Abstract
This study describes health-related quality of life (HRQoL) among older Medicare beneficiaries with hormone receptor-positive (HR+) early breast cancer (eBC). Women aged ≥65 years diagnosed with stage I-III HR+ eBC between 1997 and 2014 using the Surveillance, Epidemiology, and End Results Medicare Health Outcomes Survey Data Resource were included. HRQoL was measured using the Short Form Health Survey including physical/mental component summary (PCS/MCS) scores and subscales. Patient surveys ≤ 24 months post-diagnosis were matched to non-cancer controls. Mean differences in HRQoL were compared using analysis of covariance. Among 1880 HR+ eBC patients versus 5640 matched non-cancer controls, eBC patients surveyed ≤ 6 months post-diagnosis (n = 530) scored lower on component scores (PCS mean difference = 1.6 [95%CI: 0.6-2.6]; MCS mean difference = 2.0 [95%CI: 1.0-3.0]) and multiple subscales. Among women surveyed 19 to 24 months post-diagnosis (n = 402), mean differences in HRQoL were modest (PCS: 1.2 [95%CI: 0.1-2.4]; MCS: −1.5 [95%CI: −2.7 to −0.3]). Most differences in HRQoL following diagnosis of eBC did not indicate statistical significance or minimally important difference, emphasizing that preservation of HRQoL is an important and realistic goal among patients with eBC.
Keywords
Introduction
As breast cancer accounts for 30% of cancer in women, an estimated 290 000 new cases of breast cancer will be diagnosed in the United States in 2022. Early breast cancer (eBC) describes disease that is restricted to the breast, without distant metastasis, and comprises more than 90% of breast cancer diagnoses (1). Among breast cancer molecular subtypes, hormone receptor-positive (HR+) is the most common. Depending on risk of recurrent disease, treatment of HR + eBC may include combinations of surgery, radiotherapy, adjuvant/neoadjuvant chemotherapy, and endocrine therapy (2).
Breast cancer diagnosis and treatment can have significant impact on health-related quality of life (HRQoL) (3, 4). With increasing survival and 5-year relative survival rates of 92% in HR+/human epidermal growth factor receptor 2 negative (HER2−) breast cancer, therapeutic goals have shifted towards maintenance or improvement of quality of life in addition to preventing progression and reducing mortality (5). HRQoL may be a particularly important issue in older women, among whom many experience frailty and comorbidities that affect their breast cancer treatment and prognosis (6). Some research suggests that breast cancer and its treatment can have different impacts on older women as compared to younger women. Among older women, composing the majority of breast cancer survivors, equal or greater value is placed on HRQoL over survival when making treatment decisions given the potential adverse effects of aggressive therapies on aging and functional status (7, 8).
Little is known about HRQoL following the first primary diagnosis of eBC in the real world and there is a need to collect and measure real-world HRQoL endpoints. It is important to understand trends in HRQoL over time after an eBC diagnosis in efforts to determine whether preservation of HRQoL is an achievable goal of therapy in HR+/HER2− eBC. Similarly, it is also valuable to confirm whether patients with breast cancer can potentially reach HRQoL levels that are comparable to those of women without cancer. This comparison to the general population as a reference group can help to reveal potentially meaningful deficits in HRQoL among patients diagnosed with eBC that can be targeted by interventions aimed at improving HRQoL (9).
This study aimed to describe HRQoL among older Medicare beneficiaries within 2 years following diagnosis of HR + eBC and compare HRQoL between patients with HR + eBC and patients without cancer over 2 years following breast cancer diagnosis.
Methodology
Briefly, this study was a secondary analysis using a database linking cancer registry data with a longitudinal survey on HRQoL administered to Medicare beneficiaries. Among older women diagnosed with eBC, we describe differences in composite responses to HRQoL between cancer patients following diagnosis and matched non-cancer controls.
Study Design & Data Source
This was a population-based retrospective cohort study conducted using the Surveillance, Epidemiology, and End Results (SEER)-Medicare Health Outcomes Survey (MHOS) data resource. SEER-MHOS, sponsored by the National Cancer Institute (NCI) and the Centers for Medicare and Medicaid Services (CMS), combines population data consisting of detailed information about adults ≥65 years of age with cancer (10).
