Abstract
There are numerous opportunities for shared decision-making (SDM) in cystic fibrosis (CF) care, yet little is known about patients’ SDM experiences. This study evaluated SDM across 159 CF care programs (4024 participants) in the United States. Shared decision-making was assessed using the patient-reported
Introduction
Shared decision-making (SDM) is a process where patients and clinicians share information and make care decisions together (1). Cystic fibrosis (CF) provides numerous SDM opportunities, since it is a complex genetic, multi-organ lifetime disease (2) with emerging new therapies (3). People with CF are typically seen 4 or more times a year by a multidisciplinary care team and engagement of SDM at each visit may best ensure that treatment decisions and care plans meet the personal needs and goals of patients with CF. Evaluating decision-making experiences among patients with CF and their families would help CF clinical teams determine whether and where improvement is needed.
A brief, patient-reported measure of SDM, “
In CF care, the SDM experience is largely unknown, including whether SDM varies relative to patient or program characteristics. The CF Foundation accredits CF programs across the United States to ensure that people with CF have access to high-quality, specialized care. For teams to receive feedback from patients and families, the CF Foundation supported the national Patient and Family Experience of Care Survey (PFEC) (10), and in 2018, the survey introduced
Method
Design and Setting
This is a cross-sectional secondary research study that utilized existing data collected from the PFEC, a quality improvement activity (informed consent was optional and not required). The PFEC was voluntarily implemented at 170 accredited CF programs (located at teaching and community hospitals) across the United States. There were 3 types of CF programs: (1) Pediatric programs providing care to patients younger than 18 years and some adults, (2) Adult programs providing care to adults, and (3) Affiliate programs providing care to patients of any age in underserved areas. The CF Foundation commissioned a third-party survey vendor, Quality Data Management Inc. (QDM), to manage the PFEC data collection. This study utilized PFEC data captured between July 2018 and December 2019.
Data Collection
The survey vendor worked with CF programs to obtain each patient’s contact information and clinic visit dates. Patients became eligible for a survey invitation after a clinic visit, and no more than twice a year. Eligible adults with CF completed an adult survey and parents (or caregiver) of a child with CF completed a pediatric survey. Both pediatric and adult surveys had the same questions with the inclusion of “your child” in some pediatric questions (see Supplementary File 1).
Respondents completed PFEC in one of 3 ways: online at home, telephone at home, or tablet computer in the clinic. For online and telephone at home, an invitation to voluntarily complete a survey was sent by email, if available, or by post. The invitation contained the survey URL and telephone number to complete the survey by personal telephone interview or speech recognition system. Up to 3 reminder telephone calls were made to nonresponders and they were placed on a recontact list (survey sent again after a clinic visit in 4 months or later) if they had not completed a survey within 30 days. In rare instances, a respondent completed the survey more than twice per year, if they completed both an initial and recontact survey. For programs that chose to deliver the survey via tablet in the clinic, a clinic staff member asked the patient or family member to complete the survey toward the end of their visit (see Supplementary File 2 for detailed protocol).
Measures
The PFEC had 28 questions in total (23 closed-ended, 5 open-ended). Of these, the CF Foundation developed 19 questions and 9 were preexisting measures. Additional clinic-reported variables were also merged with PFEC data, to provide additional information about patients and their clinic.
This study focuses on
Other patient/caregiver-reported variables utilized for this study included patients’ years of relationship with their clinic, assessed via a single self-developed item with response options of “Less than 2 years,” “2 to 5 years”, or “Greater than 5 years.” Patients’ general (“overall”) health status and mental (“mental or emotional”) health status were both assessed via single item measures adopted from the Hospital Consumer Assessment of Healthcare Providers and Systems survey (13). Both items had response options of “Excellent,” “Very good,” “Good,” “Fair,” and “Poor.” The CF programs reported their program type and patients’ age-group and gender (see Supplementary File 1 for all measures).
