Abstract
Keywords
Primary Objective
Competency 2: Organ System Pathology; Topic HWC: Hematopathology-White Cell Disorders; Learning Goal 3: Classification of Leukemia and Lymphomas.
Secondary Objectives
Competency 3: Diagnostic Medicine and Therapeutic Pathology; Topic SP: Surgical Pathology; Learning Goal 5: Classification of Leukemia and Lymphomas.
Competency 3: Diagnostic Medicine and Therapeutic Pathology; Topic SP: Surgical Pathology; Learning Goal 1: Role in Diagnosis.
Patient Presentation
A 60-year-old male presents to the clinic with a lump in the left groin that he initially noticed about 4 weeks ago when he was in the shower. He reports that the lump has increased in size since then and has not been associated with any pain or tenderness. On several occasions over the last month, he has awoken at night with sweating and has felt achy and feverish at times, although he has not taken his temperature. These symptoms are relieved by acetaminophen. He is otherwise healthy except for mild hypertension controlled with diet and exercise. Review of systems is otherwise unremarkable.
Diagnostic Findings, Part 1
Vital signs are normal. Physical examination reveals a 2.5-cm firm, nontender, freely moveable mass consistent with a lymph node in the left inguinal region. No lymphadenopathy is noted elsewhere. No skin lesions are present. The remainder of the examination is unremarkable. Laboratory studies show lactase dehydrogenase 289 units/L (normal 100-190 units/L). Chest X-ray is unremarkable.
Questions/Discussion Points, Part 1
What Is the Differential Diagnosis of Lymphadenopathy in Adult Patients, and What Are the Risk Factors for Malignancy?
Lymphadenopathy is defined as lymph nodes that are greater than 1 cm in size. Etiologies of lymphadenopathy include malignancies, infections, autoimmune disorders, miscellaneous and unusual conditions, and iatrogenic causes. The most common underlying malignancies include lymphoma, leukemia, multiple myeloma, and metastatic carcinoma. Infections include bacterial infections, viral infections, tuberculosis, syphilis, and toxoplasmosis. Autoimmune disorders associated with lymphadenopathy include systemic lupus erythematosus, Still disease, Sjögren syndrome, and rheumatoid arthritis. Miscellaneous and unusual conditions are varied but include Castleman disease, Kikuchi lymphadenitis, Kimura disease, IgG4-related disease, and sarcoidosis. Iatrogenic causes chiefly include medications and serum sickness. The most likely cause of lymphadenopathy can often be identified by history and physical examination alone.
Only 1.1% of cases of unexplained lymphadenopathies are due to underlying malignancy. Risk factors for malignancy include older age, long duration of lymphadenopathy (greater than 4-6 weeks), generalized lymphadenopathy, male sex, supraclavicular location of lymphadenopathy, and systemic symptoms, including fever, night sweats, and weight loss. Age is a particularly important factor: Malignancy is identified in 4% of cases of unexplained lymphadenopathy in patients aged 40 years and older but in only 0.4% in patients younger than 40 years. 2
What Procedures Can Be Performed to Obtain a Specimen for the Evaluation of Lymphadenopathy?
The most common procedures for tissue evaluation of lymphadenopathy are fine-needle aspiration (FNA), needle core biopsy, and excisional biopsy. Fine-needle aspiration uses a narrow-gauge needle to sample cells for cytology and has the advantage of being the least invasive means to obtain diagnostic material from lymph nodes. In some situations, sampling can be performed endoscopically and may be guided by imaging methods such ultrasound or computed tomography. In addition to cytologic preparations, the material obtained can be assessed by ancillary techniques such as flow cytometry, genetic testing, and microbiologic culture. Immunohistochemical stains can be performed on cell block material. The major limitations of FNA are the total amount of material obtained and the inability to appreciate tissue architecture. The latter is particularly important in the diagnosis and classification of lymphomas. Needle core biopsies utilize a larger bore needle that is more invasive than FNA, but deep anatomic sites can still often be reached with imaging guidance, and more overall tissue can be obtained. Fine-needle aspiration is sometimes performed during the same procedure for cytological preparations and ancillary testing such as flow cytometry. Although appreciation of overall lymph node architecture remains limited, needle core biopsy is often sufficient for definitive diagnosis and classification of lymphoma. Excisional lymph node biopsy affords the most comprehensive diagnostic evaluation of lymphadenopathy but is the most invasive and may not be feasible in critical anatomic sites or in high-risk patients. False-negative results from FNA and needle core biopsies can occur if inadequate lesional tissue is sampled. Even when lesional tissue is present, it may not reflect heterogeneity in the underlying process, such as the presence of more than 1 tumor grade. Pathologists should request a larger biopsy if FNA or needle core biopsy material cannot be adequately evaluated to guide clinical management, or if discussion with the clinician suggests a discrepancy between the pathologic diagnosis and the clinical presentation.