SEER cancer registries capture clinical, demographic, and cause of death data for patients with cancer. The MHOS survey provides patient self-reported data regarding HRQoL, demographic, socioeconomic, and clinical information among Medicare Advantage Organization beneficiaries (10, 11). The SEER-MHOS linkage used in this study includes 14 MHOS cohorts (baseline and follow-up surveys from 1998 to 2014) as well as cancer data from SEER (for patients diagnosed with cancer from 1973 to 2014) for all patients included in the SEER-MHOS linked file (over 140 000 survey respondents with cancer and survivors) (12).
This study was approved by the institutional review board of the University of Illinois at Chicago and was determined to be exempt from human subjects research requiring informed consent (#2019-0170).
Study Population
The study cohort consisted of women aged ≥65 years with stage I-III HR + breast cancer (HER2 status is not consistently available in the SEER-MHOS data source). Patients were required to have completed at least one MHOS survey between 1998 and 2014 that was within 24 months post-eBC diagnosis. Therefore, eBC diagnoses among our sample could have occurred between 1996 and 2014. The first completed survey following eBC diagnosis was utilized in our analysis. Patients were excluded if their date of breast cancer diagnosis was missing, if the stage of breast cancer was unknown, or if they were missing primary outcome (i.e., HRQoL) data.
The control group was drawn from women without cancer who were also identified in the SEER-MHOS linked database. NCI and CMS created a file from the linked database in which patients who responded to at least one MHOS survey in a SEER area, were not identified in SEER, and self-reported no previous cancer diagnosis were included as non-cancer controls (10, 12, 13). We excluded non-cancer patients who were male, under 65 years old, or missing HRQoL data. Similar to the eBC group, non-cancer patients must have completed at least one MHOS survey between 1998 and 2014 and the earliest completed survey within this timeframe was utilized in our analysis.
Study Variables
Several categorical MHOS survey variables were used to describe both patient groups including age at survey, race/ethnicity, marital status, number of comorbid conditions, presence of diabetes, cardiovascular disease, education received, annual household income, plan type, geographic region, and smoking status.
Breast cancer-specific variables were described including year of diagnosis (1996-1999, 2000-2006, 2007-2013), age at diagnosis (65-69, 70-74, 75-79, ≥80), tumor size in centimeters (≤2, >2 and ≤5, >5), nodal status (+/−), overall American Joint Committee on Cancer (AJCC) stage (I, II, III), and treatment patterns of radiation therapy and surgery (breast conserving, mastectomy).
Following prior approaches, several other quality of life variables were also determined to characterize patients with eBC and non-cancer patients (14–16). The deficit-accumulation frailty index (DAFI) was calculated, with scores ranging from 0.0 to 1.0, with 0.0 being no deficits (16). Established categories of the DAFI are robust (0 to <0.2), pre-frail (0.2 to < 0.35), and frail (≥0.35 to 1) (17). Presence of depressive symptoms was determined by an affirmative response to any one of the following questions: (1) In the past year, have you had 2 weeks of depression? (2) In the past year, have you had depression much of the time? (3) in the past 2 years, have you had depression most of the time? Lastly, self-reported health was assessed using a single MHOS question asking: “In general, would you say your health is: excellent, very good, good, fair, or poor?” in which patients selected one response option.
Primary Outcome
The primary outcome was HRQoL, which was measured using the 36-Item Short Form Health Survey (SF-36) and the Veterans RAND 12-Item Health Survey (VR-12). Algorithms developed by Boston University School of Public Health for the conversion of SF-36 to VR-12 scores were used on the SEER-MHOS data resource to account for changes in the survey instrument over time (18).
