Abstract
The COVID-19 pandemic, caused by severe acute respiratory syndrome coronavirus 2, created an unprecedented need for comprehensive laboratory testing of populations, in order to meet the needs of medical practice and to guide the management and functioning of our society. With the greater New York metropolitan area as an epicenter of this pandemic beginning in March 2020, a consortium of laboratory leaders from the assembled New York academic medical institutions was formed to help identify and solve the challenges of deploying testing. This report brings forward the experience of this consortium, based on the real-world challenges which we encountered in testing patients and in supporting the recovery effort to reestablish the health care workplace. In coordination with the Greater New York Hospital Association and with the public health laboratory of New York State, this consortium communicated with state leadership to help inform public decision-making addressing the crisis. Through the length of the pandemic, the consortium has been a critical mechanism for sharing experience and best practices in dealing with issues including the following: instrument platforms, sample sources, test performance, pre- and post-analytical issues, supply chain, institutional testing capacity, pooled testing, biospecimen science, and research. The consortium also has been a mechanism for staying abreast of state and municipal policies and initiatives, and their impact on institutional and laboratory operations. The experience of this consortium may be of value to current and future laboratory professionals and policy-makers alike, in dealing with major events that impact regional laboratory services.
Introduction
In December 2019, the outbreak of novel coronavirus (severe acute respiratory syndrome coronavirus 2 [SARS-CoV-2]) began in Wuhan, China, and spread rapidly throughout the world. 1 New York State became a major epicenter, with peak case incidence and case deaths occurring early in April 2020. 2 In May 2020, the effort to manage infection rates in the New York region was integrated with efforts to establish a “new normal” for health care delivery and restoration of societal functions. 3 This “new normal” is heavily dependent on the availability and utilization of laboratory testing. 4
On April 14, 2020, senior leadership of the clinical laboratories of 11 major academic medical institutions serving New York State formed a consortium in order to share knowledge about implementation of SARS-CoV-2 diagnostic and serologic testing. The impetus for formation of the consortium was need to bring our respective expertise forward in support of the New York State response to the COVID-19 crisis. We also had a shared need to break out of our respective institutional silos so as to learn from one another on a real-time basis, and perhaps more importantly, have a clear sense of the intentions of New York State as it dealt with the crisis. We now report on the experience of this consortium, as regard to operations, communication, and information content that helped empower laboratory leadership of the state academic medical institutions over the length of this crisis.
Methods
Establishing the Consortium
The Association of Pathology Chairs (APC) maintains a listserv for its membership. On March 12, 2020, the first posting to this listserv regarding the emerging COVID-19 pandemic and the clinical laboratory response was from the University of Washington, followed the next day by a report from Northwell Health. Over the next 3 months, the APC listserv remained highly active at the national level, as the leadership of departments of pathology and laboratory medicine at academic health systems learned from one another and dealt with the emerging crisis. Specific to the State of New York, laboratory leadership from Northwell Health, Mount Sinai Hospital, State University of New York Downstate Health Sciences University, and other New York academic institutions provided our March and early April updates through the APC listserv to the national academic pathology community. This occurred simultaneous with the New York academic clinical laboratories working intensively with the State of New York Department of Health through the regulatory testing approvals process, so as to deal with the regional pandemic, for which both New York State case incidence and hospitalizations peaked in the first week of April.
On Friday April 10, 2020, an invitation was extended from the state Commissioner of Health, Howard Zucker, MD, to laboratory leadership of the state’s academic medical institutions to discuss COVID-19 testing options with him in teleconference on April 13. In that discussion, he asked the academic institutions to bring COVID-19 testing recommendations back to him by Friday April 17. The Commissioner instructed the Greater New York Hospital Association (GNYHA) to be the convening authority for our further discussions, and the primary mechanism for communicating recommendations back to the Commissioner and the Governor’s Council. This request served as the catalyst for formation of the consortium.
The Chief Executive Officers of our respective institutions already were meeting virtually with the Commissioner of Health on a regular basis. In the founding discussion of this consortium on April 14, we acknowledged that each institutional laboratory was accountable to our respective institutional leadership. It would be the responsibility of each institutional delegation to report as appropriate to our respective institutional leadership on the activities of the consortium, and especially to bring information obtained through the consortium back to our respective institutions. In turn, the formalism of the consortium, with agendas posted prior to each meeting, and minutes recorded thereof, helped ensure that the activities of the consortium were transparent. This formalism: (a) provided a safe harbor for interinstitutional discussions about our respective capabilities and weaknesses; (b) facilitated consortium communication through the GNYHA to state leadership; and (c) helped ensure that the consortium remained focused on the mission of addressing the COVID-19 crisis, without risking transgression of regulations regarding trade activities.
Consortium Membership
The initial consortium in April 2020 consisted of 11 institutions by invitation, each with an institutional delegation selected by the academic department of pathology and/or laboratory medicine chair (Table 1). In August and September 2020, 2 additional institutions were added, including the Larner School of Medicine, University of Vermont, on the basis of laboratory services provided in the upper Adirondack region of New York State.
Consortium Membership.

*Founding Member.
Consortium Operations
Northwell Health served as the Moderator and Secretariat for the consortium, working with GNYHA for preparation of agenda items for each meeting, and providing follow-up as required. The agenda for consortium meetings was set on the basis of state and municipal policies that impacted New York State laboratories; and operational issues, strategic initiatives, and advocacy issues of concern to consortium institutions. Agendas and minutes were maintained by the Secretariat. Following an initial 4-meeting sequence of April 14-16, 2020, and delivery of our recommendations to the Commissioner of Health, the consortium members unanimously agreed that there was need for continuation of the consortium. We met semiweekly April 22 to May 15; weekly May 22 to June 9; and have met monthly ever since. Teleconferences were converted to virtual video meetings on June 9, 2020.
Consortium Deliverables
The first deliverable of the consortium, at request of the Commissioner of Health on April 13, 2020, was to advise New York State on testing platforms for COVID-19; these recommendations were delivered to the Commissioner on Friday April 17, 2020.
The second deliverable was informing the state on the actual deployment of instrumentation platforms and assays being used for laboratory diagnosis of SARS-CoV-2 infection, and consortium institutional capacities for daily testing. These data were provided to the state from April 17, 2020, to May 25, 2020. By late May, this level of detail was not required for state communication purposes, and reporting was stood down.
For the duration of the consortium’s existence, the third key deliverable has been communication between consortium institutions, and provision to consortium institutions of timely information and updates from the State of New York and City of New York as communicated by GNYHA.
