Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Objective
Objective TM1.2: Transfusion Reactions. Compare and contrast the pathophysiology, presentations, prophylaxis, and acute management of the different types of transfusion reactions.
Competency 3: Diagnostic Medicine and Therapeutic Pathology; Topic TM: Transfusion Medicine; Learning Goal 1: Concepts of Blood Transfusion; TM.
Patient Presentation
A 21-year-old African American female with sickle cell disease and history of multiple blood transfusions at different hospitals presents with a vaso-occlusive crisis. She has a history of hemoglobin SS (HgbSS) disease complicated by pulmonary embolism, cerebrovascular accident, and multiple episodes of acute chest syndrome. A blood sample is sent to the blood bank for workup.
Diagnostic Findings, Part 1
The patient’s red blood cells (RBCs) agglutinated with anti-B reagent and forward typed as blood group B. Also, the patient’s serum agglutinated with A1 reagent cells and backward typed as blood group B with appropriate anti-A1 isohemagglutinin. Additionally, the patient’s RBCs agglutinated strongly with anti-D reagent; thus, the patient is determined to be positive for the D antigen (RhD). The RBC antibody screen was positive. The antibody screen consists of mixing patient’s serum with 2-3 type O donor cells of predetermined phenotype cells. Further investigation revealed the presence of anti-D, anti-C, and anti-E alloantibodies. Additional panels were utilized to confirm the findings and to rule out other significant common alloantibodies. Due to discrepancy in patient’s positive RhD type and presence of anti-D antibody, more investigation is needed.
Questions/Discussion Points, Part 1
Explain Transfusion Medicine-Specific Testing
Heredity (genotype) and therefore expression (phenotype) of blood group type and certain RBC antigens is predetermined by respective genes which follow the Mendelian rules. The genotypes AO and AA express blood type A, BO and BB express blood type B, AB expresses blood type AB, and OO expresses blood type O. Each group can be either RhD positive or RhD negative, which means in total there are 8 main blood groups. 2 Pretransfusion compatibility testing begins with a blood group typing and RBC antibody screen. Blood group typing involves determining ABO blood group and Rh (D) type of recipient utilizing commercially available RBCs and antisera. Typing has 2 aspects: forward typing, a test for recipient’s antigens, and backward typing, a test for recipient’s antibodies. In forward typing, patient’s RBCs (a source for antigens) are mixed with commercial anti-ABO reagent sera (a source for antibodies), while in backward typing, patient’s serum (a source for antibodies) is mixed with commercial ABO RBCs (source for antigens). The results of the reaction in the form of agglutination or no agglutination in forward typing determine which ABO antigens are present on recipient’s RBCs. Whereas, the backward typing inspects for the presence of corresponding antibodies in recipient’s serum. Blood group A has A antigens on RBCs with anti-B antibodies in plasma. Blood group B has B antigens with anti-A antibodies in the plasma. Blood group O has no antigens but has both anti-A and anti-B antibodies in the plasma. Lastly, blood group AB has both A and B antigens but lacks antibodies in the plasma. 3 In this case, patient’s blood group is B with anti-A antibodies present in her plasma. The Rh(D) is positive. Results of blood group typing are presented in Table 1. Performing an antibody screen is the next step.
Serologic Blood Group Typing.*

* Forward blood typing is a test for ABO antigens on patient’s red blood cell using antisera (monoclonal antibodies). Backward blood typing is a test for ABO antibodies in patient’s plasma using commercial A1-cells and B-cells as sources for A1 antigens and B antigens. Anti-D is a reagent antibody. “+” for positive reactions, “0” for negative reactions, the number in front of “+” shows the reaction strength. As the number increases (1 through 4), the reaction is considered stronger. For example, “4+” has the strongest agglutination reaction. This patient forward type is group B with expected anti-A1 in plasma. She is also Rh(D) positive.
Diagnostic Findings, Part 2
Results of antibody screen and extended panel are presented in Tables 2 and 3 (respectively).