Diagnostic Findings, Part 2
Because of the clinical suspicion of lymphoma and the easily accessible location of the lymph node, an excisional biopsy rather than an FNA or needle core biopsy was performed. Histologically, the lymph node architecture is effaced by a diffuse proliferation of atypical mononuclear cells (Figure 1A). On high-power magnification, the cells are medium to large in size and have ovoid or irregular nuclei. Some of the cells have eccentric, kidney-shaped nuclei, the so-called “hallmark cells” (Figure 1B). Immunohistochemical stains were performed. The atypical cells are diffusely and strongly positive for CD30 (Figure 2A) and negative for anaplastic lymphoma kinase (ALK; Figure 2B). They were also positive for CD2 and negative for CD3, CD4, CD8, CD15, CD20, CD45, PAX5, T-cell-restricted intracellular antigen-1 (TIA-1), and granzyme B (not shown).
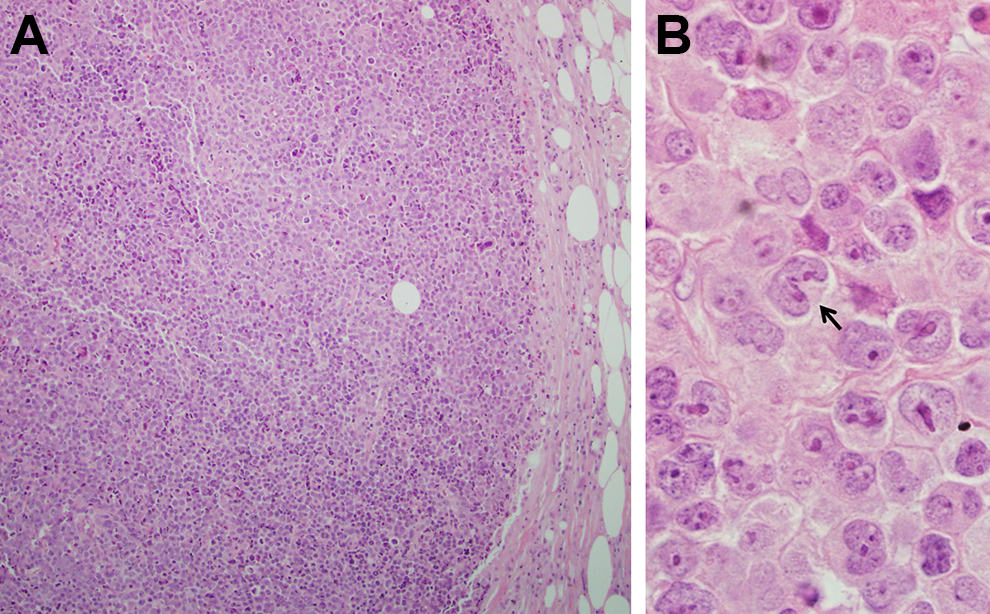
Histological findings of the left inguinal lymph node. A, A low-magnification image shows the lymph node architecture to be effaced by a diffuse proliferation of atypical mononuclear cells (hematoxylin and eosin [H&E], ×100). B, At higher magnification, medium-sized to large atypical lymphocytes are seen with ovoid or irregular nuclei, including some cells with eccentric, kidney-shaped nuclei (the so-called “hallmark cells,” arrow; H&E, ×1000).