Statistical Analysis
The analysis only included respondents who completed all 3
For study aim 1, chi-square analyses compared the overall percentage of
Two sensitivity analyses were conducted for the multivariable models. First, with respondents’ first or only survey with complete data on
Results
Characteristics of Respondents
There were a total of 5263 surveys completed at pediatric and adult CF programs. Of these, 4024 surveys (76%) had complete data on
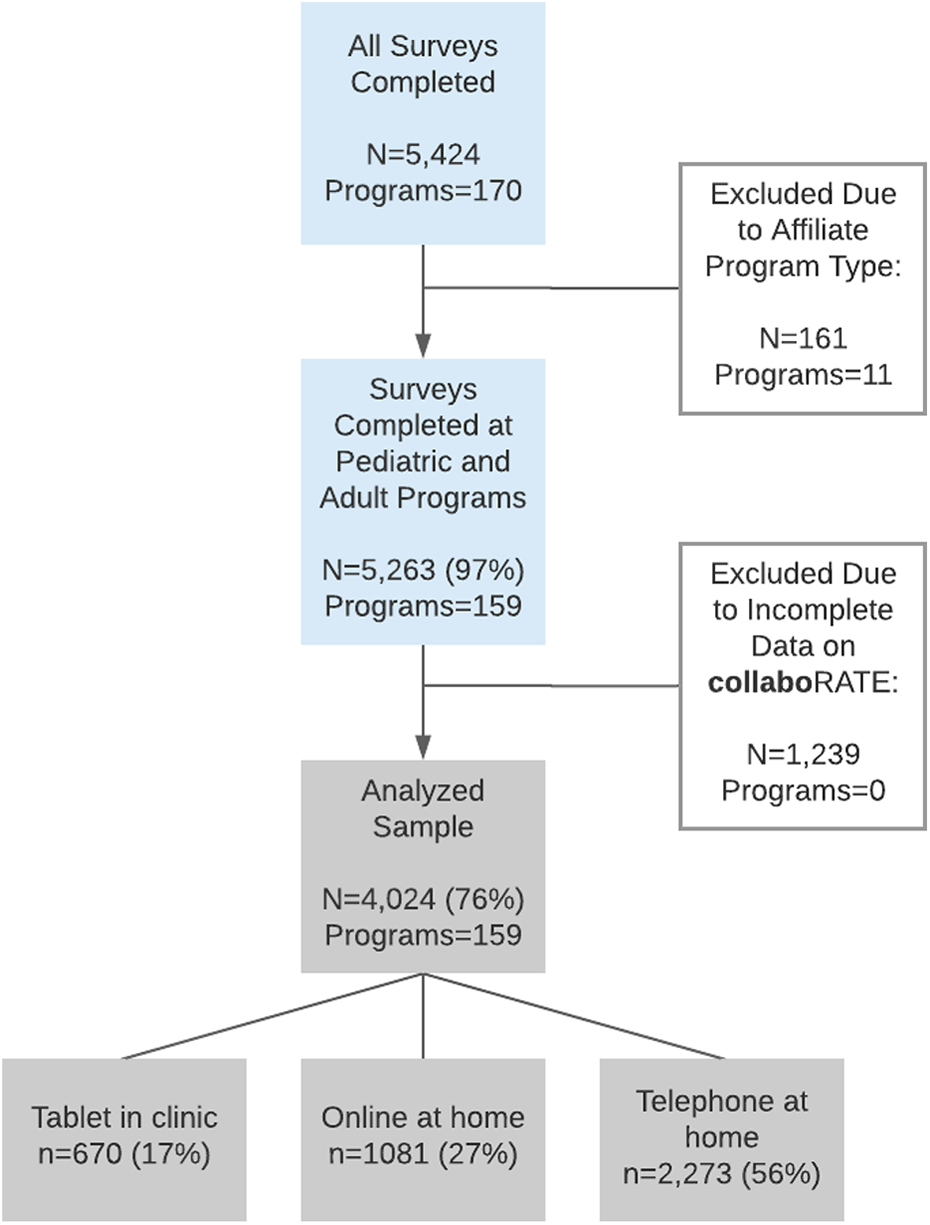
Participant flowchart.
Characteristics of Respondents Who Completed

a 95% CIs not presented for age groups as comparisons were deemed unnecessary due to program type inclusion being largely age-dependent.
b Significant difference across program type.