Antibody Screen.*

* Cells 1 to 3 represent 3 donors with known RBC antigen phenotypes. These cells are commercially available and used routinely in antibody screen panels. There are many RBC antigens, but the ones shown here are clinically significant and/or their antibodies are commonly identified. The Rh system includes the following antigens: D, C, c, E, e, f, Cw, and V. The antigens in Kell system are K, k, kpa, kpb, Jsa, and Jsb. Duffy group has Fya and Fyb antigens. Jka and Jkb are Kidd system antigens. Lewis group has Lea and Leb antigens. Antigens S, s, M, and N are within MNS system. P1 is the only clinically encountered in P system. Lastly, Lutheran group has Lua and Lub antigens. “+” for positive reactions, “0” for negative reactions, the number in front of “+” shows the reaction strength. As the number increases (1 through 4), the reaction is considered stronger. For example, “4+” has the strongest agglutination reaction. This patient has a positive antibody screen (2/3 cells are positive). A more extended panel with more cells is required to identify antibody(ies) specificity(ies).
Extended Antibody Panel.*
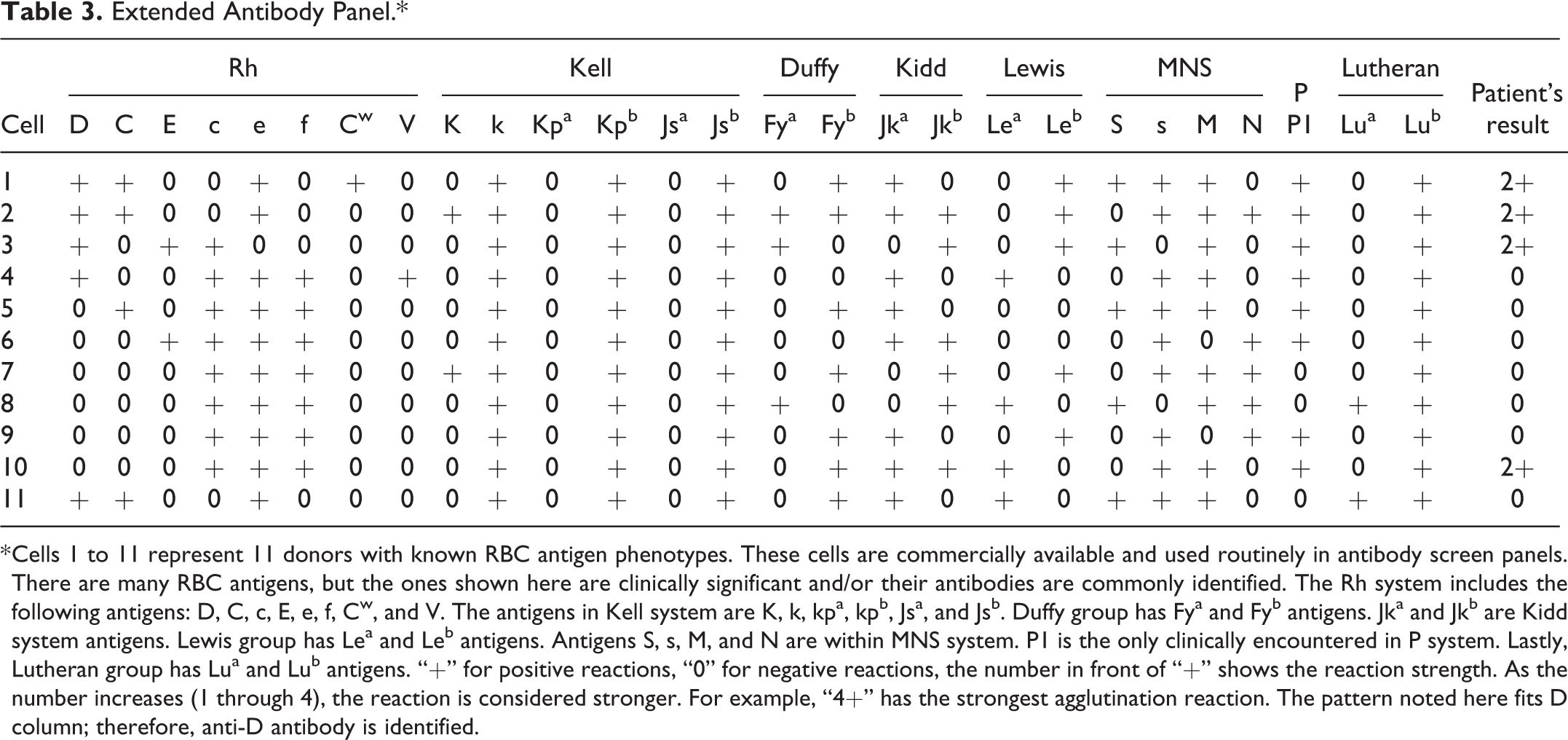
* Cells 1 to 11 represent 11 donors with known RBC antigen phenotypes. These cells are commercially available and used routinely in antibody screen panels. There are many RBC antigens, but the ones shown here are clinically significant and/or their antibodies are commonly identified. The Rh system includes the following antigens: D, C, c, E, e, f, Cw, and V. The antigens in Kell system are K, k, kpa, kpb, Jsa, and Jsb. Duffy group has Fya and Fyb antigens. Jka and Jkb are Kidd system antigens. Lewis group has Lea and Leb antigens. Antigens S, s, M, and N are within MNS system. P1 is the only clinically encountered in P system. Lastly, Lutheran group has Lua and Lub antigens. “+” for positive reactions, “0” for negative reactions, the number in front of “+” shows the reaction strength. As the number increases (1 through 4), the reaction is considered stronger. For example, “4+” has the strongest agglutination reaction. The pattern noted here fits D column; therefore, anti-D antibody is identified.
Questions/Discussion Points, Part 2
What Is the Purpose of Red Blood Cell Antibody Screening and How Is It Performed? What Is Identified in the Antibody Screen of This Patient?