The fourth deliverable was to examine emerging data and clearly articulate the performance of testing assays for this new infectious agent. The shared expertise of consortium membership helped empower consortium members to communicate effectively within our own institutions, and with the public.
Fifth, from the outset, observational and investigative research was deemed by consortium membership to be a major priority for the consortium response to the pandemic. PubMed literature searches and consortium self-reporting as of December 19, 2020, were used to identify publications with authorship that included named faculty members of the departments of pathology and/or laboratory medicine of the consortium institutions.
Results
A timeline for consortium activities is given in Figure 1; the deliverables are as follows.
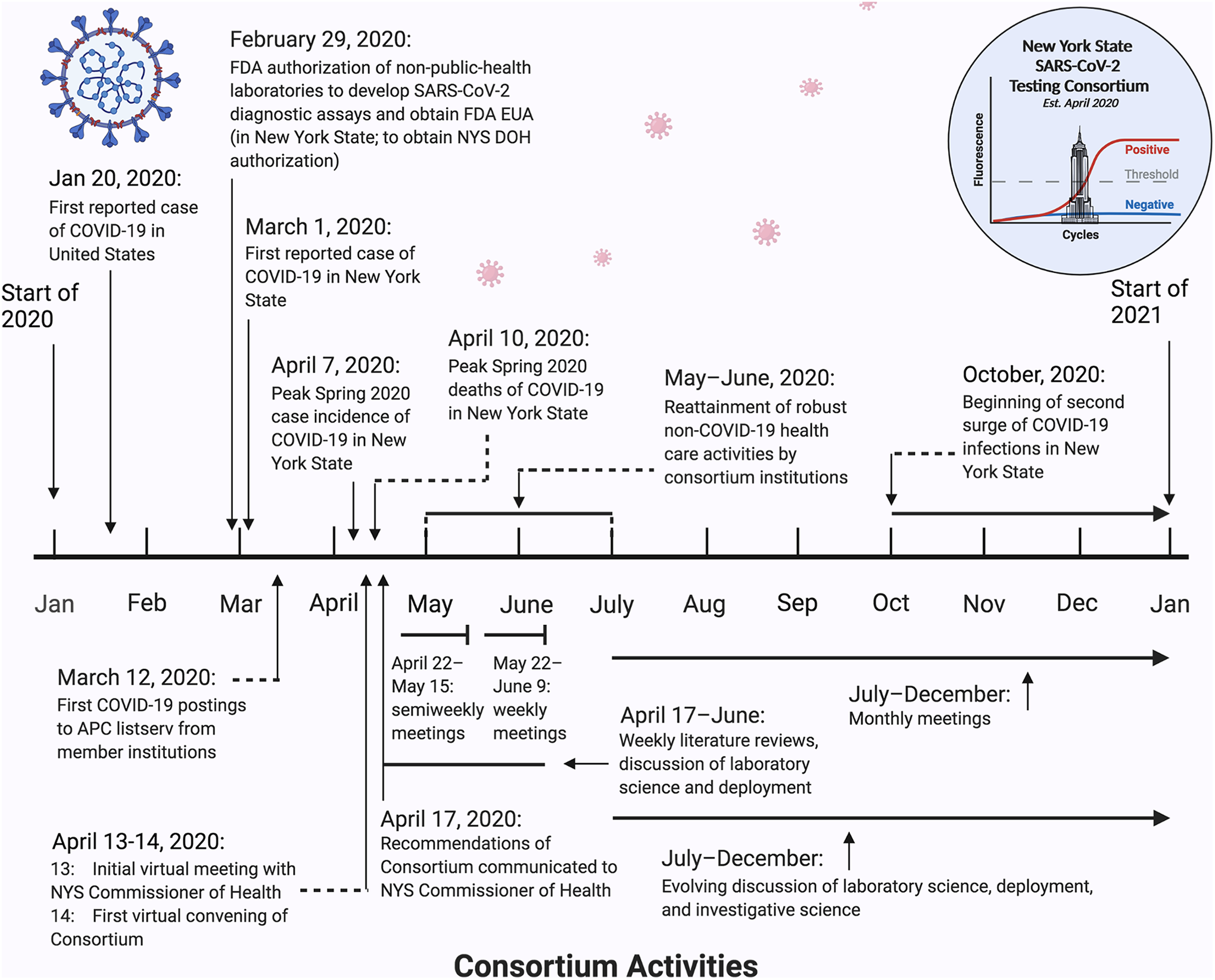
Timeline for Consortium Activities. The dates above the timeline reflect regional events. The dates below the timeline reflect the specific activities of the consortium. Note: APC, Association of Pathology Chairs; NYS, New York State.
Deliverable 1: April 17, 2020, Report to the New York State Commissioner of Health
The consortium met telephonically 4 times in 3 days (April 14-16, 2020) to develop recommendations for the Commissioner. The consortium recommendations are given in Table 2. The reasoning behind these recommendations was as follows.
Consortium Recommendations to the New York State Commissioner of Health, April 17, 2020.

Consortium deployment of test platforms
The key request put before the consortium by the Commissioner was to recommend the “best tests” for molecular and serological diagnosis of SARS-CoV-2 infection. In response, the consortium noted that an extensive variety of commercial platforms for COVID-19 molecular and serologic testing was entering the market, and that it was important for consortium members to be able to use this plurality in our choice of testing platforms. This applied both to the use of commercial platforms and the use of consortium laboratory developed tests (LDTs). In the latter instance, commonalities among the consortium serologic LDTs included the viral antigenic targets being tested and the technical testing approaches. Besides the need for flexibility so as to deal with uncertainties in reagents, consumables, and instrumentation supply, through the plurality of consortium efforts the state was gaining both additional testing capacity and important knowledge about SARS-CoV-2 testing. The consortium thus constituted a valuable resource for the State, in support of the consortium premise that the statewide effort should be driven by Science as well as by Need.
As of April 17, 2020, consortium members were deploying a spectrum of commercial testing platforms and LDTs for SARS-CoV-2 molecular diagnostic testing, using polymerase chain reaction (PCR; Table 3). Collectively, the aggregate capacity of consortium laboratories was anticipated to increase from >5000 tests/day (week of April 20-26) to >20 000 tests/day (week of April 25-May 3), with further increases over following weeks. These estimates did not include additional molecular diagnostic testing capacity from commercial platforms that were currently being validated and would be coming on line; as well as anticipated enhancements and scalability achieved by consortium efforts to further develop our LDT platforms.
SARS-CoV-2 Testing Platforms in Use by Consortium Member Institutions at height of Spring 2020 COVID-19 Pandemic (March-June, 2020).