Compared to national CF Foundation Patient Registry (CFFPR) data for 2019 (16), the study sample as a whole was representative of gender, but underrepresented patients 18 years and over (52.4% vs 56.0%, P < .001). CF Foundation Patient Registry data were not available for direct comparisons of other characteristics in the PFEC. However, adults in the CFFPR had higher rates of screening positive for anxiety disorder and depression, than those aged 12 to <18 years (24% vs 5% and 28% vs 4%, respectively), which mirrors trends in self-reported mental health in the PFEC study sample.
Overall collaboRATE Scores
Sixty-nine percent (n = 2794) of all respondents reported a
Association of collaboRATE Scores With Patient Characteristics, by Program Type
Table 2 details result from the univariable and multivariable logistic regression analyses. For the pediatric programs, there were significant univariable associations with
Univariable and Multivariable Logistic Regression Analyses: Association of

Abbreviations: OR, odds ratio; ref, reference group.
a P < .001.
For the adult programs, there were significant univariable associations with
collaboRATE Variation by Individual Program
There were 35 pediatric programs (n = 1559) and 25 adult programs (n = 1329) with 25 or more survey responses. Compared to the

Sensitivity and Subgroup Analyses
The analysis of respondents’ first or only survey showed minor differences to the main multivariable models and these pertained to patients’ age and general health (see Supplementary File 3). The analyses by survey completion mode revealed some significant associations consistent with the main multivariable models pertaining to age, and mental and general health, for surveys completed via telephone and online at home. Age was also a stronger predictor in the telephone completion group and there were no significant associations in the tablet completion group (see Supplementary File 4).
The subgroup analysis of 18- to 24-year-old respondents showed that those with a length of relationship of 2 to 5 years (OR: 2.1, CI: 1.1-4.1) were more likely to report a
Discussion
Key Findings
In this study, we sought to identify the overall and program-level experience of SDM in CF care, as well as whether patient characteristics were associated with higher or lower rates of SDM. We found variation in SDM experiences across individual CF programs, program types, survey completion mode, and that patient characteristics were associated with SDM. At both pediatric and adult programs, there were lower rates of SDM among respondents reporting fair or poor mental health, compared to those reporting better mental health. A similar pattern occurred for general health at pediatric programs. At both pediatric and adult programs, there were lower rates of SDM among respondents aged 18 to 24 years, than in some other age groups. Among the subgroup of respondents aged 18 to 24 years specifcally, there were also associations between SDM and length of relationship, general health, and mental health.
Context and Implications
This is the first study to conduct a large-scale examination of
Despite a tendency for people in this study reporting more health burden to also report poorer SDM experiences, it is perhaps most critical for this population to experience SDM. In CF care, patients with more health burden may be in the midst of acute health crises that spur changes to their existing care plans. Shared decision-making is a powerful process for ensuring that these often complex plans continue to fit the resources and constraints each individual and family brings to self-management, which is undertaken primarily outside the health care context (18,19). A clinic coordinator or other team members could be leveraged as an additional resource for patients and families, to better facilitate SDM.
The disparity we observed in
Preparing adolescents for in-depth participation in health care service through the use of developmentally appropriate SDM interventions tailored for pediatric practice, is a promising approach to encouraging SDM in this population (22) and could serve as a foundation for optimal decision-making experiences throughout the transition to adulthood. However, a current lack of SDM interventions tailored for use by adolescents with CF is a barrier (23). To effectively bridge this gap, future research should explore the decision support needs of adolescents and young adults with CF, and then work with patients and CF teams to develop SDM interventions that respond to these needs.
Lastly, for the large majority of pediatric program patients (those <18 years), PFEC items were modified for parent/caregivers as proxy reporters. Considering research indicating that adolescents’ with CF respond more positively to health-related measures than their parents (24), there may be benefits in further exploring the impact of parent/caregiver versus self-completion of patient experience and health-related measures, among adolescents with CF.
Strengths and Limitations
The PFEC is the most comprehensive source of experience data for people with CF and their families in the United States. However, it is voluntary for CF programs to implement the survey and for patients and families to complete it. Although our sample was representative of gender, the lack of available data on other patient characteristics (eg, race, ethnicity, education) means we cannot estimate the extent to which it is representative of the broader population of people with CF in the United States. Further, given the possible range in time post-visit for PFEC completion, recall bias is a possibility. Our overall survey mode comparison suggests that those completing the survey at the time of their visit are more likely to report SDM, although this finding could also be attributed to the specific programs that chose to implement in-clinic completion or potential differences in patient populations. Finally, while we prioritized comparisons by program type to better inform practice, a limitation of this study is combining the 2 respondent groups (patient and proxy) for pediatric programs. Our analyses also did not account for varied care delivery systems (eg, community vs teaching hospitals) that may impact the experience of care.
