Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Objective
Objective HWC3.1: Morphology of Acute Leukemia and Lymphoma. Describe the morphologic features that characterize typical cases of acute leukemia and lymphoma.
Competency 2 Organ System Pathology; Topic HWC: Hematopathology—White Cell Disorders, Lymph Nodes, Spleen, and Thymus; Learning Goal 3: Classification of Leukemia and Lymphomas
Secondary Objectives
Objective SP5.1: Special Studies. Describe the roles of immunohistochemistry, flow cytometry, cytogenetics, and molecular diagnostics in the diagnosis and classification of lymphoma and explain how, with examples, different techniques are most appropriate in diagnosis, staging, and management of disease.
Competency 3 Diagnostic Medicine and Therapeutic Pathology; Topic SP: Surgical Pathology; Learning Goal 5: Classification of Leukemia and Lymphomas.
Patient Presentation
A 64-year-old female with past medical history of acute myeloid leukemia (AML) diagnosed 5 years prior presentation and in complete remission after induction and consolidation chemotherapy presented to her primary care physician for a gluteal cleft mass. She noticed it approximately 2 months prior presenting to her physician, initially thinking it to be an insect bite. However, it gradually worsened in severity with increasing pain, swelling, and redness. On physical examination, there is a 4-cm mass in the right superior gluteal cleft with surrounding area of erythema. The mass appears as a reddish ulceration with some yellow crusting with raised borders. No fluid can be expressed and no palpable underlying fluctuance is noted. The overlying skin is intact without bleeding and there are no adjacent vesicular lesions.
