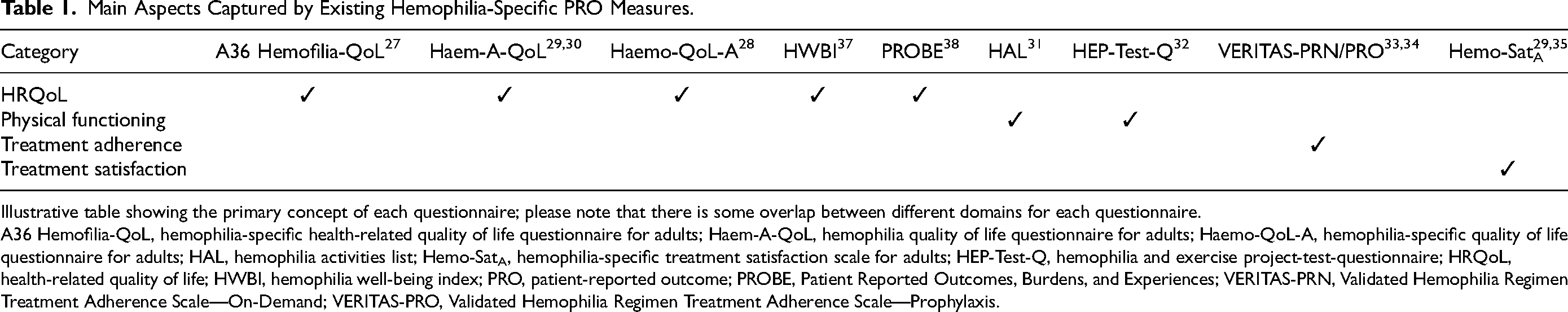
Illustrative table showing the primary concept of each questionnaire; please note that there is some overlap between different domains for each questionnaire.
A36 Hemofilia-QoL, hemophilia-specific health-related quality of life questionnaire for adults; Haem-A-QoL, hemophilia quality of life questionnaire for adults; Haemo-QoL-A, hemophilia-specific quality of life questionnaire for adults; HAL, hemophilia activities list; Hemo-SatA, hemophilia-specific treatment satisfaction scale for adults; HEP-Test-Q, hemophilia and exercise project-test-questionnaire; HRQoL, health-related quality of life; HWBI, hemophilia well-being index; PRO, patient-reported outcome; PROBE, Patient Reported Outcomes, Burdens, and Experiences; VERITAS-PRN, Validated Hemophilia Regimen Treatment Adherence Scale—On-Demand; VERITAS-PRO, Validated Hemophilia Regimen Treatment Adherence Scale—Prophylaxis.
While of proven utility, current hemophilia-specific PRO measures have their associated challenges and knowledge gaps, including sensitivity to changes over time and differences within subgroups (eg, age, geographical region, disease heterogeneity, severity/progression), which have not been conclusively established. 39 There is also the risk of a ceiling effect, defined as a measurement limitation occurring when patients record the highest/close to highest possible score, thereby decreasing the likelihood that the testing instrument has accurately measured the intended domain. 40 Additionally, it is not always clear whether HRQoL correlates with clinical outcomes, given that efficacy of treatment will impact perceived HRQoL 41 ; likewise, patients with hemophilia have reported continued HRQoL impairment despite receiving standard care.42,43 As GT is a promising novel treatment option and HRQoL data for PWH treated with GT are still limited, there is a need to determine whether existing PRO tools are appropriate for the assessment of HRQoL in PWH receiving GT and, moreover, to develop treatment-specific PROs.
Aim
The primary aim of this article was to go beyond disease-specific PRO tools and discuss hemophilia treatment-specific issues (ie, GT) by summarizing what participants of a multistakeholder focus group discussion consider to be the essential aspects of HRQoL. Participants included PWH and researchers. The secondary aim was to communicate some of the participants’ unique perspectives of undergoing a hemophilia A or B GT, including their expectations and goals.