The VR-12 is a brief, generic health survey comprised of 12 items that can be summarized into 8 subscales scores including physical functioning (PF), role limitations due to physical problems (RP), bodily pain (BP), general health perceptions (GH), general mental health (MH), role limitations due to emotional problems (RE), social functioning (SF), and vitality (VT). The physical component summary (PCS) and mental component summary (MCS) scores utilize all 8 scales in their calculation and are computed from weights derived from the 1999 Veterans Health Study. These scores are a linear transformation of the 0-100 possible range scoring for the SF-36/VR-12 scales that gives every scale a mean of 50 and standard deviation (SD) of 10, normed to the US general population.
Statistical Analysis
Patient surveys were exact-matched (3:1) to non-cancer controls’ surveys on multiple demographic factors, including age at survey, race, education, marital status, smoking status, patient/proxy respondent, number of comorbid conditions, SEER reporting region, and survey year (all variables are categorical).
Demographic and clinical characteristics for the matched patient groups were summarized using descriptive statistics. Between-group comparisons were assessed using chi-square and Fisher's exact tests for categorical variables and t-tests and Wilcoxon rank-sum tests for continuous variables. The Bonferroni method was used to correct for multiple comparisons. No imputation of missing values was performed for the linked SEER-MHOS surveys. Values for responses as none/unknown/missing were reported as appropriate.
HRQoL scores were described by mean and SD for both study groups across each 6-month interval following eBC diagnosis up to 24 months (≤6 months; >6 and ≤12 months; >12 and ≤18 months; >18 and ≤24 months). HRQoL scores were then compared between patients with eBC and exact-matched non-cancer patients for each 6-month interval. Mean differences in HRQoL scores were determined using analysis of covariance (ANCOVA) models and interpreted in the context of published minimally important differences (MID) (PCS: 2-3 points; MCS: 3 points; PF: 2 points in the score range below 40 and 3 points in the score range at or above 40 points; RP: 2 points; GH: 3 points; BP: 2 points in the score range below 40 and 3 points in the score range at or above 40 points; VT: 2 points in the score range below 40 and 3 points in the score range at or above 40 points; MH: 3 points; RE: 4 points; SF: 3 points) (19). ANCOVA models were adjusted for plan type and clustered by match ID. Mean differences and 95% confidence intervals (CI) in PCS, MCS, and subscale scores between study groups were reported for each 6-month interval following eBC diagnosis up to 24 months.