The RBC antibody screening is a way to look for any antibodies already present in recipient’s plasma. A certain RBC antibody is formed when one lacks the cognate antigen. Exposure to a certain antigen that RBCs lack is recognized as foreign substance; thus, antibodies to that antigen are formed. Transmission of RBC antigens can happen through blood transfusion, pregnancy, and/or transplantation. This test includes using 2-3 type O donor RBCs of predetermined phenotype (known antigens markup) which are tested against patient’s serum (a source for antibodies). On this panel, the column headings correspond to the antigens for which each red cell has been previously tested. Each row contains the phenotype for a single red cell donor, a “+” indicates that cells have those specific antigens present, while “0” indicates the absence of certain antigens. There are more than 20 RBC antigen group systems, but only clinically significant ones are tested which include the following groups: Rh, Kell, Duffy, Kidd, Lewis, MNS, P, and Lutheran, as those antibodies can provoke hemolytic transfusion reactions and hemolytic disease of fetus and newborn (HDFN). 4 The last row shows patient’s results. The detail of the phases of the antibody identification testing is beyond the scope of this article. But to summarize, no agglutination is equivalent to a negative reaction. This is scored as a “0” and assists in ruling out the antibodies. In a positive reaction, antibodies in the patient’s plasma bind to RBCs and are scored as a “+” which prompts for further testing called extended antibody panel. In extended panel, similar to antibody screen panel but has more donor cells, 8 to 11 panel cells are tested, and based on pattern of reactivity, specific antibodies are identified. In this case, the reactivity pattern fits with an anti-D antibody; therefore, anti-D antibodies have been determined to be present in this patient’s plasma. This finding signifies a discrepancy from the positive Rh(D) noted in Rh test. Results of antibody screen and extended panel are presented in Tables 2 and 3 (respectively).
Discuss the Significance of the Rh Blood Group System. What Is Partial D?
Rh blood group system is one of the most important antigen systems after ABO. Rh antigens are expressed on RBCs only and have transmembrane protein structure. 5,6 The most clinically significant ones include D, C/c, and E/e. They are encoded by 2 genes: RHD, which encodes the presence or absence of D antigen, and RHCE, which encodes C/c and E/e. This blood group system is highly immunogenic and polymorphic. RhD has over 30 different epitopes, and in order to be RhD positive, one must express all these epitopes. The lack of part of the epitopes on D antigen leads to partial D. Thus, it is a qualitative defect in D antigen. Potentially, a person with partial D phenotype can make anti-D antibody against those missing epitopes. A blood donor with partial D phenotype should be considered to be a D positive, but a partial D recipient should always be considered as a D negative to prevent alloimmunization. 7
Why Is RhD Antigen Status So Critical in Females of Childbearing Age?
