Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Learning Objective
Objective TM1.3: Infectious Risk. Discuss infectious disease risks of transfusion.
Competency 3: Diagnostic Medicine and Therapeutic Pathology; Topic TM: Transfusion Medicine; Learning Goal 1: Concepts of Blood transfusion.
Secondary Learning Objective
Objective M6.4: Malaria and Babesiosis. Contrast Plasmodium falciparum with other malaria species and babesiosis on a blood smear and explain the role of thick and thin smears in the diagnosis and management of malaria.
Competency 3: Diagnostic Medicine and Therapeutic Pathology; Topic M: Microbiology; Learning Goal 6: Parasitology.
Patient Presentation
A 76-year-old female underwent liver transplant due to nonalcoholic steatohepatitis cirrhosis. Her hospitalization was prolonged. Posttransplant she received 50 units of packed red blood cells (PRBCs), 1 unit of platelets, and 2 units of plasma (Figure 1). On hospital day 21, she had a positive direct antibody test immunoglobulin G; she was found to have developed new anti-RBC surface antigen alloantibody: anti-K.
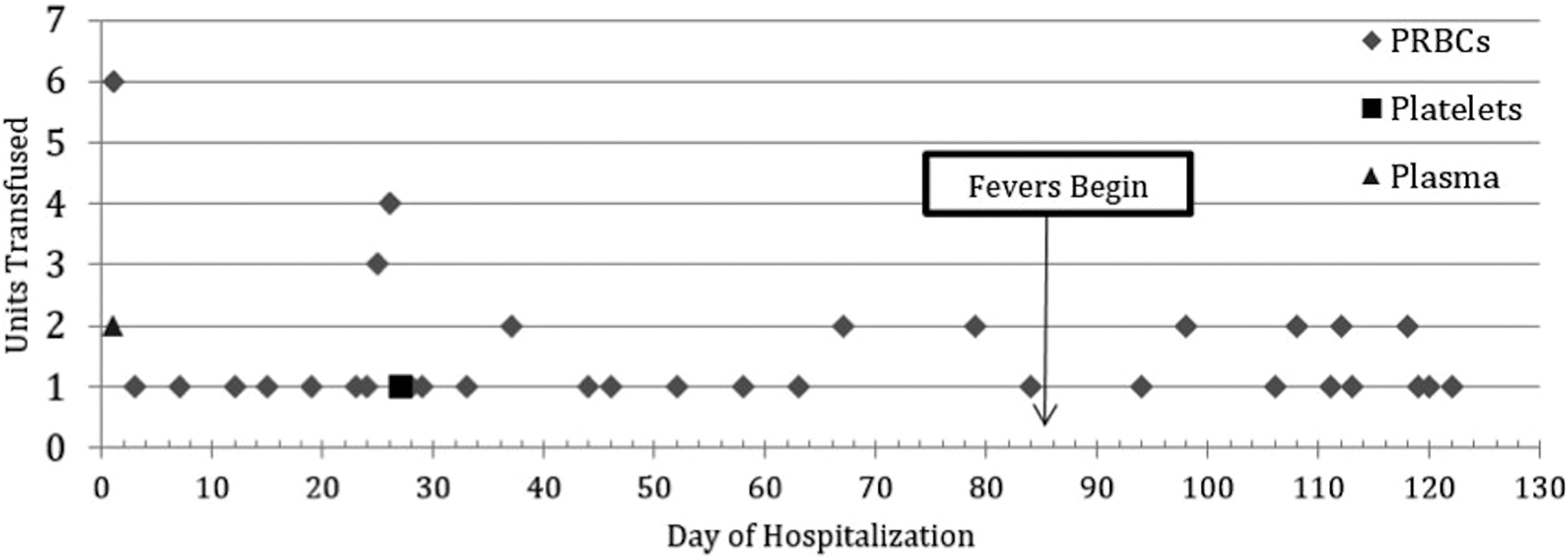
The patient received 53 blood transfusions: 50 units of PRBC, 1 unit of platelets, and 2 units of plasma. A total of 35 units of PRBCs, 1 unit of platelets, and 2 units of plasma were transfused before she became febrile. PRBC indicates packed red blood cells.
Diagnostic Findings, Part 1
Three months into her hospitalization, she developed recurrent, noncyclic fevers (up to 39 °C). Fevers persisted despite broad-spectrum antibiotic treatment. Physical examination was pertinent for new altered mental status.
Questions/Discussion Points, Part 1
Formulate a Differential Diagnosis Based on This Presentation and Propose a Plan for Further Workup
Given this presentation, the differential diagnosis included infection and transplant rejection. Infection was high on the differential because infection can cause fevers and altered mental status. Further, the patient was immunocompromised due to immunosuppression necessary after transplant. This put her at increased risk of bacterial, fungal, and viral infections. Acute transplant rejection was also high on the differential because it occurs weeks to months after transplant and can present with fever and flu-like symptoms. 2
Diagnostic Findings, Part 2
An extensive infectious disease workup was negative, including blood and spinal fluid cultures, acid-fast stains of cerebral spinal fluid, Cryptococcus antigen testing and cytomegalovirus (CMV) polymerase chain reaction (PCR). A liver biopsy was performed and displayed in Figure 2.