Diagnostic Findings, Part 1
Considering the patient’s immunocompromised state due to history of cancer and chemotherapy along with classic signs of inflammation (gluteal mass with color, dolor, rubor, and tumor), a complete blood count (CBC) was ordered. The CBC result is shown in Table 1.
Patient’s Complete Blood Count Results at Presentation.*
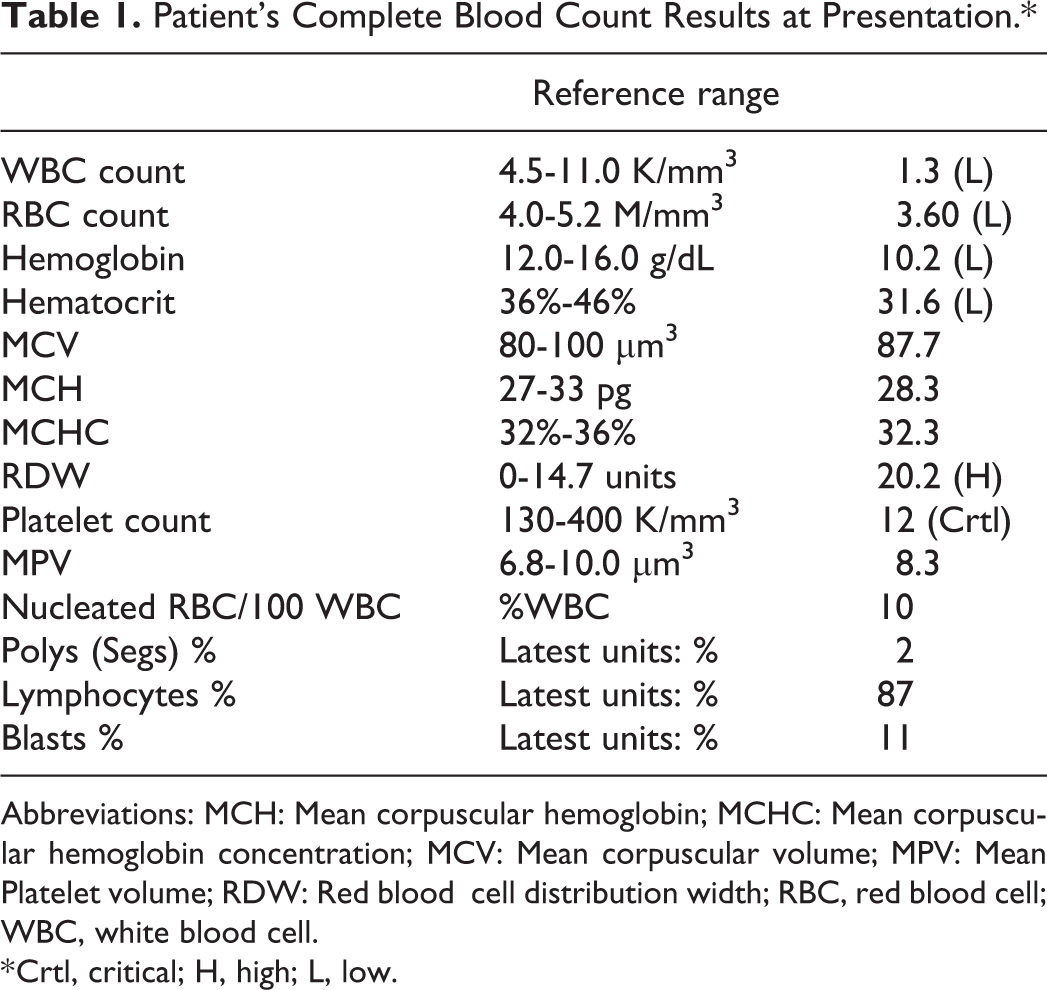
Abbreviations: MCH: Mean corpuscular hemoglobin; MCHC: Mean corpuscular hemoglobin concentration; MCV: Mean corpuscular volume; MPV: Mean Platelet volume; RDW: Red blood cell distribution width; RBC, red blood cell; WBC, white blood cell.
* Crtl, critical; H, high; L, low.
Questions/Discussion Points, Part 1
What Is the Differential Diagnosis of the Clinical Findings in This Patient?
Given patient’s history of underlying malignancy status post-chemotherapy, the differential diagnosis to consider is malignancy (a newly developed malignancy versus recurrent AML) versus an infectious process. The infections to consider in an immunocompromised host will be opportunistic infections, viral, fungal, and mycobacterial infections.
What Does the CBC Show?
The CBC showed normocytic anemia (Mean corpuscular volume (MCV): 87.2 and hemoglobin: 7.9 g/dL) with marked leukopenia and thrombocytopenia (white blood cell count: 0.9 K/mm3 and platelet: 24 K/mm3). Based on the CBC results, a peripheral blood smear (Figure 1) was prepared and subsequently.
