Abstract
What Does This Paper add to the Literature?
With the introduction of straight-to-test CTC and an aging population worldwide, the volume of frail patients being referred direct from primary care will increase. Our study has highlighted the various components of CTC to consider when referring for this test that some patients find difficult.
Introduction
Colorectal cancer is the third most common cancer for both men and women in England (1) and is the second most common cause of cancer-related deaths in the United Kingdom (2). Colonoscopy is the gold-standard investigation for suspected colorectal cancer (3), but computed tomography colonography (CTC), developed in 1994 (4), has now superseded barium enema as the investigation of choice when colonoscopy is unsuccessful or deemed inappropriate (5).
CTC is a radiological investigation used to identify neoplastic lesions in the colon and rectum. The procedure involves the use of abdominal and pelvic computed tomography (CT), enhanced by the use intra-luminal contrast and gaseous distension. Patients are required to perform repositioning maneuvers in the confined space of a CT scanner in order to obtain optimal prone and supine diagnostic images (6).
Frailty describes an important multifaceted medical syndrome in which individuals experience increased vulnerability to dependency and death due to reduced physiological and physical reserve (7). There are several validated models used to screen for frailty, of which the Clinical Frailty Scale, developed by Rockwood et al, provides a simple and rapid assessment tool and is commonly used in the United Kingdom (8). On a scale of 1 (very fit) to 9 (terminally ill), patients who score 5 or higher are considered frail.
CTC has a relative sensitivity to colonoscopy of up to 96% (9) but requires less cathartic preparation (10) and is therefore frequently recommended in frailty, with 25% of patients undergoing CTC being assessed as mild to severely frail (11). It is increasingly recognized that many frail patients struggle with the preparation, gas insufflation, and mobilization required for optimal CTC images.
The demand for CTC investigations has been rising in recent years (12) and direct referral from primary care has been introduced in certain regions of the United Kingdom (13). In addition to this, the COVID-19 pandemic has added pressure to the growing waiting lists of endoscopic and radiological investigations. Appropriate referrals will be vital to help ensure services can address the backlog.
This study aims to better understand the experience and tolerability of patients undergoing CTC in order to better prepare prospective patients and aid clinical decision-making in the investigation of potential colorectal cancer.
Methods
The authors undertook a single-centered, observational cohort study at a large volume tertiary colorectal center. We hypothesized that various aspects of the CTC protocol, aimed at enhancing diagnostic imaging, may have a significant impact on a patient's experience of the investigation.
Survey Tool
A patient experience survey was designed by the authors from the colorectal surgery department, and through collaboration with the trust's patient survey team, was refined to maximize content validity and optimize layout. Following approval for use by the Questionnaire, Interview & Survey Group (QIS), the survey was piloted on 5 patients, and subsequently adapted in consultation with the department of radiology to improve useability. The 13-item survey consisted of 2 Likert scales, 10 discreet questions and the option to expand in 4 free-text boxes and was accompanied by a cover letter. It was designed to investigate various aspects of the patient's experience, including the quality of the pre-test information, overall and specific experience of the test including side effects, discomfort from gas insufflation and difficulty performing the required maneuvers. The full survey is available in Figure 1.

Patient experience survey.
Patient Sampling and Data Collection
All patients who underwent a CTC at the radiology department were eligible to participate in this study. This cohort included patients referred from the outpatient departments, inpatient admissions and direct referrals from primary care. Indications for the investigation included: clinical suspicion of colorectal malignancy where patients were deemed unsuitable for colonoscopy, failed colonoscopy and surveillance imaging of previously identified colorectal pathology. A convenience sample of 41 patients was obtained over a 7-month period. There was an interruption of 12 weeks due to the COVID-19 pandemic, where scanning and data collection was suspended, before the radiology department restarted at a reduced operating capacity.
Patients were invited to participate in the study upon checking into the department. They were then provided with an information leaflet and verbally consented by the radiographer for their anonymous participation in the study. Responses were completed before and after the procedure and collected in a designated box.