Results
1880 patients with first primary eBC met our inclusion criteria (Supplemental Figure 1). Table 1 reports the breast cancer-specific clinical and tumor characteristics among the women with first primary eBC only. The 1880 eBC patients were compared to 5640 matched non-cancer controls. Table 2 presents general clinical and demographic characteristics of women with first primary eBC and their matched non-cancer controls.
Clinical Characteristics of Older Women With HR + eBC at Diagnosis (N = 1880).
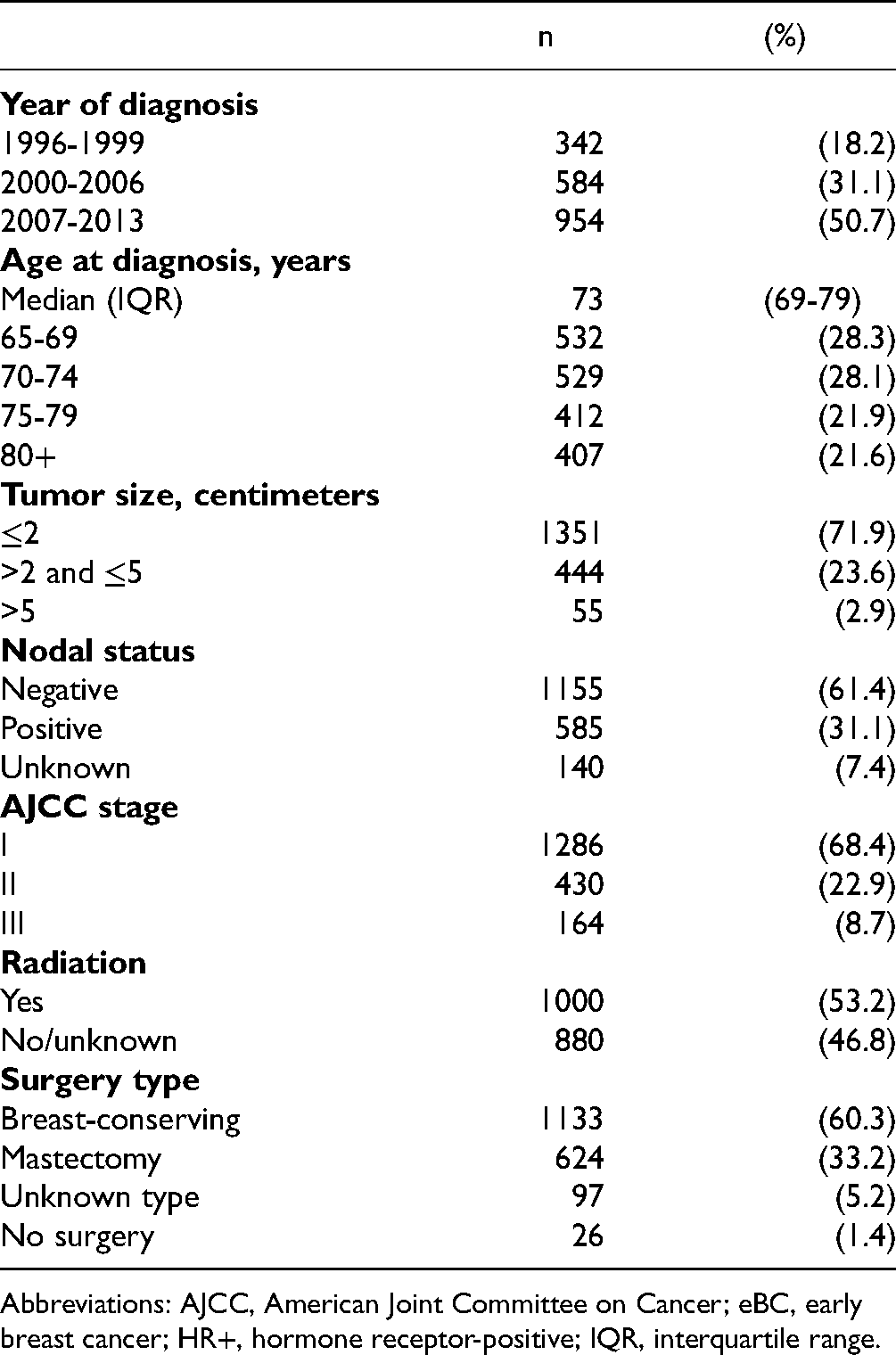
Abbreviations: AJCC, American Joint Committee on Cancer; eBC, early breast cancer; HR+, hormone receptor-positive; IQR, interquartile range.
Clinical and Demographic Characteristics of eBC Patients and Matched non-Cancer Controls at the Time of Survey.

Note: Between-group comparisons were assessed using chi-square and Fisher's exact tests for categorical variables (Wilcoxon rank-sum test was used for median age). The Bonferroni method was used to correct for multiple comparisons. Therefore, α = 0.003.
Abbreviations: CMP, competitive medical plan; DAFI, deficit-accumulation frailty index; eBC, early breast cancer; GED, general educational development; HMO, health maintenance organization; HMO-POS, health maintenance organization-point of service; HR+, hormone receptor-positive; IQR, interquartile range; PPO, preferred provider organization.
Number of comorbid conditions totaled from presence/absence of: angina pectoris/coronary artery disease, congestive heart failure, myocardial infarction, other heart conditions, stroke, emphysema/asthma/chronic obstructive pulmonary disease (COPD), Crohn's disease/ulcerative colitis/inflammatory bowel disease, arthritis of the hip/knee, arthritis of the hand/wrist, sciatica, diabetes/high blood sugar/sugar in urine, and hypertension. If patient is missing data on at least one comorbidity, then number of comorbid conditions is considered missing.
Higher frailty index indicates more deficits. Score ranges from 0.0 to 1.0, with 0.0 being no deficits. Robust: 0.0 ≤ DAFI < 0.2; Pre-frail: 0.2 ≤ DAFI < 0.35; Frail: 0.35 ≤ DAFI≤1.