Methods
Study Procedures and Participants
A multidisciplinary advisory board entitled “My gene therapy experience” was held in June 2020 to bring together PWH and hemophilia GT professionals. The group convened to discuss patient expectations throughout hemophilia GT treatment, ways of increasing patient engagement at early stages of the treatment journey, and the need for patient-friendly clinical trial design. The group included 7 PWH A or B who had previously undergone GT as part of a clinical trial, an expert on PROs, and representatives of Bayer. Before the advisory board, all 7 PWH were invited to participate in an in-depth interview, as well as to answer a detailed questionnaire on their experience. The output of this 2-step approach was then discussed in the advisory board.
Participants were invited as authors of the current article; 5 of the 7 PWH who took part in the advisory board accepted this invitation, but all 7 provided informed consent for their input and contributions to the discussion to be presented here. Their ages ranged from 26 to 73 years at the time of treatment, received between 2015 and 2020 (average 5 years before the advisory board). Participants were from Canada, Germany, the United Kingdom, and the United States.
Focus Group Discussion
Hemophilia GT PRO Domains
Participants discussed different hemophilia-specific PRO tools covering the concepts of HRQoL, subjective physical functioning, treatment adherence and satisfaction (summarized in Table 1). Currently used PROs are designed for SOC treatment but not for emerging therapies such as GT. Therefore, the group discussed existing PROs (Haem-A-QoL, EQ-5D-5L, Work Productivity and Activity Impairment Hemophilia Specific (WPAI + CIQ:HS) and the short form of the PROBE)30, 44–46 what key elements new PROs should capture and which HRQoL aspects are crucial to patients. PWH within the group were sent a brief questionnaire based on the topics to be discussed before the advisory board. Key questions to initiate this focus group discussion were:
What are the personal relevant aspects impacted by GT? How do we capture the transformational impact GT has on patient lives?
The elements that should be captured in a specifically designed tool were discussed further by the group with reference to existing PRO measures. Common themes were identified, mapped to PRO domains, and ranked according to personal preference.
Participant Experiences and Drivers of Clinical Trial Participation
The PWH on the advisory board shared their general feelings associated with hemophilia and experiences before and after GT. The key focus group question to initiate the discussion was:
Why are we embarking on GT/what drives patients’ decision to go for it?
As part of a preworkshop exercise, participants were asked to prepare 2 slides where they shared how they defined themselves before and after the initiation of their GT journey. The suggested approach was to generate a collage using pictures from clipart, internet, or other resources. In addition, participants were asked to provide 5 words each describing how they viewed themselves before and after GT treatment. The picture collages and chosen descriptor words were then discussed during the advisory board. They were also asked to identify the key drivers that motivated them to enroll in a hemophilia GT clinical trial.
Results
PRO Domains Identified
Figure 1 depicts the common themes identified by the 7 PWH and their aligned PRO domains. Overall, the ability to participate in sport, physical activity, and leisure, their future condition, and treatment were considered as most important aspects. Existing PRO tools were used as a basis for the discussion, and participants evaluated the importance of common PRO categories to their situation undergoing GT.30, 44–46 However, priorities varied by participants, with, eg, “treatment” listed as a primary priority for some participants but not mentioned by others. Moreover, 1 participant listed “all factors” as important. Pain was generally considered the lowest priority outcome compared with the other lifestyle- or treatment-related outcomes.

Outcomes/endpoints that matter to hemophilia patients. *Domains were rated by each patient based on an index value (1-100) and added for a cumulative score.
Participant Experiences of Hemophilia
Participant experiences before GT are summarized in Table 2. Treatment burden, pain, and barriers to physical activity or employment associated with hemophilia were frequently described. Although feelings before GT were mostly negative, positive sentiments included hemophilia being motivational for self-education, treatment complexity easing over time, and participants generally feeling in control of their own condition.
Summary of Patient Experiences with Hemophilia Before and After GT.