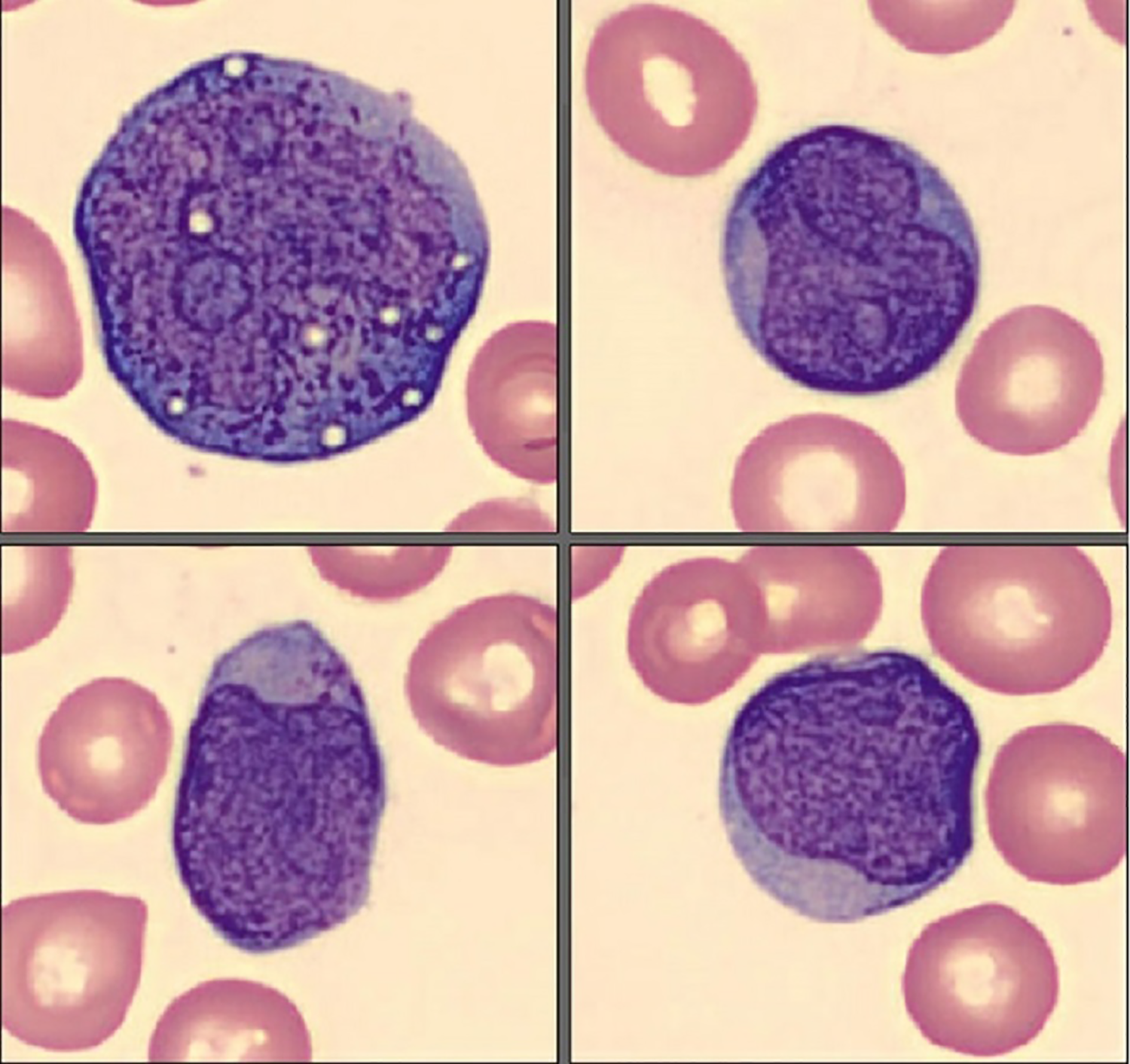
High power (Cellavision software image of peripheral blood smear equivalent to 100×) view of peripheral blood smear showing peripheral blasts. The cells are large in size with open chromatin, irregular nuclear membrane, high Nuclear-Cytoplasmic ratio, and occasional prominent nucleoli.
Describe the Cells in the Peripheral Blood Smear
The cells in the peripheral blood smear (Figure 1) are large with large nuclei, high N:C ratio, lacy chromatin, some with prominent nucleoli and some with small cytoplasmic granules, which are characteristic of immature cells.
Diagnostic Findings, Part 2
Based on the presence of immature cells in the peripheral blood, concern was raised for recurrent AML in the patient. Subsequently a bone marrow biopsy and a biopsy of the gluteal mass was performed.
Questions/Discussion Points, Part 2
What Are the Histologic Findings on the Gluteal Mass Biopsy in this Case?
Histologic evaluation by hematoxylin and eosin stain shows skin with underlying soft tissue diffusely infiltrated by immature neoplastic cells with complete distortion of the papillary and reticular dermis (Figure 2). The cells are immature with scant cytoplasm, high nuclear to cytoplasmic ratio, round to oval nuclei with finely dispersed chromatin, and prominent nucleoli (Figure 3). Numerous immature cells are noted (Figure 4).