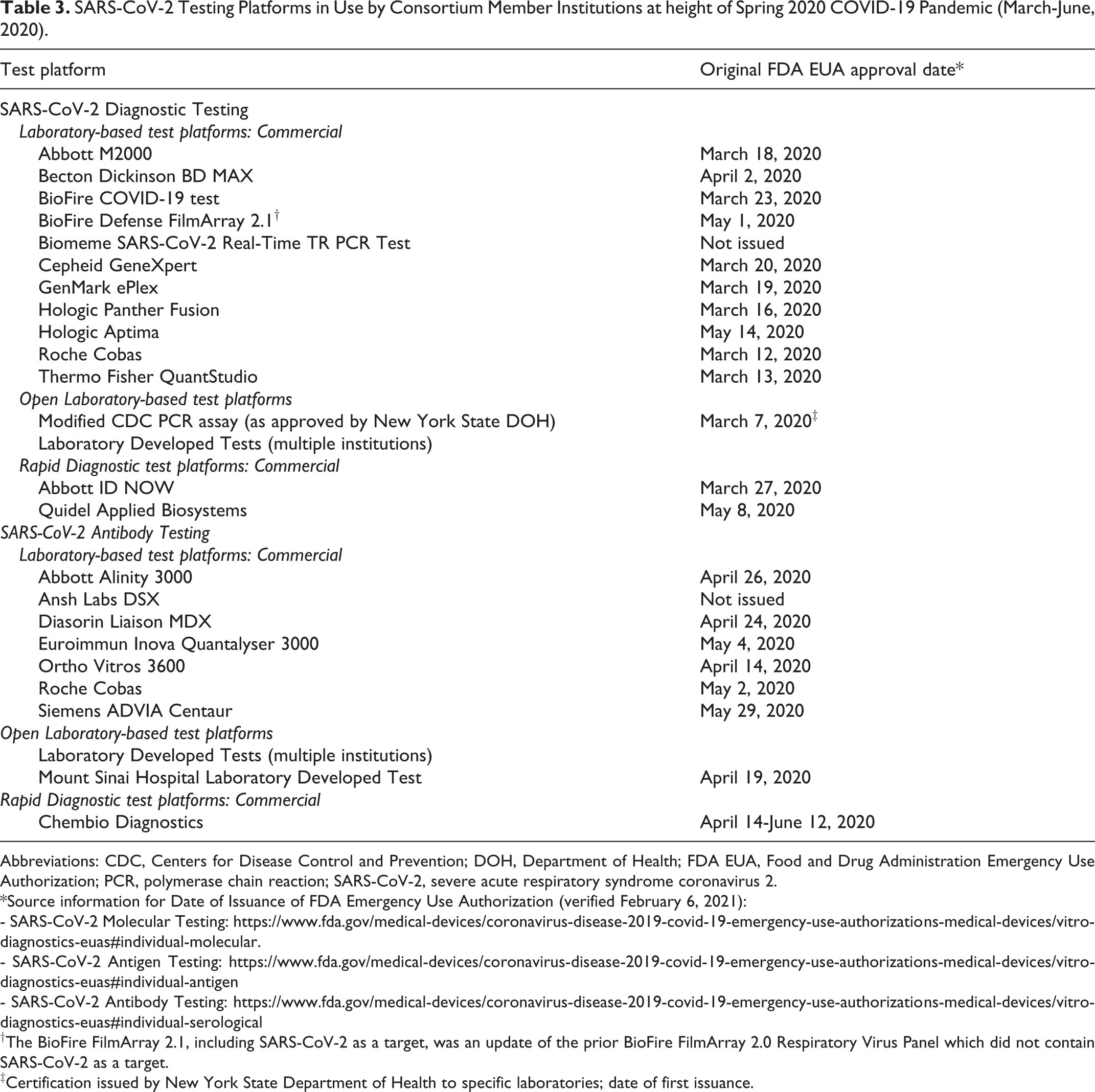
Abbreviations: CDC, Centers for Disease Control and Prevention; DOH, Department of Health; FDA EUA, Food and Drug Administration Emergency Use Authorization; PCR, polymerase chain reaction; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
*Source information for Date of Issuance of FDA Emergency Use Authorization (verified February 6, 2021):
- SARS-CoV-2 Molecular Testing: https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/vitro-diagnostics-euas#individual-molecular.
- SARS-CoV-2 Antigen Testing: https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/vitro-diagnostics-euas#individual-antigen
- SARS-CoV-2 Antibody Testing: https://www.fda.gov/medical-devices/coronavirus-disease-2019-covid-19-emergency-use-authorizations-medical-devices/vitro-diagnostics-euas#individual-serological
†The BioFire FilmArray 2.1, including SARS-CoV-2 as a target, was an update of the prior BioFire FilmArray 2.0 Respiratory Virus Panel which did not contain SARS-CoV-2 as a target.
‡Certification issued by New York State Department of Health to specific laboratories; date of first issuance.
Regarding SARS-CoV-2 serologic testing, the top priority for consortium institutions that had activated such testing (with both commercial platforms and LDTs, Table 3) was for testing of our own institutions’ health care workers (HCWs). The initial HCW’s to be tested were “front line” HCW’s (including those working in emergency departments, inpatient wards, and intensive care units); and/or HCW’s who had PCR-proven COVID-19 infection and had recovered. The latter group provided valuable opportunity to gain initial insights into the “host immune response” to documented SARS-CoV-2 infection. Severe acute respiratory syndrome coronavirus 2 serologic testing was also being performed on selected patient populations of immediate high priority.
Initiation of state serological testing
The second request before the consortium was to provide recommendations for initiation of SARS-CoV-2 serological testing in New York State. The consortium observed the following. First, a statewide perspective on supply chain distribution would be of benefit to consortium laboratories, to ensure that the full testing capacity of consortium laboratories could be leveraged in fulfillment of state needs for SARS-CoV-2 testing. Second, the specific logistics of bringing SARS-CoV-2 serological (and molecular) testing to the workplace and to communities must have state and local community support and management. Third, state support for testing performed by consortium members in support of public health needs would be essential, owing to the diverse communities being served including individuals who did not have health plan coverage for such testing. Fourth, for the state to act knowledgeably on the basis of SARS-CoV-2 serological testing, robust epidemiologic programming, supported by a statewide data system, must be in place. The data would be needed to track success, support policy decisions, and for information dissemination. Fifth, the New York State Department of Health (NYS DOH) Wadsworth Center (the state public health laboratory) constituted an invaluable resource for supporting statewide SARS-CoV-2 serologic testing validation, creation of testing standards, and generation of knowledge about the value of such testing for our state populations. Sixth, the state could act as convening authority for coordination of consortium efforts with those of regional commercial laboratories. Seventh, as the NYS DOH was already engaged in development and dissemination of educational materials for the public, this ongoing effort was considered to be essential and could be informed in an ongoing basis by knowledge gained from consortium involvement in the state effort. Lastly, both through New York being the epicenter of the COVID-19 pandemic in the United States, and by having an extraordinary concentration of outstanding academic medical centers who were members of this consortium, New York State would be able to contribute substantively to world knowledge about COVID-19 disease. The State was encouraged to support such a critical programmatic effort.