Certain RBC alloantibodies including anti-D antibody are associated with HDFN. Anti-D antibody is mostly immunoglobulin G (IgG) and can cross the placenta. If a mother is Rh negative and is carrying an Rh-positive fetus, the antibody can hemolyze fetal RBCs that bear the D antigen. It is crucial to know the RhD antigen status to decide whether a pregnant female should be offered Rh immunoglobulin (RhIG, RhoGAM) to minimize the risk of development of anti-D antibody. Nevertheless, once a female becomes immunized, she no longer qualifies for RhIG as it is of no value in such a circumstance. Still knowing the status of anti-D is important in monitoring anti-D titers during pregnancy and keeping a close eye on fetus well-being. 8 -11
What Should Be the Next Step to Resolve the RhD Type and Antibody Screen Discrepancy?
This patient is RhD positive yet she has anti-D antibody. A more through and advanced investigation must be utilized to resolve the discrepancy. There are 2 possible scenarios when someone types as RhD positive but develops anti-D along with anti-C antibodies. That should make you think about RhD variants and anti-G antibody. Several molecular variations in RhD alleles have been reported such as partial D, weak D, DEL, and their subtypes, as well as nonfunctional alleles. 12 Patient’s molecular results confirmed the absence of C and E antigens, thereby explaining the patient developing anti-C and anti-E antibodies, respectively. Not surprisingly, the patient’s RhD phenotype was determined to be a partial D with (RHD*307C) allele. This allele has been seen in association with a predicted phenotype that is positive for partial D and negative for G antigen. Little is known about RHD*307C but is seen more commonly in African Americans with partial D phenotype. 13
Is Anti-G Clinically Significant? Why Is It Important to Test for It in Pregnant Female When Suspected?
Yes. Anti-G antibody is IgG antibody that can cross the placenta and cause HDFN although less frequently and less severe than anti-D. As any other RBC alloantibody, it forms when RBCs lack the cognate antigen, in this case, G antigen. The G antigen is present whenever D and/or C are present. Anti-G may explain why a D-negative individual who was transfused with D-negative (but C-positive) red cells or D-negative woman who delivered a D-negative (but C-positive) baby subsequently appeared to have made anti-D. It is important to differentiate between anti-G reaction pattern in a panel which reacts like anti-C and anti-D and anti-D from RhD-negative or partial D phenotypes. RhD-negative and partial D phenotypic females of childbearing age, who did not develop anti-D antibody, are eligible to receive RhIg. Rh immunoglobulin consists of IGs, predominately class IgG which are derived from human plasma donors. Those donors are male donors who are RhD negative. They are stimulated by exposing them to RhD-positive RBCs in order to produce anti-D antibodies. The product is further modified to not cross the placenta and harm a fetus who could be RhD positive. The exact mechanism of action of RhIg is not well known. It is hypothesized that it works through antibody-mediated immune suppression and rapid macrophage-mediated clearance of anti-D-coated RBCs. The role of this product in clinical practice is to fool RhD-negative mother’s immune system by making it think that the anti-D antibodies have been formed. In other words, it prevents and/or minimizes the risk of producing permanent antibodies in RhD-negative mothers when exposed to RhD-positive fetuses. Rh immunoglobulin has to be given during the first pregnancy and subsequent pregnancy(ies) at 28 weeks, at delivery, and during any maternal bleeding episode. 14
Teaching Points
Always resolve a discrepant RhD type and antibody screen by more specific methods such as molecular studies to rule out RhD variants. If RhD status is discrepant, the patient should be treated as D negative until the problem is resolved. Partial D is a qualitative defect, while weak D is quantitative defect, and so partial D can make anti-D antibodies, though in general, weak D very rarely can make anti-D antibodies. It is crucial in transfusion medicine to consider all partial D recipients as D negative and donors as D positive to avoid any alloimmunization of Rh-negative individuals. Anti-D antibodies along with antibodies to Rh antigens play a significant role in HDFN. Determine if the pregnant female is a candidate for RhIG to prevent alloimmunization and minimize risk to fetus from hemolytic disease.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