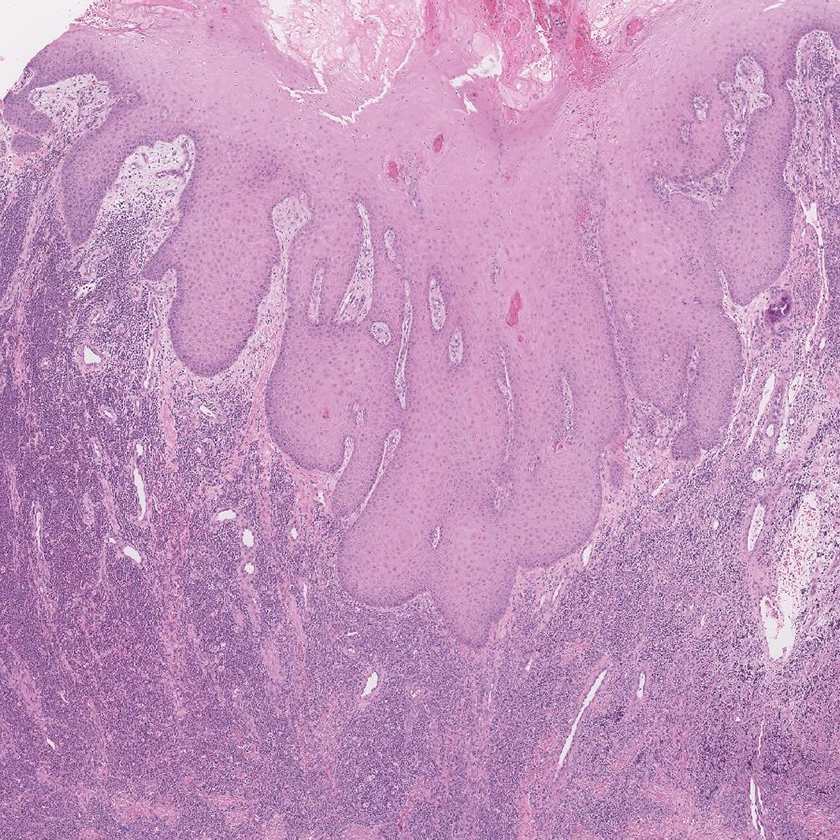
Low power (2×) view of hematoxylin and eosin (H&E) stained slide showing an expanded epidermis (acanthosis) overlying an expanded dermis infiltrated diffusely by an atypical cellular infiltrate. The cells in the infiltrate are immature cells involving the papillary and reticular dermis.
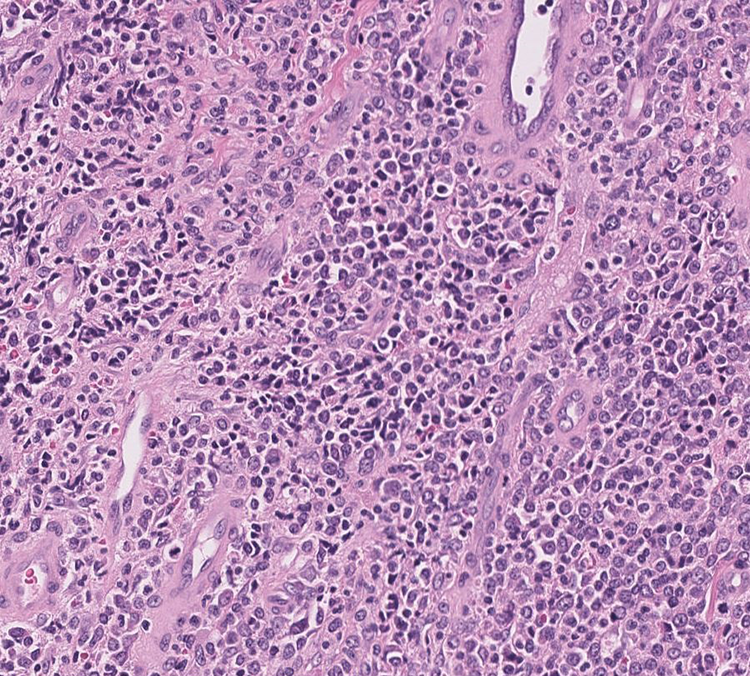
A high-power view (40×) of the tumor cells from the skin lesion. The cells are immature with scant cytoplasm, high nuclear to cytoplasmic ratio, round to oval nuclei with finely dispersed chromatin, and prominent nucleoli. There are occasional small mature lymphocytes in the background. Increased number of mitotic figures are noted. There is no necrosis and acute inflammation.
