Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040. 1
Keywords
Primary Objective
Objective FU1.3: Hereditary Colorectal Cancer and Endometrial Carcinoma. Discuss the relationship of endometrial carcinoma to hereditary nonpolyposis colorectal carcinoma.
Competency 2: Organ System Pathology; Topic: Female Reproductive—Uterus (FU); Learning Goal 1: Uterine Neoplasia.
Secondary Objective
Objective N1.1: Genetic Mechanisms of Neoplasia. Discuss and provide examples of molecular genetic mechanisms that underlie cancers, including germline mutations (including point mutations, deletions, amplifications, and translocations) and epigenetic changes.
Competency 1: Disease Mechanisms and Processes; Topic: Neoplasia (N); Learning Goal 1: Genetic Basis of Neoplasia.
Patient Presentation
A 51-year-old gravida 3, para 3 female presents to her primary care provider with intermittent vaginal bleeding for the past 4 months. The patient is postmenopausal and her last menstrual period was 2 years ago. She is sexually active with her husband and denies postcoital bleeding. She has no associated abdominal pain, dysuria, dyspareunia, bowel habit changes, or weight changes. The patient has no known allergies and is not taking any medications other than a daily multivitamin. She has no chronic medical problems. She drinks alcohol on social occasions and does not use tobacco products.
Her only abnormal pap smear was in her 20s which showed atypical squamous cells of undetermined significance with negative Human papillomavirus (HPV) test. The patient had a tubal ligation after the delivery of her third child at the age of 39. She declined screening colonoscopy last year and opted for the guaiac-based fecal occult blood test (gFOBT), which was negative.
The patient’s family history is notable for several family members with cancer. Her father was diagnosed with colorectal cancer at age 61 and is currently age 74. Her mother is alive and well at age 76. She has 2 siblings, a brother who is healthy at age 55, and a sister who was diagnosed with breast and ovarian cancer at age 37 (currently age 52). The patient’s husband was adopted with no known family history; he is healthy at age 49. The patient and her husband have 3 children. Her older son is healthy at age 31 as is his wife who is age 33. They have 3 children (son age 7, daughter age 4, and daughter age 2). The patient’s daughter was recently diagnosed with breast cancer at age 29. The patient’s younger son was diagnosed with colorectal cancer at age 23 and is currently age 25. Neither of the patient’s 2 younger children are married nor do they have children.
Diagnostic Findings, Part 1
The patient undergoes a complete physical examination. She is a well-kempt female who appears her stated age and is in no acute distress. Her cardiovascular examination reveals regular rate and rhythm without murmurs and her lungs are clear to auscultation without any wheezing. There is no abdominal pain or tenderness upon palpation. Her pelvic examination shows normal external female genitalia. There is mild, age-appropriate atrophy of the vaginal canal without overt abrasions or ulcerations, though a small amount of blood is present. The vaginal canal ends with a multiparous appearing cervical os. Her bimanual examination is unremarkable with no uterine or adnexal tenderness. She has no gross neurologic deficits. Her mood and affect are appropriate.
Questions/Discussion Points, Part 1
What Is the Differential Diagnosis for This Patient?
In a postmenopausal female with vaginal bleeding, the most common cause is atrophic vaginitis. Other possible causes include endometrial polyps, endometrial hyperplasia, endometrial carcinoma, cervical polyps, and cervical cancer. 2
The Family History Includes Several Members With Cancer. Is This a Concern?
Yes. Whenever there are many family members with various types of cancers, one must be highly suspicious that a hereditary component for these cancers exists. It is important to fully investigate because other family members will require screening tests and studies at younger ages than what is recommended for the general population with average risk of cancer.
Construct a Family Pedigree for This Patient. What Is the Likely Mode of Inheritance?
The star in the pedigree (Figure 1) denotes our patient. The likely mode of inheritance is autosomal dominant, though there is a possibility of a nongenetic explanation such as a common environmental exposure.