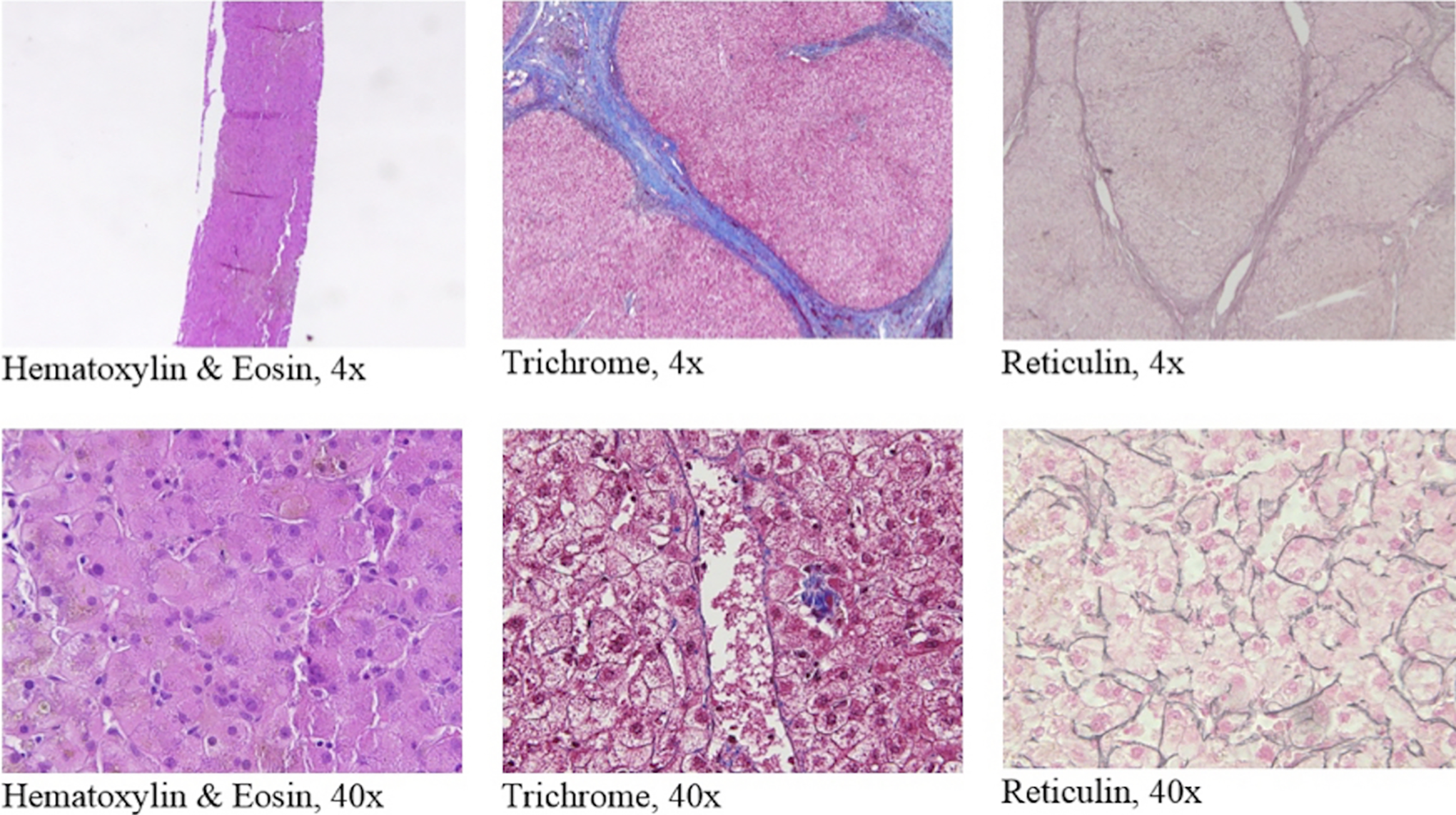
Gross microscopy of liver biopsy on hospital day 105 showed nonspecific cholestasis and no evidence of acute rejection or necrosis. Trichrome stain showed no fibrosis, confirmed by the reticulin stain. Kupffer cell hemosiderosis is present.
Questions/Discussion Points, Part 2
What Are the Findings of Acute Rejection on Liver Biopsy?
On biopsy, patients with acute rejection have bile duct lymphocytic infiltrate and necrosis due to infiltration of T-cells attacking the bile duct of the transplanted liver. 2 The patient’s biopsy demonstrated normal liver histology, including lobules and bile ducts with no lymphocytic infiltration, necrosis, or significant cholestasis (Figure 2). The biopsy was negative for acute rejection.