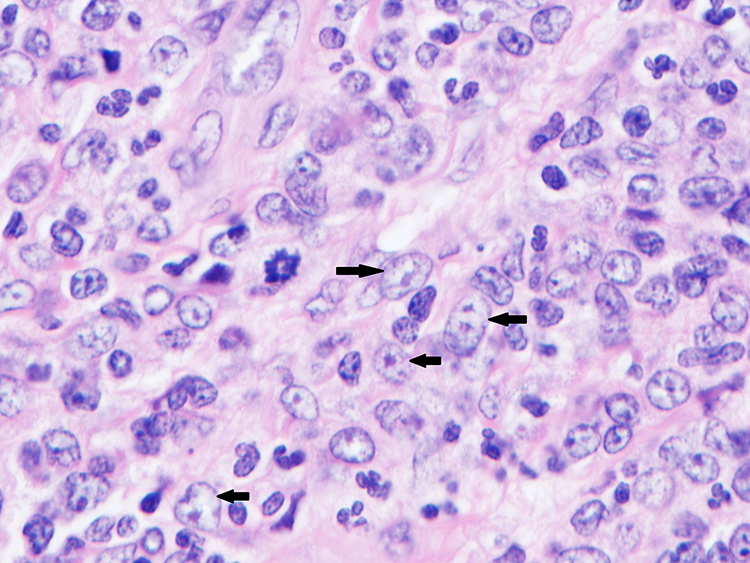
Oil emersion field (100×) view of numerous immature cells (see black arrows). The cells are large in size, have high nuclear to cytoplasmic ratio, round to oval nuclei with irregular nuclear membrane, finely dispersed chromatin, and prominent nucleoli.
Describe the Findings of the Bone Marrow Biopsy
Bone marrow biopsy shows a hypercellular marrow with diffuse proliferation of large atypical cells with high N:C ratio, lacy chromatic, and prominent nucleoli comprising 40% of the marrow. Occasional eosinophilic granules are noted in the neoplastic cells.
Diagnostic Findings, Part 3
Immunohistochemical stains are performed on the bone marrow biopsy. The neoplastic cells express CD117, CD34, and myeloperoxidase (MPO) by immunostaining and flow cytometry. The morphologic and immunophenotypic findings of the blasts and presence of more than 20% blasts in the bone marrow are consistent with an AML. The biopsy of the mass shows a diffuse proliferation of large atypical cells. The neoplastic cells are strongly positive for CD43, MPO, and CD68 and negative for CD4 (Figure 5), CD34, CD117, TdT, AE1/AE3, and Melan-A. The neoplastic cells also show a high proliferation rate with numerous mitotic figures and increased Ki-67.