CTC Protocol
At 2 days before the scan, patients were instructed to eat a low-fiber diet up until and including lunch time. After this, they were permitted a soft food diet. On the day of the scan, patients could drink clear fluids only. Patients were asked to drink 2 50 mL doses of Gastrograffin® (Bracco Diagnostics Inc.) in the 2 evenings prior to the scan. Intravenous hyoscine butylbromide was administered prior to gas insufflation. An automated carbon dioxide insuflator was used to achieve maximal pressures tolerated by patients. Two positions were used in the scanning protocol: prone and supine, and images were captured on inspiration.
Statistical Analysis
The frequency of responses per question was tabulated and analyzed using descriptive statistics. Questions were analyzed individually, using a denominator equivalent to the total number of fully completed responses. Some results were grouped to aid interpretation of scaled responses. Qualitative free-text responses were coded for content and thematic analysis (14).
Results
There was a response rate of 51%. There were no major complications or unplanned admissions following CTC. Full results are shown in Table 1.
Questionnaire Results.

Quantitative
Overall, the majority of patients (54%) found the investigation better than expected and 100% felt their dignity was maintained. However, 51% reported some form of side effect. Over a third (38%) of these experienced diarrhea from the contrast drink, of which 5 (14%) was severe. Abdominal pain (27%), followed by nausea (16%), were the next most common side effects associated with the scan (Figure 2).
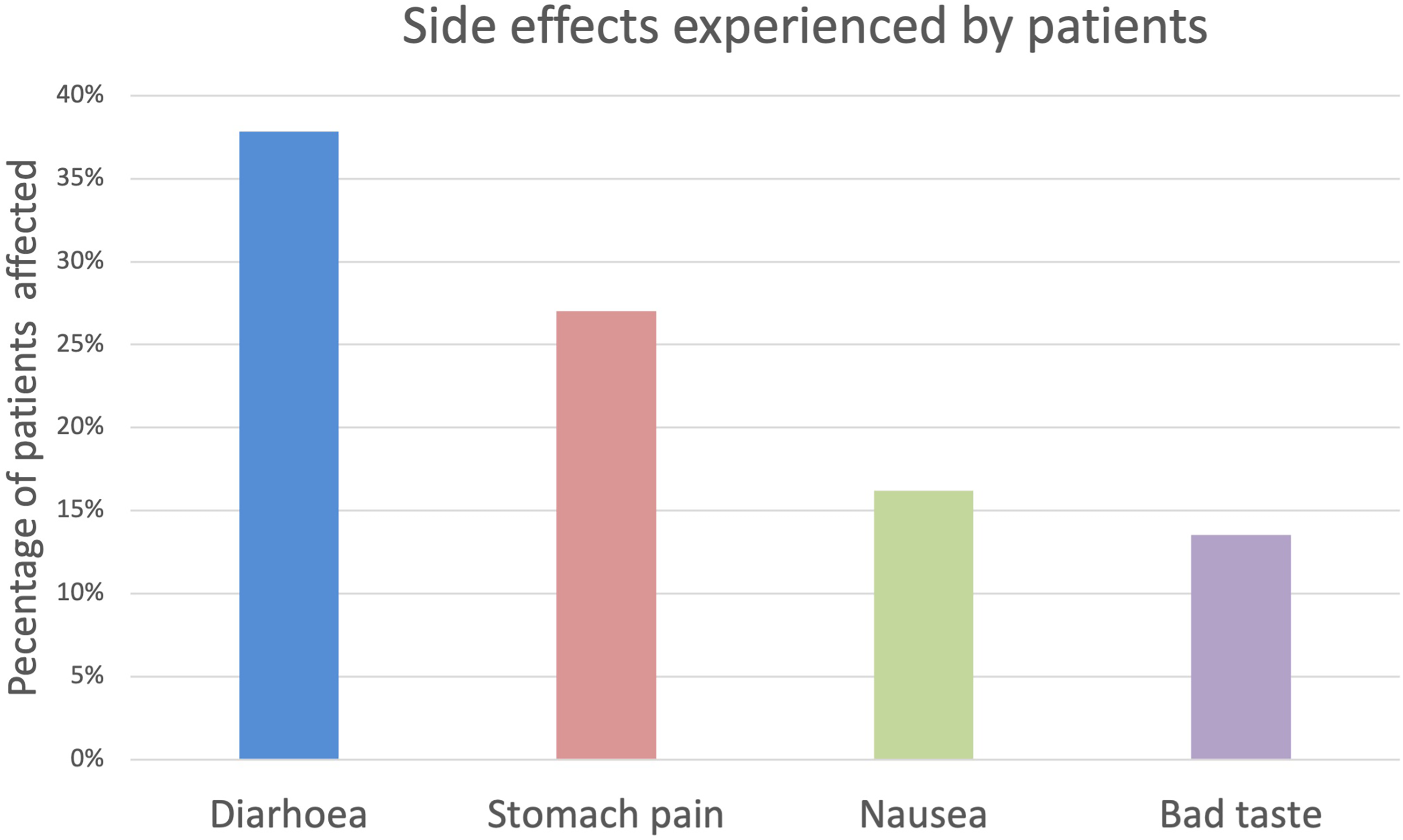
Side effects.
Around half of patients found the tube insertion uncomfortable (43%), and 59% experienced discomfort from the gas insufflation itself. None rated this discomfort extreme, but 50% rated it at a level 2 to 3 out of 5, and 9% 4 out of 5 in severity. Removing the tube was felt to be uncomfortable in only 37% of cases (Figure 3). Regarding mobility issues, 86% had any degree of difficulty performing the turning maneuvers in the scanner, of whom 59% had significant difficulty.

Patient discomfort.
About 97% understood the indication for this test and 95% found their written information helpful. Despite this, 8% felt they were not told what would happen during the procedure, 11% felt they were not informed of the risks and benefits, 9% did not know how to take the contrast dye and 18% were not aware of its potential side effects. Regarding follow-up, 31% were unsure how and when their results would be communicated to them.