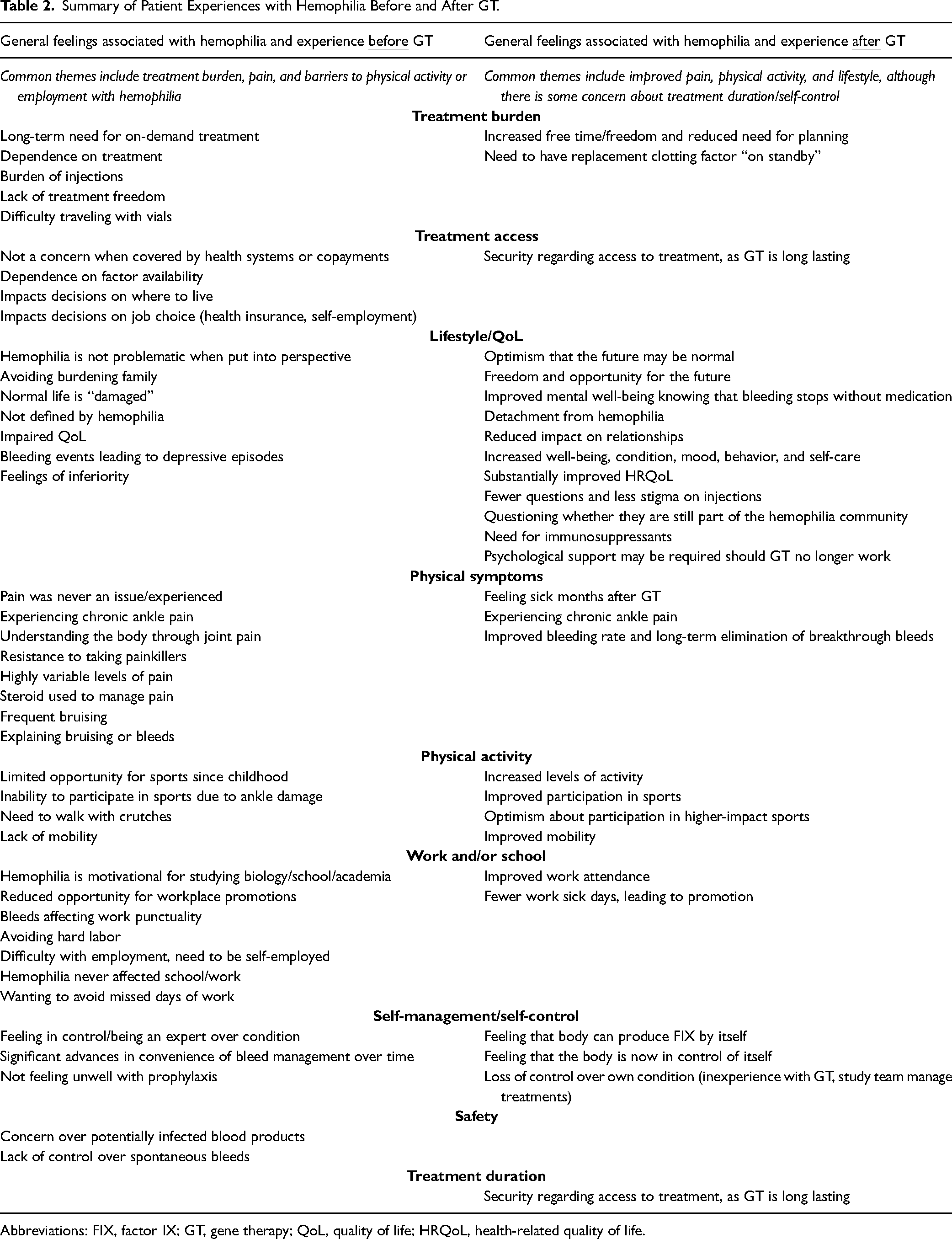
Abbreviations: FIX, factor IX; GT, gene therapy; QoL, quality of life; HRQoL, health-related quality of life.
Participant experiences following GT are also summarized in Table 2. In contrast with feelings before GT, feelings associated with hemophilia in the period after GT were generally positive, with improvements in pain, physical activity, work, and lifestyle noted. The group also generally felt more optimistic about the future and appreciated the feeling of fewer restrictions on work and leisure time. However, some reported anxiety about treatment duration and psychological impacts should GT stop working and noted that they experienced difficulty adjusting to life after GT due to a feeling of losing control over their own treatment. The need to still have replacement clotting factor available as a precautionary measure in case of bleeds was also highlighted. Collage slides summarizing each participant's experience before and after GT were shared with the group; examples of chosen descriptor words are shown in Figure 2.