Outcomes of consortium recommendations
The consortium recommendations were delivered to the Commissioner at 10:00
Deliverable 2: Consortium Instrumentation and Daily Testing Capacities
For both SARS-COV-2 molecular diagnostic testing and serologic testing, the consortium documented that member institutions were standing up a plurality of commercial instrumentation, and/or implementing both molecular and serologic LDTs. Among many frustrations were the inability to obtain instruments, even prior to the sustained severe constraints on the supply chain for consumables, kits, and reagents. For 2 consortium institutions, their LDT provided the only reliable mechanism for performing institutional SARS-CoV-2 molecular testing; for one consortium institution SARS-CoV-2 molecular testing had to be sent to a commercial laboratory for many weeks during the first 3 months of the pandemic (March-May, 2020). The aggregate consortium daily testing capacity for April 13, 2020, to May 20, 2020, is shown in Figure 2. These data helped inform the state on the ramp-up in both molecular and serologic testing in the academic laboratory sector, as arrangements were being made throughout the state for COVID-19 testing.
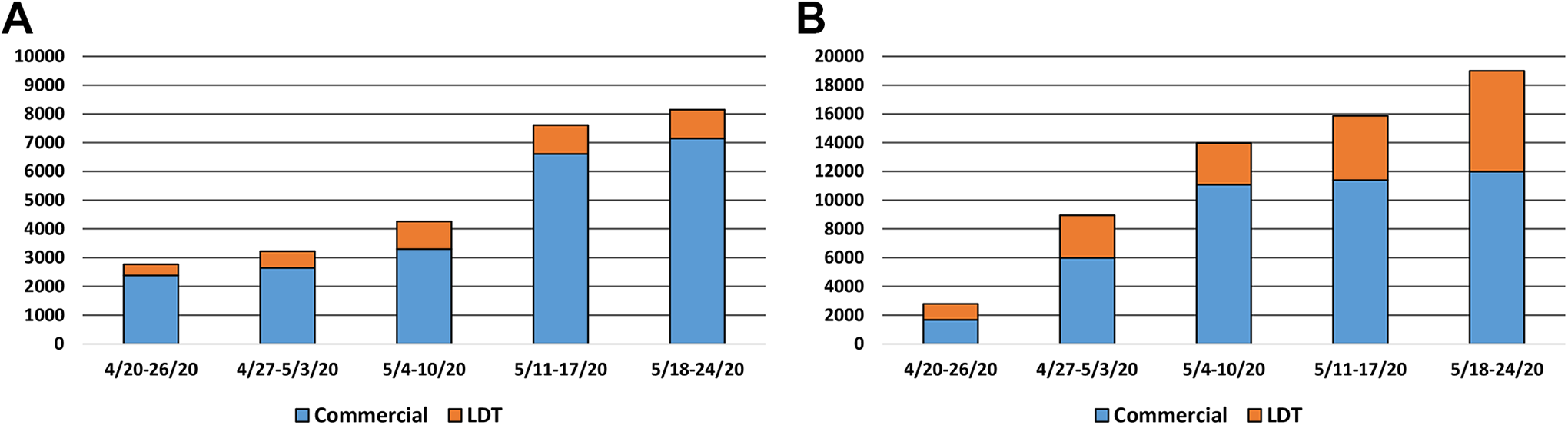
Daily SARS-CoV-2 testing by consortium institutions, April 20, 2020, to May 24, 2020. A, SARS-CoV-2 molecular diagnostic testing performed by polymerase chain reaction (PCR). B, SARS-CoV-2 serologic testing. LDT indicates laboratory developed test; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Deliverable 3: Consortium Communication and Content
In the initial weeks of consortium operation, the Secretariat performed weekly searches of the PubMed database for articles on SARS-CoV-2 laboratory diagnostics; search terms and strategy were as used by Cheng et al. 7 This function was stood down in mid-June 2020, owing to the massive volume of COVID-19-related publications emerging by that time. The Secretariat also monitored the following: websites of the in vitro diagnostics manufacturers for updates and advisories on their SARS-CoV-2 testing platforms; industry announcements of new testing platforms and their Food and Drug Administration (FDA) Emergency Use Authorization (EUA) status; the public websites of major commercial laboratories as regard to test sample requirements and reported testing turnaround times; COVID-19-related advisories to licensed clinical laboratories as issued by the US FDA, Centers for Disease Control and Prevention (CDC), NYS DOH, and Executive Orders issued by the Governor of New York State. These materials, which were made available to consortium membership prior to scheduled meetings, helped inform the semiweekly discussions of the consortium.
In parallel, consortium members populated a Secretariat-maintained roster of institutional testing capabilities, including testing platforms used, challenges being encountered, and challenges in enhancing institutional capacity through use of both commercial testing platforms and institutional LDTs; this roster was updated weekly through May 24, 2020.
The topics formally discussed in consortium meetings are given in Table 4. As is evident, there was enormous breadth of discussion, tracking the cascading issues faced by consortium institutions in dealing with the COVID-19 pandemic. Also evident is the interplay between New York State and New York City policies and decision-making and their impact on laboratory operations. In turn, both through verbal communication and with the written record of these meetings in the consortium minutes, the GNYHA, and, beginning in September 2020 the Hospital Association of New York State (HANYS) were well informed for raising consortium issues with state leadership.
Consortium Discussion Topics.

Abbreviations: CDC, Centers for Disease Control and Prevention; EMR, electronic health record; FDA EUA, Food and Drug Administration Emergency Use Authorization; IRB, institutional review board; LDT, laboratory developed test; LIS, laboratory information system; MIS-C, multisystem inflammatory syndrome of children; PCR, polymerase chain reaction; RVP, respiratory virus panel; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
*Agenda topics over-and-above selective updates on prior agenda items (noting that Supply Chain was an agenda item in every meeting).