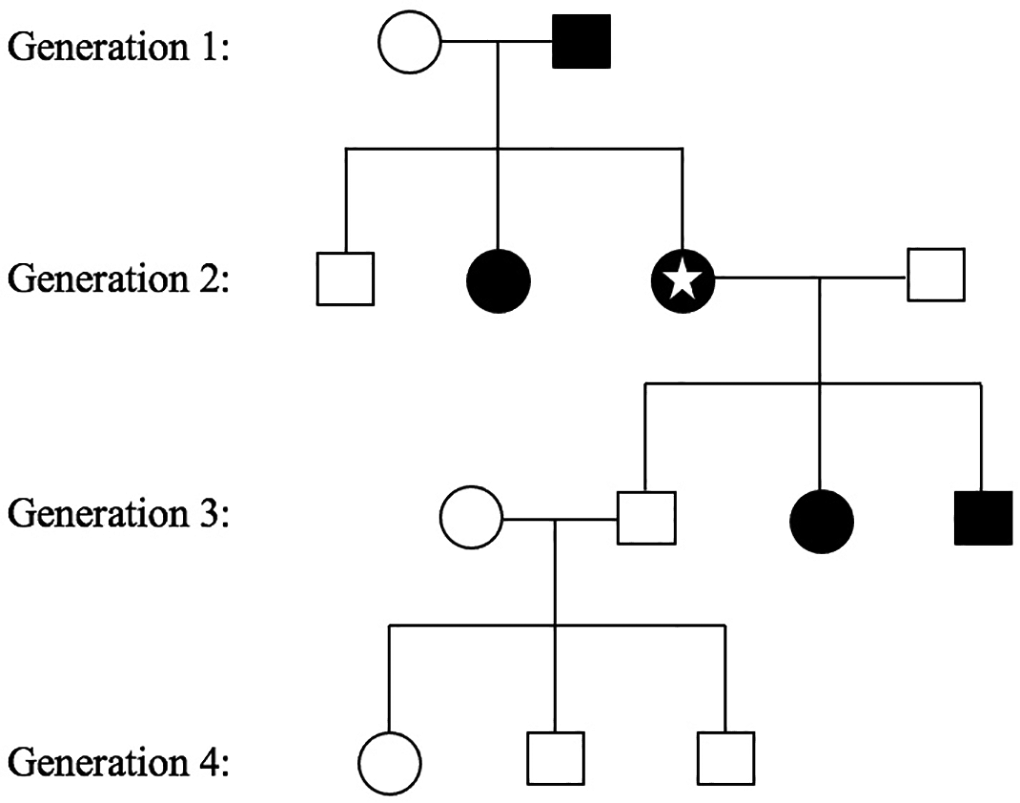
This pedigree represents the autosomal dominant inheritance pattern of Lynch syndrome. The patient described in the narrative is indicated by the star in the pedigree.
How Should One Begin to Work Up This Patient’s Bleeding?
This patient is postmenopausal and has been without a menstrual period for 2 years. Female patients presenting with postmenopausal bleeding should undergo a thorough physical examination as noted above followed by further diagnostic testing. A transvaginal ultrasound is often the first imaging study performed in patients with postmenopausal bleeding as it is readily available and noninvasive. It can significantly aid with quickly ruling in and out differential diagnoses. Baseline laboratory studies should also be obtained to rule out possible infectious causes as well as check for any severe anemia.
Diagnostic Finding, Part 2
This patient’s basic laboratory work including complete blood count, basic metabolic panel, and urinalysis are all within normal limits. Her transvaginal ultrasound reveals an endometrial thickness of 6 mm (normal endometrial thickness is considered less than or equal to 4 mm in postmenopausal women).
Questions/Discussion Points, Part 2
Based on the Ultrasound Findings, What Test Should Be Performed Next?
In a postmenopausal woman with vaginal bleeding, with or without overt risk factors, the lining of the endometrium should be closely examined. This can be done by performing an endometrial biopsy (EMB) in an office setting. A sampling curette is placed through the cervical os and tissue is collected from the endometrial cavity, which can then be sent for pathological examination.
What Results Are Expected From the EMB Given the Patient History and Presentation Above?
This patient is postmenopausal and has an abnormally thickened endometrial lining. One might expect the EMB to show anything from benign proliferative endometrium to endometrial adenocarcinoma. Figure 2 illustrates normal endometrium. Figure 3 illustrates the patient’s endometrium.