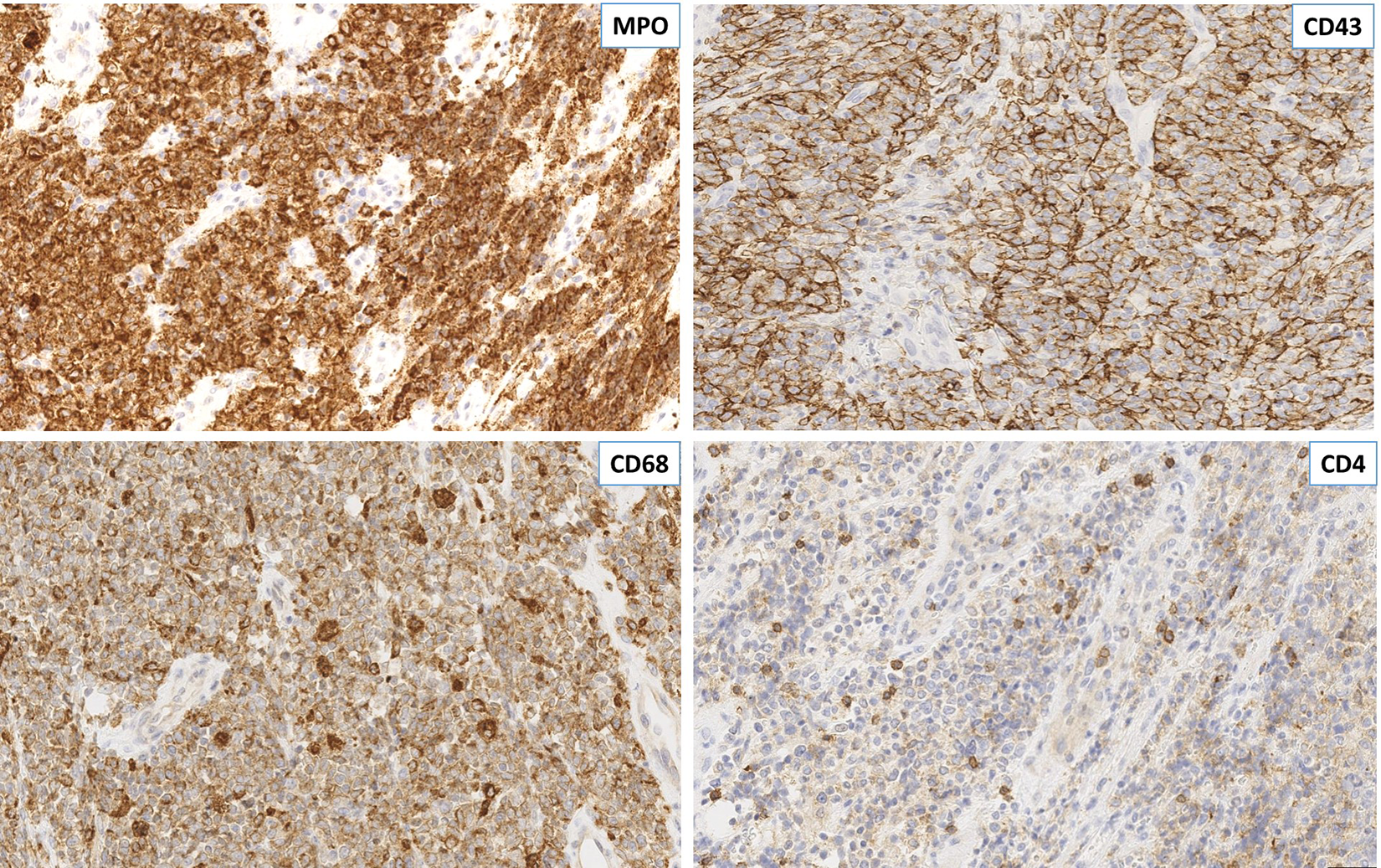
High-power view (40×) of the neoplastic cells showing strong positivity for CD43, myeloperoxidase (MPO), and CD68 and are negative for CD4.
Questions/Discussion Points, Part 3
What Is Your Diagnosis Based on the Immunohistochemical Findings?
CD34 is expressed on the immature cells along with CD117 which also indicated a myeloid origin of the cells. Myeloperoxidase is expressed on myeloid lineage cells. CD68 is a histiocytic marker and can be expressed in myeloid cells. The neoplastic cells staining for these markers show them being immature and of myeloid lineage. TdT is a lymphoblastic marker, AE1/AE3 highlights epithelial cells, and Melan-A is expressed in melanoma cells. The lack of expression of these markers help exclude epithelial neoplasm, melanoma, and lymphoblastic origin of these cells. Based on the immunoprofile of the neoplastic cells expressing CD34, CD117, and MPO in the bone marrow and MPO and CD68 in the biopsy section along with immature morphologic features on the cells findings are consistent with an immature myeloid process.