Deliverable 4: Examination of Test Performance
The consortium used the weeks of April and May 2020 to critically examine all aspects of test performance for SARS-CoV-2 laboratory science, both in the diagnostic and serologic realms. Information was drawn both from publicly available information as above and from real-world experience being gained by consortium members. Although new test assays and emerging technologies have continued to appear in the months since, the conceptual foundations articulated by the consortium in April and May have stood the test of time. Importantly, the shared expertise brought forward by consortium members empowered members to be effective communicators within our own institutions (for which there was great need), and to the public. In the latter instance, consortium members were heavily sought after subject matter experts for public dialogue and media communications, as through newspaper interviews, television news broadcast, podcasts, and social media postings. The components of this critical examination are as follows.
Issues affecting deployment of SARS-CoV-2 testing
The laboratory science of SARS-CoV-2 molecular and serologic testing is reviewed elsewhere. 7 On the basis of consortium real-world experience, issues affecting successful deployment of diagnostic and serologic testing strategies are given in Table 5. Specifically, these issues pertained to Pre-analytical (pretesting), Analytical (testing), and Post-analytical (post-testing and reporting) deployment of SARS-CoV-2 testing. That there may be significant clinical consequences if these factors are not taken into account is reviewed elsewhere. 8 The dominant “real-world” issues raised in Table 5 as experienced by consortium members were:
Preanalytical, Analytical, and Post-Analytical Variables Affecting Deployment of SARS-CoV-2 Testing.

Abbreviations: rRT-PCR, real time reverse transcriptase-polymerase chain reaction; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
*For detection of viral nucleic acid by rRT-PCR.
‡Harmonization: Patient identification and tube labeling, ensuring that the correct testing kit (appropriate swab device; Viral Transport Media with appropriate lysate) is used at the right collection site, and delivered to the right testing platform.
SARS-CoV-2 diagnostic test performance: Sample procurement was a major starting point for consortium members in educating clinical colleagues about test performance and expectations for positive predictive value (PPV) and negative predictive value (NPV). Sample source, including educating a broad health care workforce in proper procurement of nasopharyngeal swabs; and being mindful of the timing of viral infection in setting expectations for test sensitivity, were important teaching points. In turn, extensive education was required for the fundamental laboratory science of test Specificity and Sensitivity, as regard to the likelihood for false negatives and false positives. These discussions then informed consortium institutional development of algorithms for repeat testing, in the triage and cohorting of patients suspected of having COVID-19, especially in the early stages of the pandemic when test availability was severely constrained.
SARS-CoV-2 diagnostic test delivery: Extensive coordination of consortium clinical laboratories with our respective health systems was required for sample logistics and sample prioritization. In the latter instance, even as the height of the Spring 2020 crisis subsided, testing volumes increased relentlessly owing to the reopening of our health systems for regular health care. Hence, these 2 preanalytical issues have continued to be major variables for the overall ability of consortium laboratories to meet the needs of our parent health systems. Given the continuing constraints on the testing supply chain, whether a SARS-CoV-2 diagnostic test can be performed in-house or is sent out to a commercial reference laboratory has been a key determinant of test turnaround time. This is true simply owing to the logistics of sample transport before consideration is given to the in-laboratory turnaround times of the respective laboratories.
SARS-CoV-2 serologic testing: Given the intensity of the COVID-19 pandemic in the greater New York metropolitan area in March to May 2020, there was great need to obtain insight into the exposure of the regional health care workforce to SARS-CoV-2 during their “front-line” care of COVID-19 patients. This was at a time when the biology of the host immune response to SARS-CoV-2 was only first being mapped out. Publications from 2 of the consortium institutions, based on serologic testing deployed as soon as FDA EUA was issued, provided assurance that the stringent protocols for protection of HCWs from COVID-19 infection were effective. 9,10 Serologic testing also was a critical tool for qualification of convalescent plasma for use in COVID-19 patients. 11,12 The real-world experience of these initiatives helped inform all consortium members on the variables affecting SARS-CoV-2 serologic testing performance, as given in Table 5.
Test utility
At every phase of the COVID-19 pandemic, test utility has been a critical issue, including the potential for “false negatives” and “false positives.” 13 Table 6 was developed as a reference table for NPV and PPV, using the algebraic formulation given in Figure 3. This table was constructed on the basis of published reports for the test performance of laboratory-based diagnostic (molecular) assays and rapid point-of-care diagnostic assays (both PCR-based and then antigen-based 14 -31 ); and for test performance of laboratory-based and rapid point-of-care serologic (antibody) assays. 32 -42 This chart or versions thereof found its way into numerous intrainstitutional presentations given by consortium members, to give example of how sensitivity, specificity, and case prevalence all impact NPV and PPV.