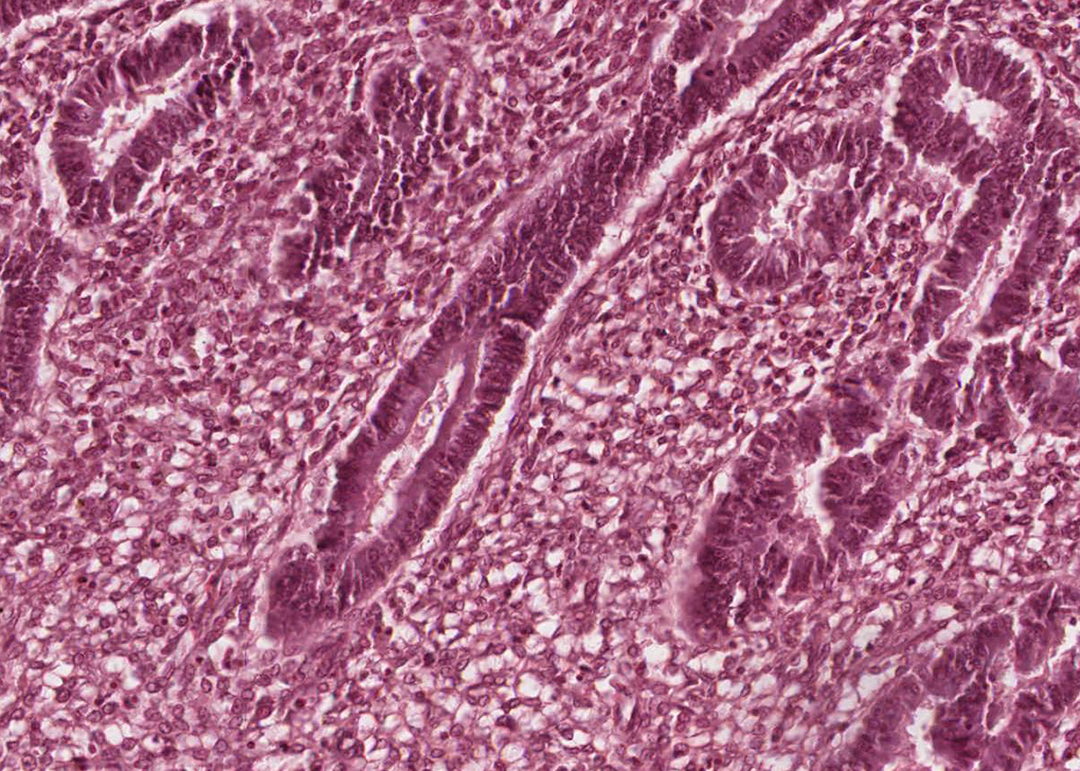
Normal endometrium during proliferative phase. The glands in this proliferative phase endometrium are widely spaced, and the nuclei are regular in size and basally oriented (hematoxylin and eosin [H&E], ×200).
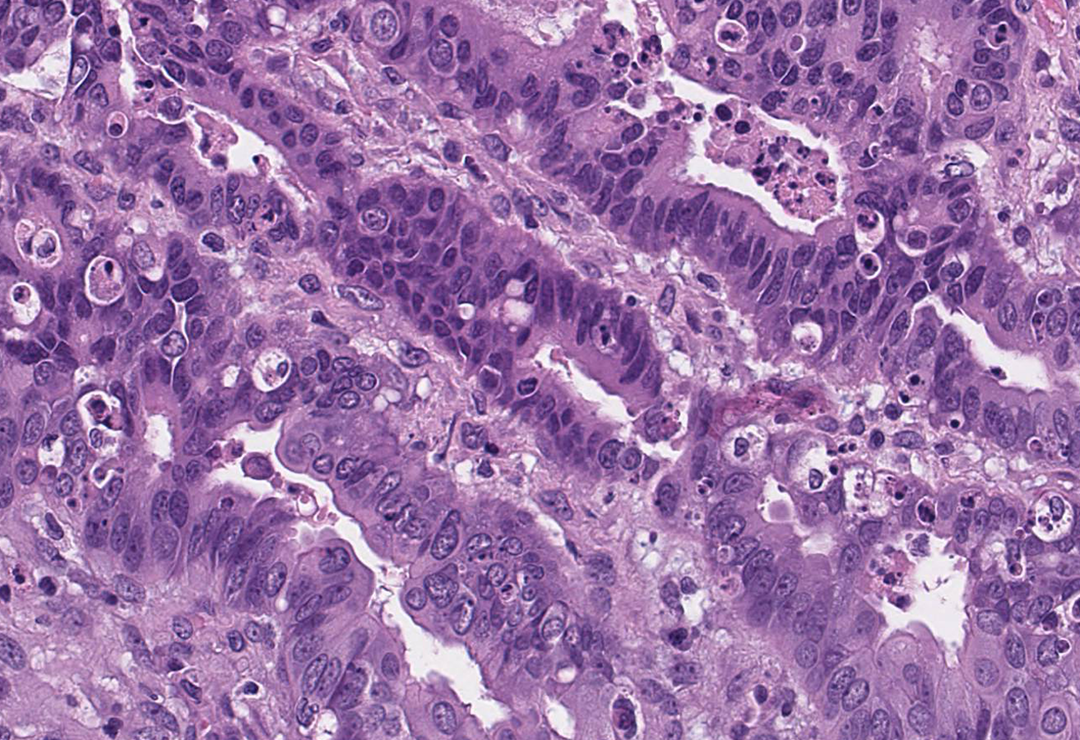
Endometrioid carcinoma (FIGO 1). The glands are packed tightly together within minimal intervening stroma. Although the nuclei are disorganized and variable in size and shape, the tissue retains some resemblance to normal endometrium in having a glandular architecture (hematoxylin and eosin [H&E], ×400).