What Explains the Difference in Immunomarkers Expressed in the Bone Marrow and Gluteal Mass Neoplastic Cells?
There can be different staining of the blasts in the marrow and the tissue, depending on the maturation pattern of the blasts present in each tissue examined. Tumors with more immature myeloid profile express CD33, CD34, CD117, CD68, and staining for MPO is sometimes inconsistent. However, in promyelocytic cases of myeloid sarcoma (MS), generally the blasts express MPO and are negative for CD34.
How Would You Interpret the Immunophenotypic Findings of the Neoplastic Cells in the Biopsy?
The neoplastic cells express myeloid markers such as MPO and CD68 without expression of cytokeratin and melanin markers. The morphologic and immunophenotypic findings are consistent with an MS. The proliferation index (Ki67) is generally high with 50% to 95% cells being positive. 2,3
What Is Myeloid Sarcoma?
According to World Health Organization (WHO) 2017, MS is a mass lesion consisting of blasts of myeloid lineage, which may or may not have maturation, and characteristically present at an anatomical site outside of the bone marrow. 4 However, presence of myeloid blasts infiltrating any organ of the body cannot be considered as MS unless there is complete effacement of the tissue architecture. 5 Myeloid sarcoma has been described in the earlier literature as a green colored tumor due to the greenish hue on fresh specimens as result of MPO production and has its alternate name as “chloroma.” Later, it was named as “granulocytic sarcoma” by Rappaport followed by “MS” by WHO in 2002, due to the higher expression of MPO staining.
What Is the Epidemiology of Myeloid Sarcoma?
Generally older males (male-to-female ratio is 1.2:1) with a median age of 56 years are affected, although case reports have described MS occurring in 1 month olds to 89 year olds. 6,7
What Is the Clinical Presentation of Myeloid Sarcoma?
Myeloid sarcoma has no predilection toward any specific anatomical site, but literature describes skin, lymph nodes, gastrointestinal tract, bone, soft tissue, and testes to be the most favorable sites of involvement.3-7 Rarely (<10%), MS presents at multiple sites of the body. 6,7 Clinical presentation of MS depends on its location and generally caused by the mass effect of the tumor or due to its infiltration of the organ system. Most importantly, it is to be kept in mind that, in 25% of cases, MS can occur in absence of an underlying AML or any other myeloid neoplasm. Diagnosis of MS is equivalent to diagnosis of AML even in the absence of bone marrow infiltration of blasts. Sometimes the development of MS can herald the onset or overlap with the development of AML. It can also represent a blast transformation of myelodysplastic syndromes or myeloproliferative neoplasms. 6,7
What Is the Differential Diagnosis and Immunophenotype of Myeloid Sarcoma?
The main differential diagnosis to consider is that of B or T lymphoblastic lymphomas. However, these diagnoses can be easily discerned using cytochemical and immunohistochemical stains as well as with flow cytometry. The main cytochemical stains to consider for MS are Chloroacetate esterase, MPO, and nonspecific esterase. Immunohistochemically, the paraffin embedded sections of MS can be stained with CD68, CD43, MPO, CD117, lysozyme, and CD34. To eliminate other differential diagnoses, B- and T-cell markers, especially CD20, Pax-5, and CD3, should be tested. 8 Using flow cytometry, CD13, CD33, CD117, and MPO can help to identify blasts with myeloid differentiation. Immunohistochemistry shows CD68 as the most commonly expressed marker, 9 followed by MPO, CD117, CD99, lysozyme, CD34, terminal deoxynucleotidyl transferase, CD56, CD61, CD30, glycophorin A, and CD4. 10,11 This panel helps in the differentiation of MS from lymphoblastic lymphoma, Burkitt lymphoma, and diffuse large B-cell lymphoma, blastic plasmacytoid dendritic neoplasm, neuroblastoma, rhabdomyosarcoma, primitive neuroectodermal tumor, and medulloblastoma. 9 The proliferation index (KI-67/MIB-I) is frequently high (50%-95%). 3,8
What Are the Treatment Options and Prognosis of Myeloid Sarcoma?