Summary of descriptor words provided by patients to illustrate their general feelings associated with hemophilia and experiences before (A) and after (B) GT. Red: Negative, blue: Neutral, green: Positive. GT, gene therapy.
Factors driving clinical trial participation vary for every individual, but the focus group discussion identified several common themes. These included excitement about participation in a pioneering trial, the prospect of a new therapy that does not require repeat injections, and benefits from access to the clinical trial team as well as enhanced healthcare treatment and monitoring.
Discussion
The “Patient Voice” in Hemophilia Gene Therapy
PRO Measures
While decision-making bodies have historically marginalized PRO tools and the patient experience data they generate, these are now included in regulatory guidelines to support labeling claims.47,48 Furthermore, PROs can provide comprehensive data and important nuances to inform the risk-benefit profile of a new product, particularly in rare diseases such as hemophilia where trial populations may be smaller than ideal 49 ; they could also provide more accessible and relevant data for PWH to enable shared decision-making. 50 However, studies are needed to develop GT-specific PRO tools in PWH or adapt existing tools for PWH receiving GT. Baseline data may be difficult to interpret as PWH may have adjusted to living with the constraints of hemophilia and will have access to more freedom and activity choice after treatment. Therefore, hemophilia-specific PRO tools should accommodate GT while benchmarking the HRQoL change in people with a lifelong condition, now living without the disease.25,51 This may initially be challenging due to the relatively low numbers of patients with access to GT, 37 but such refinements can hopefully be improved over time as more patients receive this treatment. Progress has already been made in this direction with the coreHEM project, in which a mixed group of stakeholders (including patients and clinicians) identified a core set of important outcome measures for the assessment of GT efficacy, safety, comparative effectiveness, and value in hemophilia. 38
For the PWH who were participants of the advisory board, the most important domains relating to HRQoL were related to physical activity, leisure, self-perception, and the future; however, it is important to highlight the differences between individual priorities. New PRO measures for individualized treatment should be designed to capture the aspects identified here, which in turn may increase patient engagement/acceptance of PRO assessment. 52 This is especially relevant since current approaches (eg, questionnaires, scale completion) are sometimes perceived as burdensome (eg, intrusive/poorly worded) and/or lacking in interest (repetitive/irrelevant). Therefore, a novel, more personalized approach to assessment may boost participation rates. 53 In addition, patient responses to questionnaires may vary by mode (ie, paper-and-pencil form/electronic system) and setting (ie, clinic setting or at home) 53 ; therefore they should be kept consistent, and they should be engaging. Taken together, PRO tools for PWH should accommodate patients who receive GT and should be individualized whenever possible. However, traditional PRO tools for PWH will still be of use for comparisons between different treatment types.
The Patient Experience
Overall, sentiments shifted from negative (before) to positive after GT, with key changes including reduced pain, increased physical activity, and general lifestyle improvements. However, concerns about treatment duration/efficacy and self-control of treatment remain. Physicians and patients will inevitably have different perspectives on the opportunities associated with GT. To help patients make an informed decision about choosing this treatment, physicians gain a clear understanding of what their patients can expect from the process, including benefits and risks based on the most recent available data. 13
Clinical Trial Participation
Consistent with our findings, PWH who have participated in EU- and US-based surveys54,55 showed general support for clinical trial participation despite potential logistical/emotional difficulties, although preferences are likely to vary between individuals. Interestingly, some people were more likely to consider participation in a clinical trial if they were already familiar with clinical research modalities, or after receiving additional information, supporting the importance of education and awareness as a factor influencing patient decision-making. Others explained that the availability of accurate and complete information on the topic of GT and hemophilia was highly important to them and agreed that it is necessary for anyone considering participation in a GT clinical trial to understand the associated risks and benefits. Finally, some participants felt that the success of the GT trial outweighed the personal inconvenience of frequent study site visits/testing and long-term follow-up, indicating a general altruistic interest in supporting treatment advances in this area. This is encouraging, as patient willingness to participate in clinical trials is essential for this treatment to become more established.