Diagnostic Finding, Part 3
The patient’s pathology report confirms type I endometrioid adenocarcinoma (Figure 3). Compared to normal postmenopausal proliferative endometrium found in Figure 2, the endometrial glands are closely packed together with minimal intervening stroma. The nuclei vary in size and shape and appear disorganized. The patient was referred to a gynecologic oncologist. In addition, she elects to seek guidance from a genetic counselor.
Questions/Discussion Points, Part 3
Given the Above Patient History, Physical Examination, and Diagnostic Results, What Syndrome Is the Most Likely Diagnosis?
The patient has postmenopausal bleeding, thickened endometrium on ultrasound, and a biopsy confirming endometrial cancer. Because of her significant family history of malignancies, Lynch syndrome, formerly known as hereditary nonpolyposis colorectal cancer, should be highly considered in this patient.
What Genes Are Involved in the Pathogenesis of Lynch Syndrome?
Lynch syndrome is the most common form of hereditary colorectal cancer and is estimated to account for 2% to 4% of all colorectal cancer cases. The inheritance pattern is autosomal dominant. It is contrasted with the next most common form of hereditary colorectal cancer, familial adenomatous polyposis, by the lack of multiple polyps and the detection of germline mutations in various mismatch repair (MMR) genes. 3 The most frequently implicated MMR genes are: MLH1, MSH2, MSH6, and PMS2. In patients with identified germline MMR mutations, the incidences of those genes in colorectal tumors are 46%, 35%, 20%, and 10%, respectively. 4 Deletion in the EPCAM gene is also involved in Lynch tumors as it results in loss of MSH2 expression. 5 Consistent with Knudsen’s classic “two-hit” hypothesis, individuals inherit one mutated allele and then acquire inactivation of the second allele. Epigenetic hypermethylation at promoter regions and somatic mutations can lead to gene inactivation. The accumulation of mutations at regions with short repeating sequences, known as microsatellites, results in genomic instability. 6,7 It is important to remember microsatellite instability (MSI) is not specific to Lynch syndrome. In fact, 15% to 20% of sporadic colorectal cancer can be attributed to MSI pathways. 8
What Malignancies Are Associated With Lynch Syndrome?
Patients with Lynch syndrome have a higher lifetime risk of developing colorectal cancer (up to 80%) and other extracolonic cancers compared to the general population. In addition, these malignancies are discovered at earlier ages. 9 Extracolonic cancers associated with Lynch syndrome involve the uterus, ovaries, stomach, small intestine, hepatobiliary tract, kidney, bladder, skin, and brain. These cancers are often described as synchronous, meaning they manifest at the same time, or metasynchronous, meaning they present and occur temporally spaced in time. 10
What Criteria May Indicate Which Patients Should Undergo Further Molecular Testing for Lynch Syndrome?
The initial 1991 Amsterdam Criteria were originally developed for research purposes; however, they did not take into account extracolonic tumors for diagnosis and missed identifying many individuals. The 1999 Amsterdam II Criteria do take into account extracolonic tumors (Table 1) and are also regarded as being highly specific (61%), yet remain impractical for clinical use due to its low sensitivity (78%). 11,12 The 1997 Bethesda Guidelines were developed with the purpose of identifying tumors for further molecular testing and were later revised in 2004 (Table 2). The Revised Bethesda Guidelines are more sensitive (94%) than the Amsterdam II Criteria but are less specific (25%). 12,13
1999 Amsterdam Criteria II for Molecular Testing for Lynch Syndrome. 10

1997 Bethesda Guidelines for Molecular Testing for Lynch Syndrome. 12

What Molecular Tests Can Be Performed to Help Identify Individuals Who May Need Further Germline Testing?