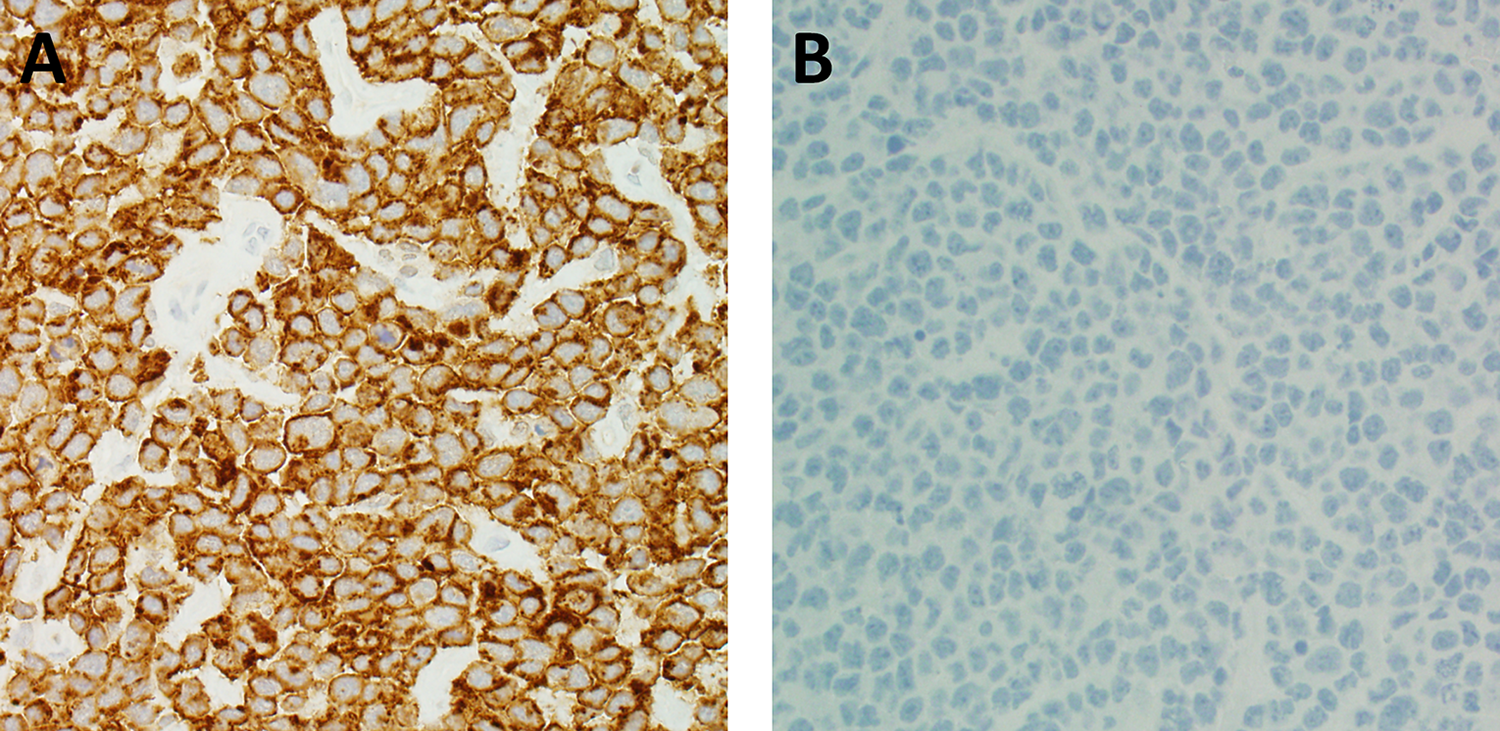
A, The tumor cells are diffusely and strongly positive for CD30 by immunohistochemistry (×400). B, The tumor cells are negative for ALK by immunohistochemistry (×400). ALK indicates anaplastic lymphoma kinase.
Questions/Discussion Points, Part 2
What Is the Diagnosis Based on Pathological Examination of the Lesion and Correlation With Clinical Findings?
Based on the histologic and immunohistochemical features of the tumor and the clinical presentation, the diagnosis is ALK-negative anaplastic large cell lymphoma (ALCL). Classic Hodgkin lymphoma (CHL) is another malignant lymphoma containing large neoplastic CD30-positive cells. However, CHL more typically shows scattered neoplastic cells in a mixed inflammatory background rather than sheets of tumor cells as in this case. In addition, although immunophenotypic features may vary, CHL often co-expresses CD15 and the B-cell transcription factor PAX5 and is less commonly positive for pan-T-cell markers such as CD2.
ALCLs comprise a group of T-cell lymphomas with common pathologic features but with varying genetics and clinical features. The tumor cells are typically large, and hallmark cells should be present at least focally for a diagnosis of ALCL. These cells have eccentric horseshoe-shaped or kidney-shaped nuclei and a prominent Golgi zone. Immunophenotyping is mandatory for diagnosis. By definition, all ALCLs express CD30, a type 1 transmembrane protein expressed by activated lymphocytes that is also known as tumor necrosis factor receptor superfamily 8 (TNFRSF8). CD30 is evaluated by flow cytometry in some centers, but the mainstay of phenotyping is immunohistochemistry. In addition to CD30, immunohistochemistry should also evaluate a range of T- and B-cell antigens and often cytotoxic markers. Although ALCLs are of T-cell origin, most cases show loss of several T-cell antigens or sometimes all T-cell antigens (“null” cell type). Immunohistochemistry for ALK should also be performed, as described below.
Describe the Classification of Anaplastic Large Cell Lymphoma
According to the World Health Organization (WHO) classification, ALCLs are classified based on clinical presentation (systemic or localized) and the expression of ALK.