Columns do not sum to total sample size due to missing values.
*P < .003.
Survey Characteristics Among eBC and Non-Cancer Cohorts (Data not Shown)
The median number of months from first primary eBC diagnosis to time of survey was 11 months. In both patient groups, over 80% of MHOS surveys were conducted by mail and the rest by telephone. In both groups, 8.8% of surveys were completed by a person other than the patient. Most (58.7%) of the surveys in both patient groups were conducted between 2006 and 2014.
HRQoL Scores Among eBC and Non-Cancer Cohorts
Table 3 shows the HRQoL scores (mean, SD) for both patient groups across each 6-month interval following eBC diagnosis up to 24 months. Figure 1a–d illustrates the differences in mean scores and corresponding CI between the 2 patient groups (a) within the first 6 months of diagnosis, (b) within 6 to 12 months of diagnosis, (c) within 12 to 18 months of diagnosis, and (d) within 18 to 24 months of diagnosis. Compared to matched non-cancer controls, eBC patients surveyed within 6 months post-diagnosis (n = 530) demonstrated presence of MID on 2 scales compared to non-cancer patients: mean difference in RP was 3.6 (95% CI: 2.5, 4.7); mean difference in SF was 3.0 (95% CI: 1.9, 4.2). Between 6 and 12 months post-diagnosis (eBC patients = 497), only RP (mean difference = 2.0 [95% CI: 0.9, 3.1]) reached MID. Between 12 and 18 months post-diagnosis (eBC patients = 451), PCS (mean difference = 2.0 [95% CI: 0.8, 3.1]) and RP (mean difference = 2.4 [95% CI: 1.2, 3.7]) reached MID. Among women with eBC surveyed between 18 and 24 months post-diagnosis (n = 402), differences in component summary scores between patients with eBC and non-cancer controls were modest: mean difference in PCS was 1.2 (95% CI: 0.1, 2.4), and mean difference in MCS was −1.5 (95% CI: −2.7, −0.3). After 18 months, differences in subscale measures also attenuated.
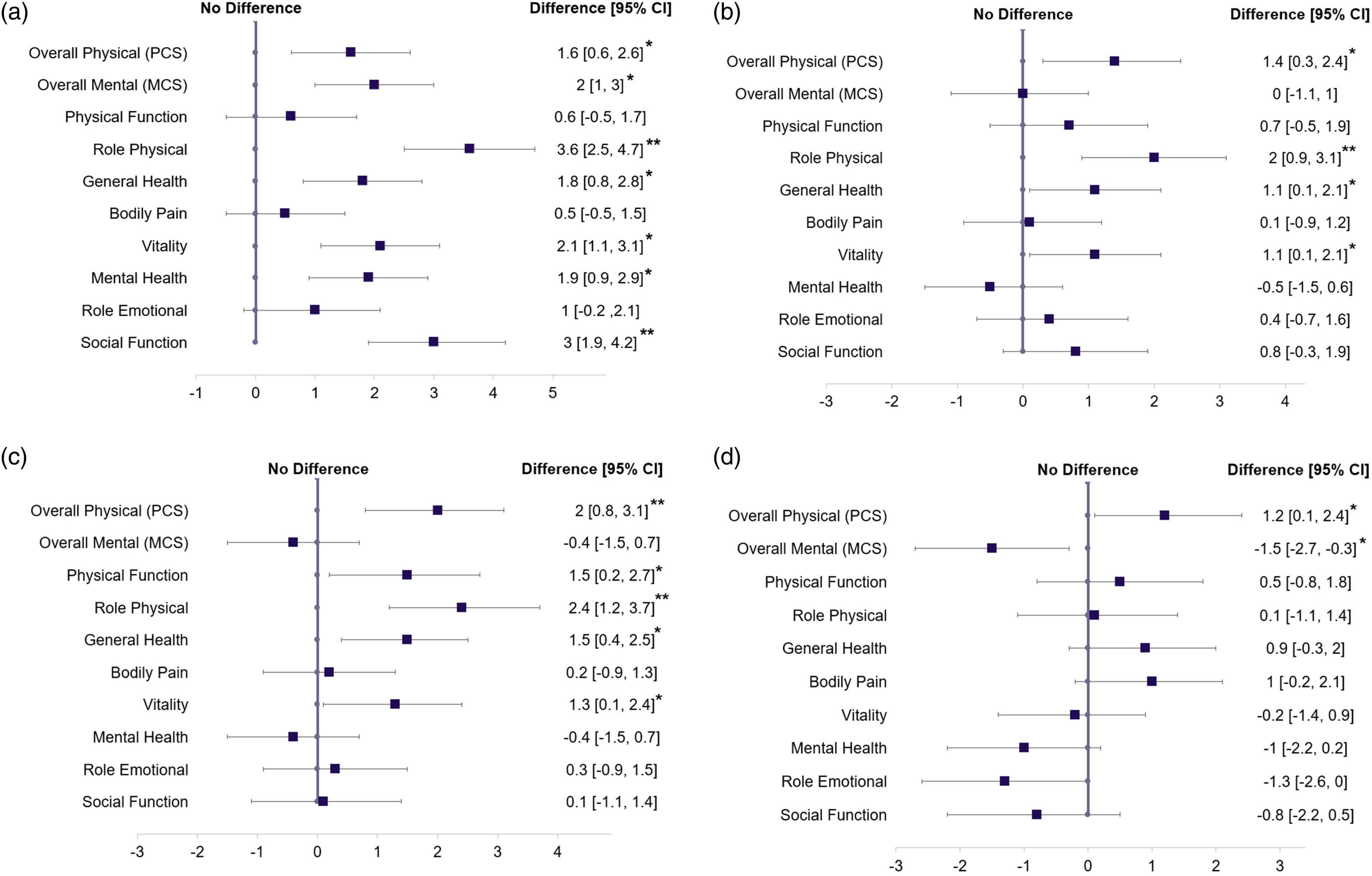
Differences in mean VR-12 HRQoL scores between HR+ eBC patients versus matched non-cancer controls surveyed (a) within 6 months after diagnosis; (b) 6-12 months after diagnosis; (c) 12-18 months after diagnosis; (d) 18-24 months after diagnosis.