Tumors are tested for defective DNA MMR gene expression. Immunohistochemistry is used to detect the lack of staining of MMR proteins, particularly MLH1, MSH2, MSH6, and PMS2. 14 These tumors also often exhibit MSI. Microsatellite instability analysis utilizes PCR techniques to amplify specific gene sequences. In recent years, this has been accomplished with next-generation sequencing in which multiple genes are sequenced in parallel to save time. 15
What Are Distinct Pathological Features of Lynch-Associated Colorectal Cancer?
In contrast to sporadic colorectal cancer, Lynch-associated colorectal cancer has a penchant to affect the right side of the colon. In fact, approximately 70% of Lynch colorectal cancer cases are located proximal to the splenic flexure. These tumors have distinct histologic features characteristic of high levels of MSI: poorly differentiated, mucinous, signet ring cells, medullary growth pattern, infiltrating lymphocytes, and Crohn-like lymphocytic reactions. 5,6
Compare and Contrast Type I and Type II Endometrial Carcinoma and Their Respective Histology
The risk of having endometrial cancer in the general population is 2% to 3% while that of individuals with Lynch syndrome is estimated as 40% to 60%. 16 Of note, approximately 50% of female patients with Lynch syndrome present with endometrial cancer as the initial manifestation of this genetic disorder, making it the sentinel cancer found in many Lynch patients and rivaling colorectal cancer. 17,18 Both type I and type II endometrial carcinoma are seen in patients with Lynch syndrome. The slower growing type I endometrial carcinoma is diagnosed at a mean age of 60, is related to excess unopposed estrogen, is associated with PTEN gene mutations and MSI, has endometrioid histology, and is typically lower grade. Figure 2 shows normal endometrium for comparison, while Figure 3 depicts an increased ratio of endometrial glands to stroma with features typical of endometrioid carcinoma. The faster growing and more aggressive type II endometrial carcinoma is not related to hyperestrogenism, is associated with p53 gene mutations, usually has serous or clear cell history, and is higher grade. 19 Figures 4 and 5 below are histological examples of papillary serous and clear cell subtypes, respectively.
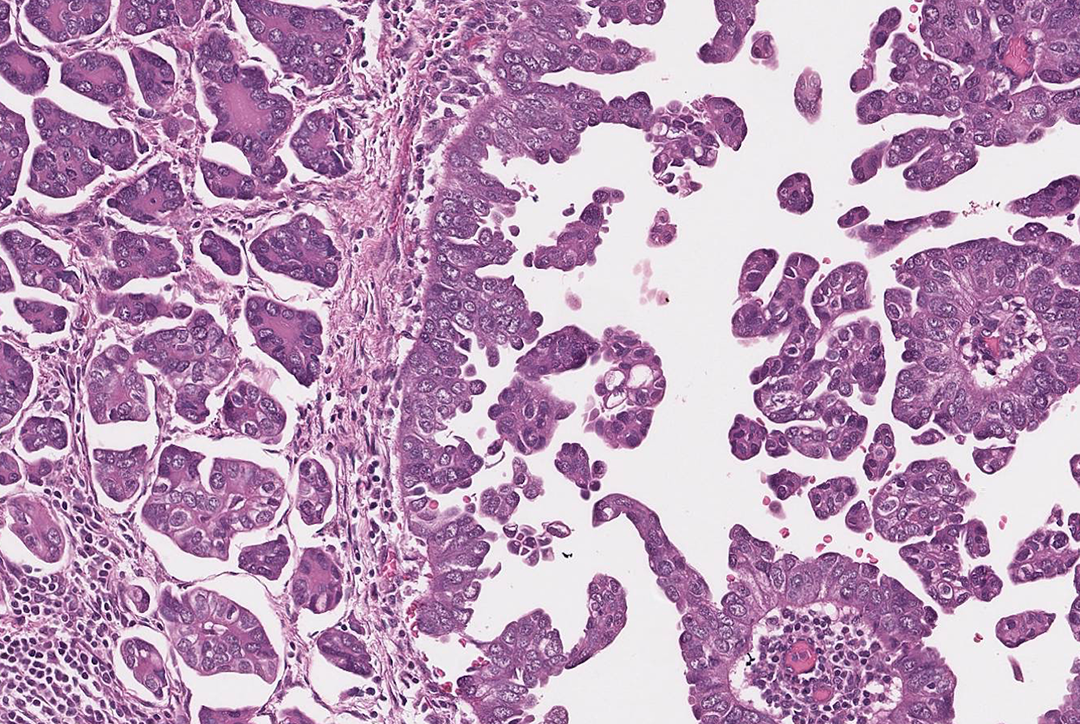
Nonendometrioid, papillary serous subtype. The neoplastic cells in papillary serous carcinoma have a papillary architecture as well as cellular features of neoplasia (pleomorphism, lack of resemblance to cells of origin, loss of polarity, hyperchromasia) and retain no resemblance to normal endometrium (H&E, ×200).