3
ALK-positive ALCLs express ALK fusion proteins as a result of chromosomal rearrangements involving the
While systemic ALK-negative ALCL is considered an aggressive lymphoma, the WHO also recognizes 2 localized forms of ALK-negative ALCL, primary cutaneous ALCL and breast implant–associated ALCL, that typically have more indolent clinical behavior. 5,6 However, ALCL is not graded cytologically, and cases with aggressive and indolent clinical behavior may have similar cytologic features. In general, there is not a formal grading system for T-cell lymphomas. This is in distinction to B-cell lymphomas, where some entities such as follicular lymphoma have specific cytologic grading criteria, and other entities are specifically designated as high grade. Some more indolent lymphoid neoplasms carry the designation “lymphoproliferative disorder” rather than “lymphoma,” such as indolent T-cell lymphoproliferative disorder of the gastrointestinal tract.
What Additional Tests Could Be Done to Predict the Clinical Course of ALK-negative ALCL?
FISH studies to evaluate chromosomal rearrangements involving the
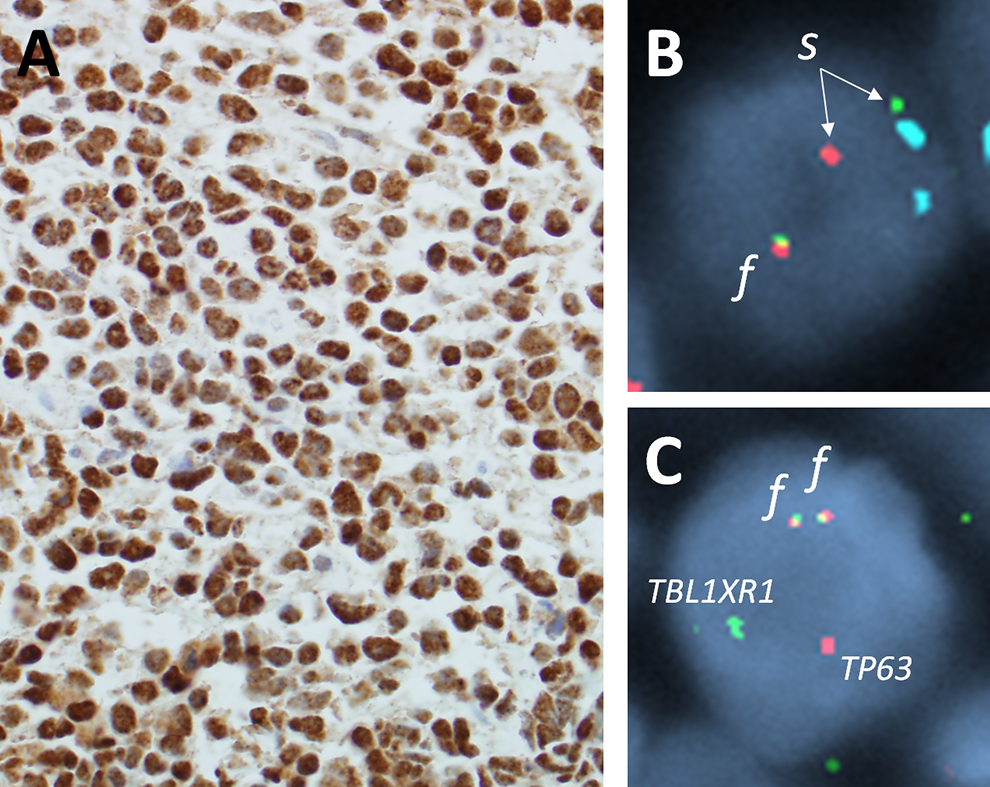
A, The tumor cells are positive for p63 by immunohistochemistry (×400). B, Interphase fluorescence in situ hybridization (FISH) using a break apart probe for the
Teaching Points
Anaplastic large cell lymphomas are malignant T-cell lymphomas characterized by the presence of distinct morphologic features, including hallmark cells and the expression of CD30; ancillary studies including immunohistochemistry as well as clinical correlation are necessary for accurate diagnosis and classification.
Anaplastic large cell lymphomas are classified by the WHO based on clinical presentation (systemic or localized) and the expression of ALK.
Systemic ALCLs include ALK-positive ALCL and ALK-negative ALCL; both tend to present at advanced stage and may involve extranodal sites.
ALK-positive ALCL occurs mostly in young patients and has a relatively good prognosis, whereas ALK-negative ALCL occurs in older adults and overall has a poorer prognosis.
Localized ALCLs include primary cutaneous ALCL and breast implant–associated ALCL.
Among ALK-negative ALCLs, those with
ALK-negative ALCLs with
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