Conclusion
This large-scale study of patients’ and families’ CF care experiences contributes a novel look at SDM trends in routine chronic care. Disparities in the experience of SDM highlight a need to improve CF decision-making, especially for adolescents transitioning to adult CF care and for people experiencing a mental health burden. The CF Foundation is well positioned to support the enhancement of SDM experiences and to further investigate the variation observed across CF programs.
Supplemental Material
Supplemental Material, sj-pdf-1-jpx-10.1177_23743735211034032 - Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs
Supplemental Material, sj-pdf-1-jpx-10.1177_23743735211034032 for Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs by Karen Homa, Gabrielle Stevens, Rachel Forcino, Peter Scalia, Pamela Mertz and Glyn Elwyn in Journal of Patient Experience
Supplemental Material
Supplemental Material, sj-pdf-2-jpx-10.1177_23743735211034032 - Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs
Supplemental Material, sj-pdf-2-jpx-10.1177_23743735211034032 for Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs by Karen Homa, Gabrielle Stevens, Rachel Forcino, Peter Scalia, Pamela Mertz and Glyn Elwyn in Journal of Patient Experience
Supplemental Material
Supplemental Material, sj-pdf-3-jpx-10.1177_23743735211034032 - Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs
Supplemental Material, sj-pdf-3-jpx-10.1177_23743735211034032 for Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs by Karen Homa, Gabrielle Stevens, Rachel Forcino, Peter Scalia, Pamela Mertz and Glyn Elwyn in Journal of Patient Experience
Supplemental Material
Supplemental Material, sj-pdf-4-jpx-10.1177_23743735211034032 - Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs
Supplemental Material, sj-pdf-4-jpx-10.1177_23743735211034032 for Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs by Karen Homa, Gabrielle Stevens, Rachel Forcino, Peter Scalia, Pamela Mertz and Glyn Elwyn in Journal of Patient Experience
Supplemental Material
Supplemental Material, sj-pdf-5-jpx-10.1177_23743735211034032 - Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs
Supplemental Material, sj-pdf-5-jpx-10.1177_23743735211034032 for Assessing Shared Decision-Making in Cystic Fibrosis Care Using collaboRATE: A Cross-Sectional Study of 159 Programs by Karen Homa, Gabrielle Stevens, Rachel Forcino, Peter Scalia, Pamela Mertz and Glyn Elwyn in Journal of Patient Experience
Footnotes
Authors’ Note
Karen Homa and Gabrielle Stevens are Joint lead authors. This study was approved for secondary research purposes by the Dartmouth Committee for the Protection of Human Subjects (#31976). All procedures in this study were conducted in accordance with Dartmouth College Committee for the Protection of Human Subjects approved protocols. The PFEC itself is considered a quality improvement activity, thus, obtaining informed consent was optional under the HIPAA Privacy Rule for all covered entities (i.e., participating CF programs). We are not aware of how many CF programs chose to do this. The use of PFEC data for this study was considered secondary research for which consent was not required.
Acknowledgments
The authors thank the adults with Cystic Fibrosis (CF) and families with a child or children with CF that completed one or more Patient and Family Experience of Care Surveys (PFEC). The authors also thank the CF programs that provide information for the national PFEC dataset and Fadi Asfour, MD, Medical Director at Primary Children’s Cystic Fibrosis Center, Salt Lake City, Utah, for his input in revising this manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Pamela Mertz and Karen Homa were paid consultants for the CF Foundation during the conduct of this study. Gabrielle Stevens and Glyn Elwyn are supported by an award from the CF Foundation (see 'Funding') to conduct other research projects in addition to this work. Glyn Elwyn’s academic interests are focused on shared decision-making and coproduction. He edited and published the book Shared Decision Making (Oxford University Press) that provides royalties on sales by its publishers. He owns copyright in measures of shared decision-making including
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported (in part) by an award from the CF Foundation [ELWYN18QI0]. The CF Foundation solicited Dartmouth College to conduct an analysis of
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