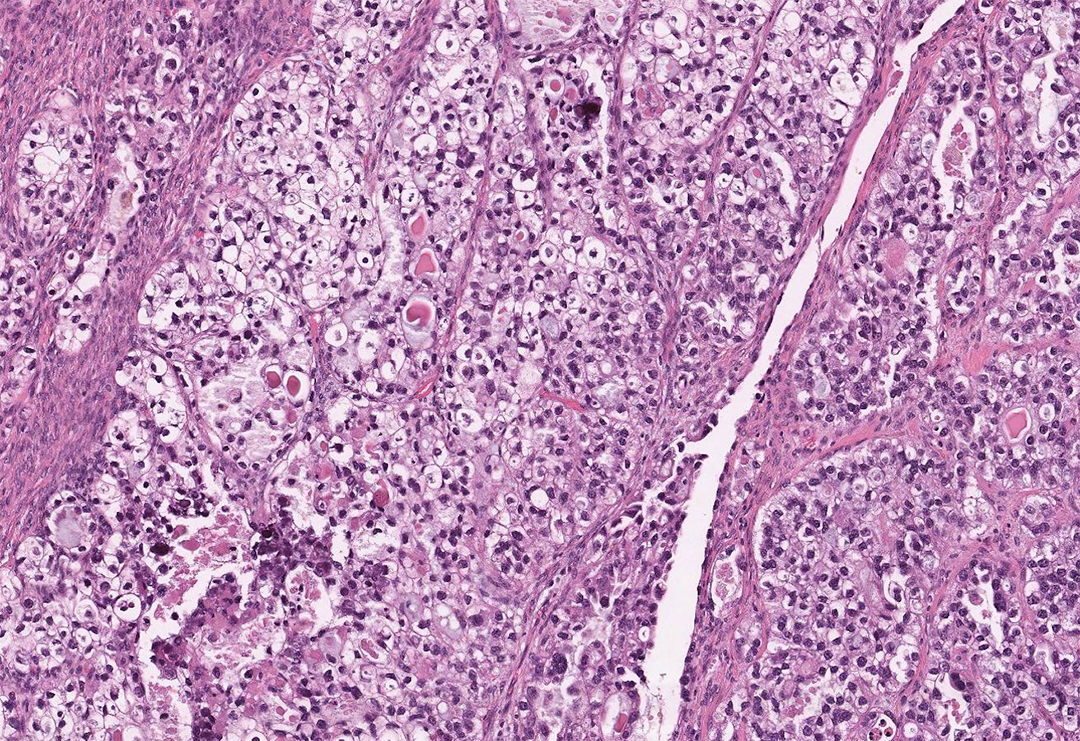
Nonendometrioid, clear cell subtype. The neoplastic cells in clear cell carcinoma have a clear appearance as well as the cellular features of neoplasia (pleomorphism, lack of resemblance to cells of origin, loss of polarity, hyperchromasia; H&E, ×200).
What Are the Similarities and Differences in the Clinical Presentation, Genetics, and Histology Between Endometrial Cancer in Patients With Lynch Syndrome Versus Sporadic Cases?