Radiation, chemotherapy, targeted molecular therapy, and surgery with debulking with or without bone marrow transplantation have been considered as current modalities for MS treatment. 8 Tumor behavior and response to treatment does not depend on age, sex, anatomical site of involvement, or type of presentation. 7 Bone marrow transplantation may or may not be helpful in MS; however, people do consider it to be a therapy after induction chemotherapy in patients with remission. 8 Isolated MS patients have comparable disease-free survival rates with patients with AML without MS when managed with AML-based induction regimens. 12,13
Studies show that overall survival of patients receiving systemic chemotherapy at diagnosis was significantly longer. 14,15 Published reports also mention that systemic chemotherapy along with radiotherapy, although there is no improvement in overall survival in isolated MS, radiotherapy may prolong failure-free survival. 16 In the era of molecular medicine, targeted therapies with tyrosine kinase inhibitors have found a role in the treatment of AML cases with complex cytogenetic abnormalities. 17 The emerging data on the molecular profile of different AML and their targeted treatment options might shine light on different treatment options with prognostic implications in subgroups of MS patients.
What Are the Different Subtypes of AML as per WHO 2017?
WHO classifies AML into 6 main categories with multiple subcategories
18
: Acute myeloid leukemia with recurrent genetic abnormalities: This classification of AML is based on the genetic findings noted with chromosome analysis or other molecular techniques. A few subtypes of this category include AML t(8;21), AML inv 16, or t(16;16) and APL t (15;17) which generally are considered to have a good prognosis. Acute myeloid leukemia with myelodysplasia-related changes: This subtype of AML occurs in patients with a prior history of myelodysplastic syndrome, and these patients have dysplastic features in their blood and bone marrow cells. Therapy-related myeloid neoplasms: This type of AML occurs following cytotoxic chemotherapy (alkylating agents/topoisomerase inhibitors) and radiation therapy. Acute myeloid leukemia not otherwise specified: This encompasses the cases that do not fulfill the criteria to be included in any of the above groups and examples of this subtypes include AML with minimal differentiation, AML without maturation, AML with maturation. Myeloid sarcoma. Myeloid proliferations associated with Down syndrome: This subtype of AML is related to the patients with Downs syndrome.
Teaching Points
Myeloid sarcoma is a subtype of AML based on WHO classification.
Myeloid sarcoma, an extramedullary tumor mass of myeloid blasts which may or may not have maturation, is equivalent to AML regardless of involvement of the bone marrow.
In 25% of cases, MS can occur in absence of an underlying AML or any other myeloid neoplasm.
In some cases, the development of MS can herald the onset or overlap with the development of AML.
Myeloid sarcoma can represent a blast transformation of myelodysplastic syndromes or myeloproliferative neoplasms.
It may involve any site of the body although a predilection for skin, lymph nodes, gastrointestinal tract, bone, soft tissue, and testes is reported.
Presence of myeloid blasts infiltrating extramedullary site cannot be considered as MS unless there is complete effacement of the tissue architecture.
CD68 has been shown to be the most commonly expressed marker by MS, followed by MPO, CD117, CD99, lysozyme, CD34, terminal deoxynucleotidyl transferase, CD56, CD61, CD30, glycophorin A, and CD4 with a high proliferation rate.
Diagnostic modalities include a combination of immunohistochemistry, flow cytometry, cytogenetic, and molecular studies besides radiology studies.
Current treatments for MS include radiation, chemotherapy, targeted molecular therapy, and surgery with debulking with or without bone marrow transplantation.
Footnotes
Authors’ Note
Each author participated in the design, writing, and editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