Limitations and Future Directions
A few limitations should be considered when interpreting these results. Important PRO domains and patient experiences are derived from a small group and can only provide a snapshot of the key issues relevant to PWH. This small focus group did not discuss their hemophilia diagnosis (A or B) or the specific GT treatments received. While there are many common challenges associated with living with the disease, unique issues are also relevant, highlighting the importance of considering PWH as individuals rather than as a homogenous group. This discussion should be considered only a starting point for future stakeholder discussions.
The topic of GT is a complex and important one for the hemophilia community and society as a whole. There will be ethical considerations that should be explored around issues such as safety and access. The discussion reported here is focussed on HRQoL aspects and broader considerations were not captured. Other topics should also be considered between HCPs and patients in wider conversations on the “patient voice” in hemophilia GT; these are beyond the scope of this article but are detailed briefly here. Notably, the functional implications of GT (eg, reduced bleeding) need to be made clear; ie, patients should understand that they may still experience bleeding (due to pre-existing joint damage) and will still require monitoring even if their condition has been stabilized. Also, factor expression may fall over time and any benefits of GT will not be passed on to future children. 13 In addition to current PRO measures, there has been a recent increase in research into the use of wearable devices and activity trackers in hemophilia. 56 Direct, objective measurement of patients’ physical activity has the potential to complement and contextualize subjective PRO data, although further investigation is needed in this emerging field as there are currently no validated algorithms considered by regulatory authorities that include the use of wearables. Finally, safety aspects need to be communicated transparently to patients and in a timely fashion, as current data are based on a small number of patients and for a limited follow-up period; new safety concerns may also arise as the quantity of clinical evidence increases. 13
Conclusions
In the opinion of this author group (including PWH and psychology/medical/industry experts), the transformational process of GT should be assessed based on elements that are relevant to individual patients. This should include an assessment of the importance of particular PRO domains to the individual, while keeping in mind that, due to their unique treatment perspective, their opinion may differ from that of HCPs or scientists. Existing PRO measures assess common endpoints and are not personalized to individual patients so may fail to capture the complexity of the GT experience. The development of new and individualized PRO measures will hopefully allow the transformational experience of GT to be more accurately captured. Based on the authors’ experiences, PWH generally experience a positive impact on their sense of confidence, freedom, and opportunity to engage in society and physical activity following GT. Moreover, patient attitudes toward participation in GT clinical trials are generally positive. In conclusion, PROs with domains capturing physical activity, treatment type/access, and the future, among others, would allow a more holistic measure of GT success beyond bleeding outcomes.
Supplemental Material
sj-pdf-1-jpx-10.1177_23743735231193573 - Supplemental material for Gene Therapy in Hemophilia: A Transformational Patient Experience
Supplemental material, sj-pdf-1-jpx-10.1177_23743735231193573 for Gene Therapy in Hemophilia: A Transformational Patient Experience by Enayet Rasul, Ryan Hallock, Magnus Hellmann, Jay Konduros, Luke Pembroke, Gregory LeCleir, Jean Malacan and Sylvia von Mackensen in Journal of Patient Experience
Footnotes
Acknowledgments
Editorial support (in the form of writing assistance, including the development of the initial draft based on author direction, collating authors’ comments, grammatical editing, and referencing) was provided by Hanna Mourad-Agha, PhD, and Ewa Kilinska, MSc, and submission support by Beth Lund-Yates of Fishawack Communications Ltd, part of Fishawack Health. The authors would also like to acknowledge Francesca Ferrante, Sharon Eastwood, and Frank Driessler, of Bayer, for organizing and facilitating the meeting and acting as expertise contributors during the meeting and manuscript development.
Conflicts of Interest
GL was an employee of Bayer at the time of the advisory board. JM is an employee of Bayer. SvM served as a consultant or advisor for Sobi, F. Hoffmann-La Roche Ltd/Chugai Pharmaceutical Co., Takeda, and Bayer; served in speakers’ bureaus for Biomarin, Sobi and CSL Behring; received research funding from Sobi and Novo Nordisk; and received travel, accommodations, and expenses from Sobi, Chugai and CSL Behring. ER, RH, MH, JK, and LP declare no potential conflicts of interest to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Bayer (grant number NA).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