Like most Lynch-associated cancers, endometrial carcinoma in patients with Lynch syndrome are diagnosed at a younger age (mean age of 50), irrespective of type I or type II classification. 18,20 Although Lynch-associated endometrial cancer can have very diverse and mixed histology, the endometrioid subtype is the most common which correlates with sporadic type I endometrial carcinoma. Interestingly, patients with Lynch-associated endometrial cancer tend to not have factors of excess unopposed estrogen such as higher body mass index, diabetes, and polycystic ovarian syndrome. 17 The 5-year survival rates of Lynch-associated endometrial carcinoma is equal to that of sporadic endometrial carcinoma cases and is estimated to be 88% to 92%. 21
In contrast to the incidences of the 4 main MMR gene mutations (MLH1, MSH2, MSH6, and PMS2) in Lynch colorectal cancer as described above, their incidences in Lynch-associated endometrial cancer are estimated to be 34%, 51%, 49%, and 24%, respectively. In other words, MLH1 and MSH2 are more prevalent in Lynch colorectal cancer, while MSH2 and MSH6 are more commonly seen in Lynch-associated endometrial cancer. 4 In addition, EPCAM deletions near the MSH2 promoter are associated with higher risk of endometrial cancer. 5 Anatomically, Lynch-associated endometrial cancer has been discovered in the lower uterine segment in about 29% of cases. This region is clinical significance because it can be mistaken for cervical cancer. Meanwhile, the majority of sporadic endometrial cancer are in the corpus of the uterus. 22
What Are the Next Steps in the Management of Our Patient and Her Family?
Since this patient is postmenopausal and has completed childbearing, she should be offered a total hysterectomy with bilateral salpingo-oophorectomy. For a patient with the average risk factors for colorectal cancer, yearly gFOBT is one type of screening test that may be used. However, because this patient has a higher than average risk for colorectal cancer, she should undergo a colonoscopy. In fact, a colonoscopy is considered the gold standard in detecting colonic lesions and obtaining biopsies. 23 Some altered gFOBT results have been attributed to certain fruits and vegetables, red meats, nonsteroidal anti-inflammatory medications, and excessive vitamin C supplements. 24 -26
Her family members can talk with their primary care providers regarding their individual cases and seek genetic counseling. The American College of Obstetricians and Gynecologists recommends the following screening for women with Lynch syndrome: (1) colonoscopy every 1 to 2 years, starting at age 20 to 25, or starting 2 to 5 years before the earliest cancer was diagnosed in the family; and (2) EMB every 1 to 2 years, starting at age 30 to 35. Women can also monitor themselves for abnormal uterine bleeding by keeping a menstrual calendar. 27 Oral contraceptives that contain progestins and Depo-Provera (medroxyprogesterone acetate) are chemopreventive agents that can help to reduce endometrial cancer risks in Lynch patients. 28 Prophylactic hysterectomy with salpingo-oophorectomy is the most effective way to reduce endometrial and ovarian cancer risks and essentially cuts the risks down to zero. 29 This procedure results in permanent sterility and early menopause and therefore, is offered to patients who are nearing the end of their childbearing years. American College of Obstetricians and Gynecologists recommends the risk-reducing procedure be performed between ages 40 to 45 and can be done concurrently in patients undergoing surgery for colorectal cancer. 27
Teaching Points
Molecular genetic mechanisms that underlie cancers include germline mutations of specific genes, acquired mutations (random or induced by outside forces, toxins, environment, etc), and epigenetic changes.
Hereditary nonpolyposis colorectal cancer or Lynch syndrome is an autosomal dominant familial disorder due to germline mutations in various MMR enzymes, particularly MLH1, MSH2, MSH6, and PMS2.
Tumor suppressor genes such as those mutated in Lynch syndrome likely require inactivation of both alleles to cause cancer, consistent with Knudson’s “two-hit” hypothesis. When one inactivated allele is inherited, the probability of cancer is increased.
There is a strong history of familial cancers in Lynch syndrome across generations and diagnosis occurs at approximately 10 years younger than the average for sporadic cases.
The Amsterdam II Criteria are less sensitive but more specific than the Revised Bethesda Guidelines in identifying individuals with Lynch syndrome.
Lynch syndrome is detected via direct germline testing, immunohistochemistry, and MSI testing of tumors.
Endometrial cancer is most common extracolonic manifestation. Lynch-associated and sporadic endometrial cancer share similar endometrioid histological features and prognosis.
Women with Lynch syndrome will require more frequent intervals for cancer screening including colonoscopy every 1 to 2 starting at age 20 to 25 and EMB every 1 to 2 years starting at age 30 to 35.
Prophylactic hysterectomy with salpingo-oophorectomy is a risk-reducing surgery offered to women who has completed childbearing.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Walter L. Kemp receives royalties from McGraw-Hill for pathology review book and Thieme for Q&A book.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