Mean VR-12 Scores for Women with HR+ eBC Versus Matched Non-cancer Controls.

Note: This table displays T-scores—linear transformation of the 0-100 versions of the scale that yields a population mean of 50 and a standard deviation of 10, based on 1990 population norms; higher scores denote better health. Exact-matched 3:1 ratio on age at survey, race, education, marital status, smoking status, patient/proxy respondent, comorbidities, region, and survey year (all variables are categorical).
Abbreviations: eBC, early breast cancer; HR+, hormone receptor-positive; MCS, mental component summary; PCS, physical component summary; VR-12, Veterans RAND 12-Item Health Survey.
Discussion
The present study examined differences in HRQoL between eBC cases and matched non-cancer controls in the 2 years following diagnosis. Although statistically significant differences in HRQoL scores for PCS, PF, RP, GH, VT, and SF components and subscales were observed between patients with eBC and matched non-cancer controls, few differences were considered minimally important. These differences in HRQoL scores also varied depending on when women with HR+ eBC were surveyed after diagnosis. SF subscale scores exceeded the MID threshold, but only during the first 6 months post-diagnosis (SF mean difference ≤6 months: 3.0 [95% CI: 1.9, 4.2]). Lower PCS scores and RP subscale scores among older HR + eBC patients compared to similar women without cancer were statistically significant and sometimes exceeded the MID thresholds during the first 18 months post-diagnosis (PCS mean difference ≤6 months: 1.6 [95% CI: 0.6, 2.6]; PCS mean difference 6 to 12 months: 1.4 [95% CI: 0.3,2.4]; PCS mean difference 12 to 18 months: 2.0 [95% CI: 0.8, 3.1]; RP mean difference ≤6 months: 3.6 [95% CI: 2.5, 4.7]; RP mean difference 6 to 12 months: 2.0 [95% CI: 0.9, 3.1]; RP mean difference 12 to 18 months: 2.4 [95% CI: 1.2, 3.7]) however, during months 19 to 24 post-diagnosis, no MID in these physical HRQoL scores were observed.