Free Text Responses
The majority of free-text responses were shared in the final comments section. The 4 main themes identified reflect the relative importance of aspects identified from the quantitative results.
Discussion and Conclusions
CTC’s use in Frail Populations
In a review completed in our center (9), 25% of patients with a documented Rockwood frailty score undergoing CTC were mild-severely frail. CTC provided a definitive diagnosis in 79% of cases, however 31% of the 306 patients required further investigation with endoscopy to either confirm or rule out a suspicion on the CTC. About 18% of those requiring further investigation had a Rockwood score of 5 or over.
With the introduction of straight-to-test CTC (13) and an aging population worldwide (15), the volume of frail patients being referred direct from primary care will increase. Previous studies have shown good overall tolerability of CTC (16), but our study has highlighted the various components of the test that some patients find difficult which is important for clinicians to consider when referring for CTC.
Whilst protocols for CTCs differ between units, our study has illustrated issues related to different aspects of the preparation and procedure.
Preparation
Fecal tagging is a fundamental stage in all CTC protocols and although supplemental cathartic bowel preparation is no longer used in many centers, the gastrointestinal effects of agents such as Gastrograffin® are often neglected. Our study has found that Gastrograffin® produced a number of frequently reported side effects including abdominal pain and diarrhea. Previous studies have not shown any significant biochemical abnormality (17) caused by fecal tagging agents, however clinicians may need to be aware of the potential risk dehydration may cause to those with reduced renal functions or significant comorbidities.
Emphasis should be placed by the clinical team during the consenting process on the potential effects of fecal tagging agents. Frailty may affect the patient's ability to mobilize to the toilet or administer analgesia when required and practical preparations may need to be made in their home in the period prior to the test, for example extra support or a commode. Whilst minimal preparation CTC protocols have been developed which provide an overall more tolerable investigation (18), it does not entirely prevent potentially significant patient distress.