Impact of Specificity, Sensitivity, and Prevalence on Negative and Positive Predictive Value.*
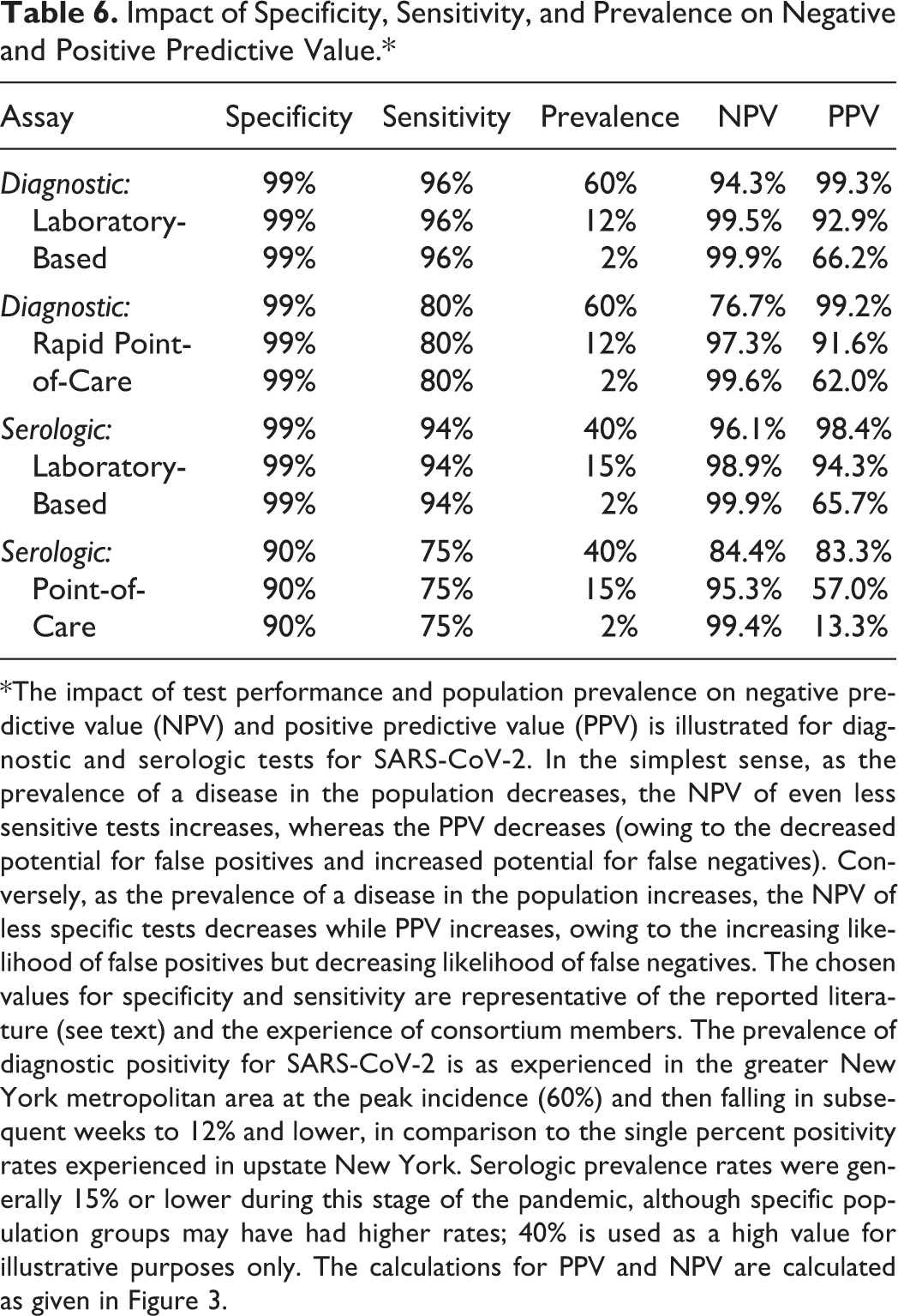
*The impact of test performance and population prevalence on negative predictive value (NPV) and positive predictive value (PPV) is illustrated for diagnostic and serologic tests for SARS-CoV-2. In the simplest sense, as the prevalence of a disease in the population decreases, the NPV of even less sensitive tests increases, whereas the PPV decreases (owing to the decreased potential for false positives and increased potential for false negatives). Conversely, as the prevalence of a disease in the population increases, the NPV of less specific tests decreases while PPV increases, owing to the increasing likelihood of false positives but decreasing likelihood of false negatives. The chosen values for specificity and sensitivity are representative of the reported literature (see text) and the experience of consortium members. The prevalence of diagnostic positivity for SARS-CoV-2 is as experienced in the greater New York metropolitan area at the peak incidence (60%) and then falling in subsequent weeks to 12% and lower, in comparison to the single percent positivity rates experienced in upstate New York. Serologic prevalence rates were generally 15% or lower during this stage of the pandemic, although specific population groups may have had higher rates; 40% is used as a high value for illustrative purposes only. The calculations for PPV and NPV are calculated as given in Figure 3.

The algebra of test performance and predictive value. FN indicates false negative; FP, false positive; NPV, negative predictive value; PPV, positive predictive value; TN, true negative; TP, true positive.
Table 7 was then developed on the basis of consortium institution real-world experience, to inform the impact of false negatives and false positives, and in turn NPV and PPV, on test result reporting. Specifically, during the peak of the pandemic, false negatives represented a significant clinical issue even with high-sensitivity laboratory-based SARS-CoV-2 molecular testing. 43 False positives then became more of an issue through Summer and into the Fall 2020, as the volumes of SARS-CoV-2 laboratory-based PCR tests continued to rise and less sensitive rapid testing (both PCR and antigen) become more and more common, applied to a regional population whose test % positivity rates fell to below 2% (through September 2020) and then began to increase again in the Fall 2020. Key impact points for SARS-CoV-2 diagnostic testing included:
Considerations for SARS-CoV-2 Test Utility.

Abbreviations: PCR, polymerase chain reaction; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
*PCR test sensitivity is a function of the number of viral RNA copies required to generate a positive result, reflected in the test Limits of Detection (LOD).
†Sensitivity in an immunologic assay, for either viral antigen (diagnostic testing) or host antibodies (serologic testing) is a trade-off between assay detection of an immunologic reaction, and setting high stringency for test specificity.
Potential for False Negatives: Triaging and cohorting of patients suspected of having COVID-19 at the time of admission to hospital, including potential for nosocomial spread of SARS-CoV-2; discharge of COVID-19 patients to the community; testing of symptomatic patients in congregate living facilities; ambulatory testing of persons following exposure to COVID-19 patients; management of quarantine for asymptomatic persons (eg, travel restrictions); testing and screening of HCWs (including for congregate living facilities and home care), frontline, and essential workers; broad community-based testing of asymptomatic persons who might be infected by SARS-CoV-2 and capable of infecting others.
Potential for False Positives: Testing and screening of HCWs, students, teachers, childcare centers, frontline, and essential workers; and other workforce populations; with ensuing impact on workforce availability and the status of facility operations.
Given the panoply of available SARS-CoV-2 diagnostic tests, and especially with the extensive deployment of rapid SARS-CoV-2 PCR and rapid SARS-CoV-2 antigen testing as Fall 2020 arrived, consortium members had to maintain a high profile in our institutions to help inform institutional decision-making for deployment (and interpretation) of different test platforms. The development of rigorous institutional algorithms for patient evaluation and testing (including repeat testing), while allowing for the potential of false negatives or false positives, was assurance that patient safety would be served successfully in the midst of the inherent limitations of laboratory tests.
These many considerations then coalesced into articulation of the practical challenges the consortium laboratories actually faced in deploying SARS-CoV-2 testing, as given in Table 8. As with the prior information, the impact of these variables on interpretation of test results helped empower consortium members in providing leadership within our home institutions for design of clinical care pathways.
Practical Challenges in Deploying SARS-CoV-2 Testing.