To our knowledge, this is the first study to examine HRQoL at different time points following eBC diagnosis among older women in a real-world setting in the United States. Similar to our findings, a Korean prospective cohort study of HRQoL among 206 survivors of breast cancer (primarily eBC) found that SF was among the most significantly impaired domains compared to the general population both immediately and 1 year after diagnosis. However, PF was comparable between survivors and the general population at both time points, as observed in our cohort as well (9). We also found that SF among women with eBC became more similar to that of the general population as time since diagnosis increased. This finding is in accordance with another study of quality of life among a cohort of about 300 Australian women, in which respondents reported significant improvements in SF between 3 and 12 months after surgery for eBC (20). Our study builds upon these by focusing on older women with eBC and by examining HRQoL up to 2 years following diagnosis, when the negative transient effects of cancer treatments on HRQoL may subside and treatment benefits may be experienced (20–25). However, we cannot conclude that changes in HRQoL are due to treatment itself, as we did not examine HRQoL among patients with untreated eBC. Additionally, breast cancer can be treated with a multimodal treatment strategy, with different therapies having different effects on HRQoL (26). Examining the effects of specific breast cancer treatments or lack of treatments on HRQoL was beyond the scope of our study and remains an important topic for future research among patients with eBC. However, since we did not observe long-term detriments in HRQoL among our sample, who almost all received at least one type of cancer treatment, our results support the idea that preservation of HRQoL is an important and realistic goal of therapy among older patients with eBC and standard therapies should not be withheld from this population (24, 25, 27).
Several studies have revealed the linkage between cancer recurrence and metastasis and the reduction in quality of life among eBC patients. Meisel et al (28) conducted a cross-sectional quality of life survey of women living with long-term metastatic breast cancer. The study found that although women tended to be physically well after long-term treatment, many faced significant emotional challenges, which led to reductions in HRQoL (28). In a cross-sectional study by McClelland et al (29) aimed at identifying factors affecting HRQoL in a sample of patients diagnosed with metastatic breast cancer, reduced quality of life was significantly associated with body image changes and active disease symptoms. Due to the negative impact of breast cancer metastasis on HRQoL, there is potentially a need for new treatments that help maintain patients’ HRQoL while preventing cancer recurrence and metastasis.
Our findings have implications for practicing clinicians as well as directions for future research. With the advancements in treatment of HR+ eBC, there is a need for more longitudinal studies capturing patient-centered experiences throughout the breast cancer treatment journey from early diagnosis, recurrence and progression to metastatic disease. Clinical studies of new therapies to prevent breast cancer recurrence in women with high-risk disease should also evaluate and report whether life-extending treatment also preserves HRQoL.
This analysis has strengths, especially the extensive and robust population-based information included in SEER-MHOS data. We were able to account for important factors such as patient sociodemographics and comorbidities that can impact HRQoL. Furthermore, this linked data source captures both HRQoL data and tumor information. Additionally, this study is among the first to examine HRQoL over a 2-year period following HR+ eBC diagnosis, which is important since HRQoL can change over time (30).
Our study also has limitations. First, SEER-MHOS has information on specific pre-existing chronic conditions reported in the health outcomes survey but lacks other comorbidities and more detailed information on the severity of these conditions. Therefore, information on other common conditions among older patients such as osteoporosis, hepatitis C virus, or dementia was not available. Although SEER-MHOS provides detailed information on cancer site, stage, sociodemographic factors, and HRQoL, it lacks information on clinical and prognostic variables specific to eBC, such as specific treatments received (including endocrine therapy and chemotherapy, which may have impacts on HRQoL), HER2 status, psychosocial factors, and disease risk at diagnosis. Due to the lack of information regarding specific treatments and the lack of a comparison group of eBC patients without treatment, changes in HRQoL among beneficiaries cannot be concluded to be due to therapy alone. Also, the Medicare Advantage enrollees that participated in the MHOS survey during or immediately following breast cancer treatment may not be entirely representative of all Medicare fee-for-service beneficiaries. Lastly, we sought to describe HRQoL following first primary diagnosis of eBC, and therefore did not aim to detect statistically significant changes in HRQoL over time, as this was beyond the scope of our study. However, this remains an important topic for future research.