Scanning Protocol
Adequate colonic distension is important in achieving good quality diagnostic imaging, with one study finding a reduction in false positive rates from 15% to 5% (19) and reduced scanning time (20). Other methods of achieving colonic distension, used in varying combinations, include turning maneuvers and the use of antispasmodic agents, although evidence for the use of the latter is weak (20), and many minimally invasive CTC protocols do not recommend their use.
A significant proportion of our patient population experienced discomfort with the overall use of gas, however the majority of these found the discomfort moderate. We are also unable to draw any direct conclusions regarding the impact of this discomfort on the patients’ overall experience and tolerability.
A significant proportion of our patient population struggled manoeuvering during the investigation, with patients reporting increased body habitus and pre-existing back pain as contributing factors.
Patients with restricted mobility, not otherwise deemed frail, referred for CTC following a failed colonoscopy due to this reason, should be assessed for their ability to complete the required turning maneuvers in the CT scanner. Using a combination of 2 scanning positions has been shown to increase the quality of colonographic imaging (21), although, for those less mobile, it has been suggested this could be using a combination of supine-left decubitus as an alternative to supine-prone, while still achieving adequate test sensitivity.
The issues raised in this study, combined with the fact CTCs are time consuming to perform and report, means other imaging strategies may need to be considered, particularly in the context of rising demand. Alternatives include:
1. Non-Colonography CT
For patients deemed unsuitable for surgical or oncological intervention but where diagnostic information will be aid in advanced and supportive care planning, intravenous contrast-enhanced CT (IVCT) without specialist colonographic protocols can be considered. IVCT has been demonstrated to have a sensitivity of 72.4% for the detection of invasive colorectal cancers but a low sensitivity for polyp detection (10.1%) (22). The inferiority of IVCT should be considered in the context of the patient and that progression from adenoma to adenocarcinoma may take 5 to 8 years (23). If IVCT is used to exclude malignancy, the negative predictive value for invasive colorectal malignancy of 78.9% to 100% (22,24). IVCT can be performed without the need for oral contrast, gas insufflation or patient turning and the scan itself requires less time in the scanner and radiologist reporting time.
CTC is clearly the superior investigation to IVCT in the identification of polyps and colorectal malignancy however for patients in whom invasive investigation or treatment would be intolerable, IVCT could provide adequate prognostic information regarding significant or extensive disease or metastasis. As such, evaluation of the clinical significance of test characteristics in relation to patient-specific needs and involving patients in these discussions will aid in clinical decision-making (25).
2. Patient Tailored CTC Protocols
The number of minimally invasive imaging technique protocols has increased globally in recent years (26). Further evaluation of how frailty affects the tolerance of each aspect of CTC is required in order to develop individualized patient tailored CTC protocols. This could allow us to balance patient experience with adequate diagnostic outcomes.
The data collected in this study contributes to a growing pool of information that referring clinicians can use to prepare and select patients for our current CTC protocols.
Limitations
The study was carried out at a single center and has a relatively small sample size. It should be considered, therefore, that this study may not have captured all experiences of patients undergoing CTC. The volume of patients recruited into the study was restricted, in part, due to the disruption caused by the COVID-19 pandemic that came into effect halfway through data collection. It is also important to consider that the restrictions imposed and anxiety around the virus transmission may have changed the demographics of patients seen in clinic and attending for outpatient investigations. Due to the relatively greater risk of the virus to older populations, our study group may have under-represented those considered more frail. Supplementation with results from a larger study will help to validate the findings presented in this paper. The requirement for personal protective equipment and non-touch interactions with reception staff may also have influenced patient experience but did not disrupt the CTC protocol used in the center.
Footnotes
Author Contribution
All authors contributed to the study design, analysis, and write-up. In addition, GD and BB completed data collection. GD and BB co-authored the manuscript submitted.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymised information to be published in this article.