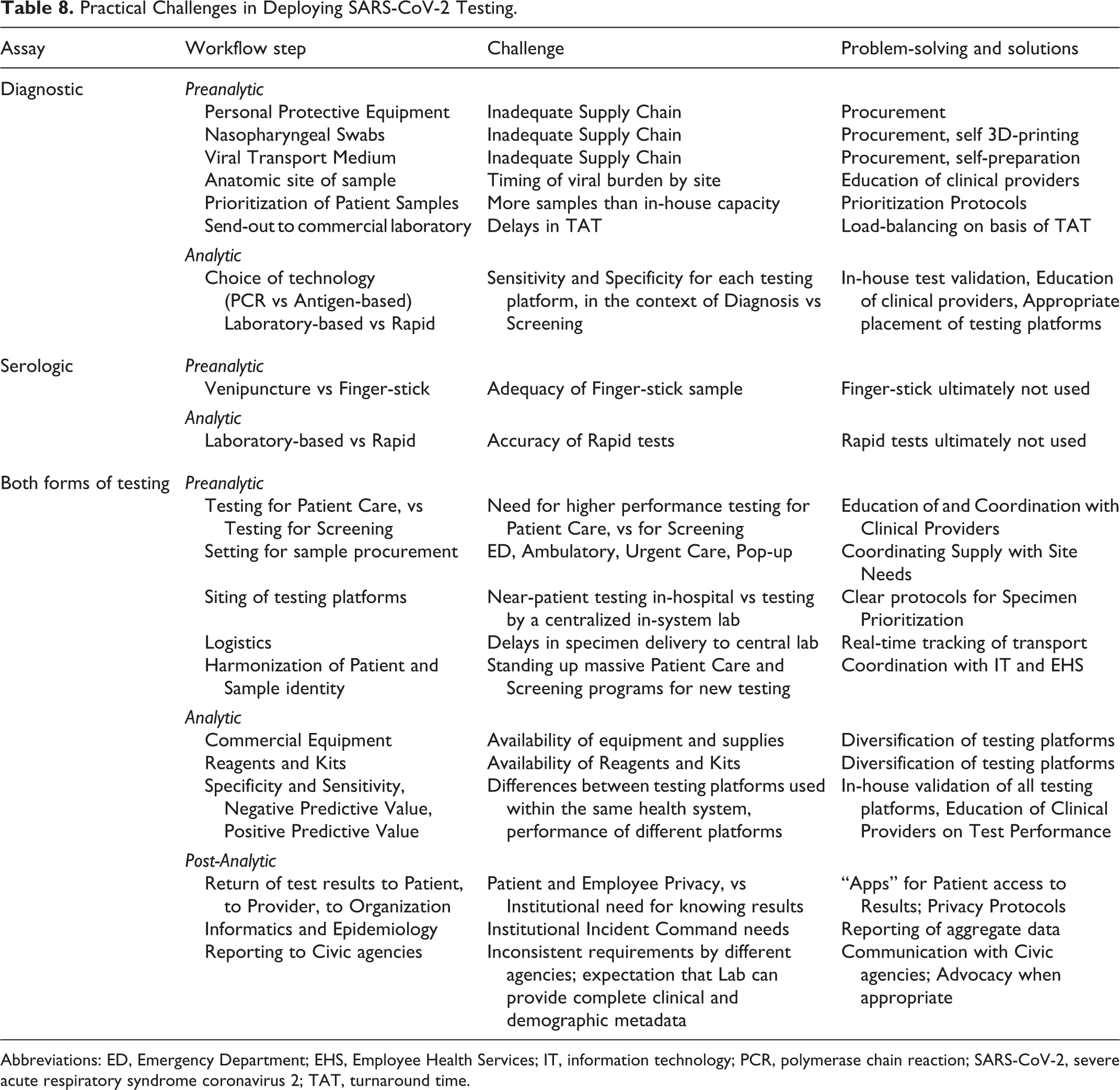
Abbreviations: ED, Emergency Department; EHS, Employee Health Services; IT, information technology; PCR, polymerase chain reaction; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; TAT, turnaround time.
Deliverable 5: Observational and Investigative Research
Once the immediate needs of April 14-17, 2020, were met, with delivery of the consortium report to the Commissioner of Health, the first formal meeting of what was agreed upon as the “consortium” was on April 22, 2020. In this founding meeting, the premise was strongly argued that the consortium must commit to the process of discovery and advancing knowledge for the new disease that was COVID-19. Table 9 gives the questions that consortium members considered of highest priority. Over the first 10 months of the pandemic (March-December, 2020), 212 peer review publications were authored by consortium departments of pathology and laboratory medicine members. These publications are listed in Supplemental Appendix 1.
SARS-CoV-2 Questions for Observational and Investigative Research: April 2020.

Abbreviations: PCR, polymerase chain reaction; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Discussion
The greater New York metropolitan area was the COVID-19 pandemic epicenter of the United States in the March to May 2020 time frame. The health systems of this region experienced unprecedented stress in providing care to COVID-19 patients who were admitted to our hospitals, with the hospitals of the consortium member institutions providing a major fraction of this inpatient care. The clinical laboratories of consortium institutions were thus faced with establishing diagnostic testing for this new infectious agent, SARS-CoV-2, on an unprecedented accelerated time frame. Ad hoc communication between institutions was occurring from mid-March 2020 onward, both regionally and at the national level. It was the coalescence of this New York State SARS-CoV-2 Testing Consortium starting Tuesday April 14, 2020, that enabled formalization of interinstitutional communication and established a regular mechanism through the GNYHA for collective communication with state leadership.
As experienced by our consortium institutions, several conclusions are evident. First, the COVID-19 pandemic is first-and-foremost a regional event, differing in timing when compared with other regions of the country. Hence, the response of medical and societal communities is and must be regional and will follow regional timelines. Second, the regional community benefits from communication and collaboration, as shared experiences can help educate and prepare all members of a statewide laboratory community in dealing with a universal and rapidly evolving health threat such as COVID-19. Third, the establishment of our regional laboratory testing consortium in mid-April 2020 established a bidirectional communication channel with civic authorities and most especially with the NYS public health laboratories. This communication channel helped inform both the state’s management of regional events and consortium members’ responses to meeting the state’s needs.
Lessons learned are as follows. First, state leadership was in need of understanding state capabilities for laboratory testing, both as regard to testing technologies and test capacities. The consortium provided a formal mechanism for the state to know what the assembled academic medical institutions were capable of doing, and to receive our recommendations for testing platform strategies to deal with SARS-CoV-2 as a new infectious pathogen. Besides making clear to state leadership that the clinical laboratories of the state’s academic health systems were already making a substantial contribution to the regional pandemic response, the consortium also made clear that coordinated work between state leadership and regional laboratories was essential. This included support at the community level of the preanalytical and post-analytical workflow and infrastructure, public education, collaboration between regional clinical laboratories and the state public health laboratory, and state support for testing costs incurred on behalf of nonpaying populations.
Second, the great challenges being experienced by consortium institutions in obtaining supply for the laboratory testing of SARS-CoV-2 was important to communicate to state leadership, so as to keep the state informed in its own national advocacy for state needs. The sharing of supply chain experiences between consortium institutions was critical in informing individual institutions about what might or might not be possible through our own institutional procurement efforts.