Conclusion
In this study of older female Medicare beneficiaries diagnosed with HR+ breast cancer, most differences in HRQoL compared to similar women without cancer were not large enough to indicate statistical significance or MID. These findings emphasize that preservation of HRQoL is an important and realistic goal of therapy in HR+ eBC.
Supplemental Material
sj-docx-1-jpx-10.1177_23743735221113058 - Supplemental material for Health-related Quality of Life in Hormone Receptor-Positive Early Breast Cancer: Analyses From the Surveillance, Epidemiology, and End Results Medicare Health Outcomes Survey
Supplemental material, sj-docx-1-jpx-10.1177_23743735221113058 for Health-related Quality of Life in Hormone Receptor-Positive Early Breast Cancer: Analyses From the Surveillance, Epidemiology, and End Results Medicare Health Outcomes Survey by Nadia A Nabulsi, Khatija W Naing, Huiwen Deng, Jenilee Cueto, Alemseged A Asfaw, Colin C Hubbard, Jifang Zhou, Inyoung Lee, Debanjali Mitra, Gregory S Calip and Ernest H Law in Journal of Patient Experience
Footnotes
Acknowledgments
This study used data from the SEER-MHOS linked data resource. The interpretation and reporting of these data are the sole responsibility of the authors. The authors acknowledge the efforts of the National Cancer Institute; the Centers for Medicare & Medicaid Services; Information Management Services (IMS), Inc.; and the Surveillance, Epidemiology, and End Results (SEER) Program tumor registries in the creation of the SEER-MHOS database.
Availability of Data and Materials
Requests for datasets used and/or analyzed during the current study were used under a limited data use agreement at the University of Illinois at Chicago. Analytic data programs and sources are available by directly contacting the corresponding author.
Authors’ Contributions
NAN: substantial contributions to the conception or design of the work; acquisition, analysis, or interpretation of data for the work; drafting the work and revising it critically for important intellectual content; and final approval of the version to be published. KWN: acquisition, analysis, or interpretation of data for the work; and final approval of the version to be published. HD: acquisition, analysis, or interpretation of data for the work; and final approval of the version to be published. JC: substantial contributions to the conception or design of the work; acquisition, analysis, or interpretation of data for the work; drafting the work and revising it critically for important intellectual content; and final approval of the version to be published. AAA: acquisition, analysis, or interpretation of data for the work; and final approval of the version to be published. CCH: acquisition, analysis, or interpretation of data for the work; and final approval of the version to be published. JZ: acquisition, analysis, or interpretation of data for the work; and final approval of the version to be published. IL: acquisition, analysis, or interpretation of data for the work; and final approval of the version to be published. DM: substantial contributions to the conception or design of the work; acquisition, analysis, or interpretation of data for the work; drafting the work and revising it critically for important intellectual content; and final approval of the version to be published. GSC: substantial contributions to the conception or design of the work; acquisition, analysis, or interpretation of data for the work; drafting the work and revising it critically for important intellectual content; and final approval of the version to be published. EHL: substantial contributions to the conception or design of the work; acquisition, analysis, or interpretation of data for the work; drafting the work and revising it critically for important intellectual content; and final approval of the version to be published. All authors read and approved the final manuscript.
Ethics Approval and Consent to Participate
Approval to conduct this study was obtained from the University of Illinois at Chicago Institutional Review Board 2019-0170.
Consent for Publication
National Cancer Institute; the Centers for Medicare & Medicaid Services; Information Management Services (IMS), Inc.; and the Surveillance, Epidemiology, and End Results (SEER) Program.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NAN, KWN, HD, AAA, CCH, JZ, IL, and GSC were employees of UIC which received financial support from Pfizer in connection with the development of the manuscript. JC, DM, and EHL are shareholders and were employed by Pfizer Inc during manuscript preparation.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was sponsored by Pfizer Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