Third, the consortium provided a formal mechanism for timely updates on policies and programs emanating from both the state and New York City, and clarification through all 3 agencies participating in the consortium (GNYHA, NYS DOH, and HANYS) about the intent and implications of those policies.
Fourth, the consortium mechanism enabled sharing on a regular basis of real-world experience between member institutions about the deployment of SARS-CoV-2 testing, leading to collaborative establishment of a foundational body of knowledge regarding the laboratory and implementation science for testing for this new infectious agent.
Fifth, real-world experience shared by the laboratory leadership of consortium institutions constituted a critical source of educational content. The timely discussions of the consortium, as reflected in Tables 3 to 9, empowered each participant in educational efforts within our own institution. Each laboratory leader could speak with authority both about the real-world experience of regional laboratories, and the shared challenges of addressing institutional needs for SARS-CoV-2 testing. This knowledge also was of immediate value in laboratory-level problem-solving and decision-making during this rapidly evolving crisis.
Sixth, and perhaps most breathtaking, the commitment of consortium institutions ab initio to the creation of new knowledge about COVID-19 helped set the tone for consortium operations. Specifically, the purpose of the consortium was not just to navigate through the crisis; it was to establish a regional community for leadership in the fight against this virus and the pursuit of scientific understanding and new treatments. Supplemental Appendix 1 lists medical literature publications by faculty in consortium departments of pathology and/or laboratory medicine faculty. One consortium publication will be called out especially: the world’s first major autopsy series of COVID-19 patients that documented widespread thrombotic microangiopathy affecting most major organs, especially the central nervous system. 44 Sadly, the senior author of this study, Mary Fowkes, MD, PhD, died of an unrelated medical event in November 2020. She will be greatly missed.
The consortium worked assiduously to avoid the risk of becoming a trade organization, with the implications of market manipulation or influence. The aggregate testing capacity reported to the state from April 20, 2020, to May 24, 2020, was for the express purpose of informing the state of how rapidly these consortium institutions were helping to address daily testing capacity required for the state response. With cessation of this reporting, all subsequent discussions were nonquantitative. Likewise, discussion of testing instrumentation being used revealed only what was becoming apparent across the nation: that clinical laboratories were required to acquire, validate, and deploy a multiplicity of testing platforms for both SARS-CoV-2 molecular diagnosis and serology. The commonality of the named manufacturers served only to underscore that we shared common challenges.
We must emphasize that the dominant theme of our regular discussions was Supply Chain. For the duration of the COVID-19 pandemic through to the present, our laboratories have had inadequate access to reagents, kits, and in some instances, instrumentation for the performance of requisite testing. We consider that our New York State institutions are sharing a common national experience: the COVID-19 testing needs of hospital-based and regional laboratories are underprioritized in the national response to this pandemic. We advocate that the national COVID-19 testing supply chains must be rebalanced with assignment of greater priority to regional and local clinical laboratories. 45
Pitfalls and potential shortcomings in consortium activities are the following. First was the delay in the consortium actually forming, April 14, 2020. In retrospect, mid-February 2020 would have been a more opportune time for bringing the regional laboratory leadership together. It is our intent to keep this regional consortium active for the indefinite future, not just in anticipation of potential future crises but to foster continued communication between our academic clinical laboratories as regional colleagues and maintain a continued state of regional preparedness. Second, the balance between reporting institutional testing capacities to the state and not acting as a trade organization was a challenge, and we were relieved to end that reporting after 5 weeks. By that time, New York State had set up survey mechanisms for obtaining the same information from individual laboratories statewide. Third, it was essential to verify with consortium participants that our regular meetings were of value. From time to time, the Agenda included formal discussion of consortium structure and operations, to verify that the time spent was useful.
Preparedness for infectious outbreaks has long been a concern. The 2009 novel H1N1 gave opportunity for articulation of the principles for the laboratory surge response to a pandemic event. 46 The intercurrent pandemics of SARS, Middle East respiratory syndrome, and Ebola virus gave further impetus for laboratory preparedness, as articulated in the national plan for pandemic response issued in 2006. 47 The COVID-19 pandemic revealed the staggering impact that a major world pandemic can have on the global supply chain for laboratory testing and the resultant inability of clinical laboratories to respond to the needs of their region. 48 Under extraordinarily trying circumstances, consortium institutions were able to stand-up SARS-CoV-2 molecular testing 49 and serological testing 11,12 rapidly and effectively. But the lessons of this pandemic must still be learned and carried forward in anticipation of future such events. In bringing this consortium experience forward, we hope that our statewide effort may serve as an exemplar for other regional laboratory communities.
Supplemental Material
Supplemental Material, sj-pdf-1-apc-10.1177_23742895211006818 - The New York State SARS-CoV-2 Testing Consortium: Regional Communication in Response to the COVID-19 Pandemic
Supplemental Material, sj-pdf-1-apc-10.1177_23742895211006818 for The New York State SARS-CoV-2 Testing Consortium: Regional Communication in Response to the COVID-19 Pandemic by James M. Crawford, Maria E. Aguero-Rosenfeld, Ioannis Aifantis, Evan M. Cadoff, Joan F. Cangiarella, Carlos Cordon-Cardo, Melissa Cushing, Aldolfo Firpo-Betancourt, Amy S. Fox, Yoko Furuya, Sean Hacking, Jeffrey Jhang, Debra G. B. Leonard, Jenny Libien, Massimo Loda, Damadora Rao Mendu, Mark J. Mulligan, Michel R. Nasr, Nicole D. Pecora, Melissa S. Pessin, Michael B. Prystowsky, Lakshmi V. Ramanathan, Kathleen R. Rauch, Scott Riddell, Karen Roach, Kevin A. Roth, Kenneth R. Shroyer, Bruce R. Smoller, Steven L. Spitalnik, Eric D. Spitzer, John E. Tomaszewski, Susan Waltman, Loretta Willis and Zeynep Sumer-King in Academic Pathology
Footnotes
Acknowledgment
The New York State SARS-CoV-2 Testing Consortium gratefully acknowledges the role of Greater New York Hospital Association in serving as convening authority for this consortium (Susan Waltman, Esq.; Laura Alfredo, Esq.; Zeynep Sumer-King), and Jill Taylor, PhD, Director, Wadsworth Center, NYS Department of Health and Brad Hutton, 2nd Deputy Commissioner, Office of Public Health at NYS Department of Health for participation in consortium discussions. We express our deepest thanks to the laboratory professionals of our respective institutions, who have steadfastly provided the highest quality of laboratory services over the length and duration of the COVID-19 pandemic.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