Diagnostic Findings, Part 3
Subsequently, the patient was found to be pancytopenic with reticulocytosis (15.3%; normal 0.5%-1.5%). She developed up-trending lactate dehydrogenase (LDH), direct and indirect bilirubin, and had undetectable haptoglobin (Figure 3). Her liver function progressively worsened, with liver function enzyme levels in the mid-100s.
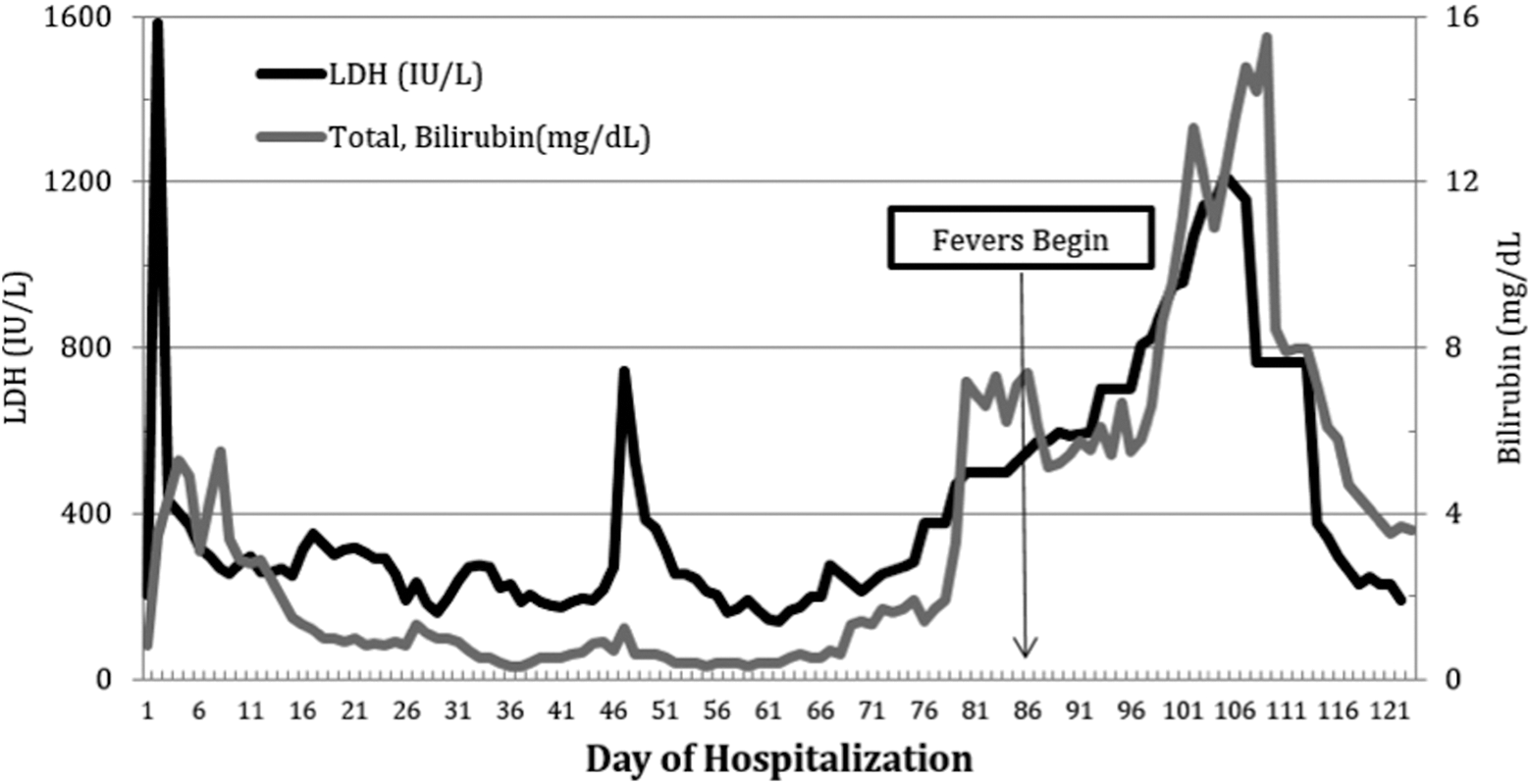
Lactate dehydrogenase (LDH) and total bilirubin levels throughout the patient’s hospitalization. There is an initial LDH spike at the time of transplant and a second elevation that temporally correlated with initiation of fevers, clinical decomposition, and additional lab aberrancies.
On hospital day 107, the patient experienced worsening mental status, hypotension (79/40 mm Hg), and an acute hemoglobin drop from 8.2 to 6.6 g/dL without an obvious source of bleeding, requiring 1 unit of PRBCs and with subsequent inappropriate posttransfusion hemoglobin response. See Table 1 for a complete set of labs.
Laboratory Values, Day 107.a
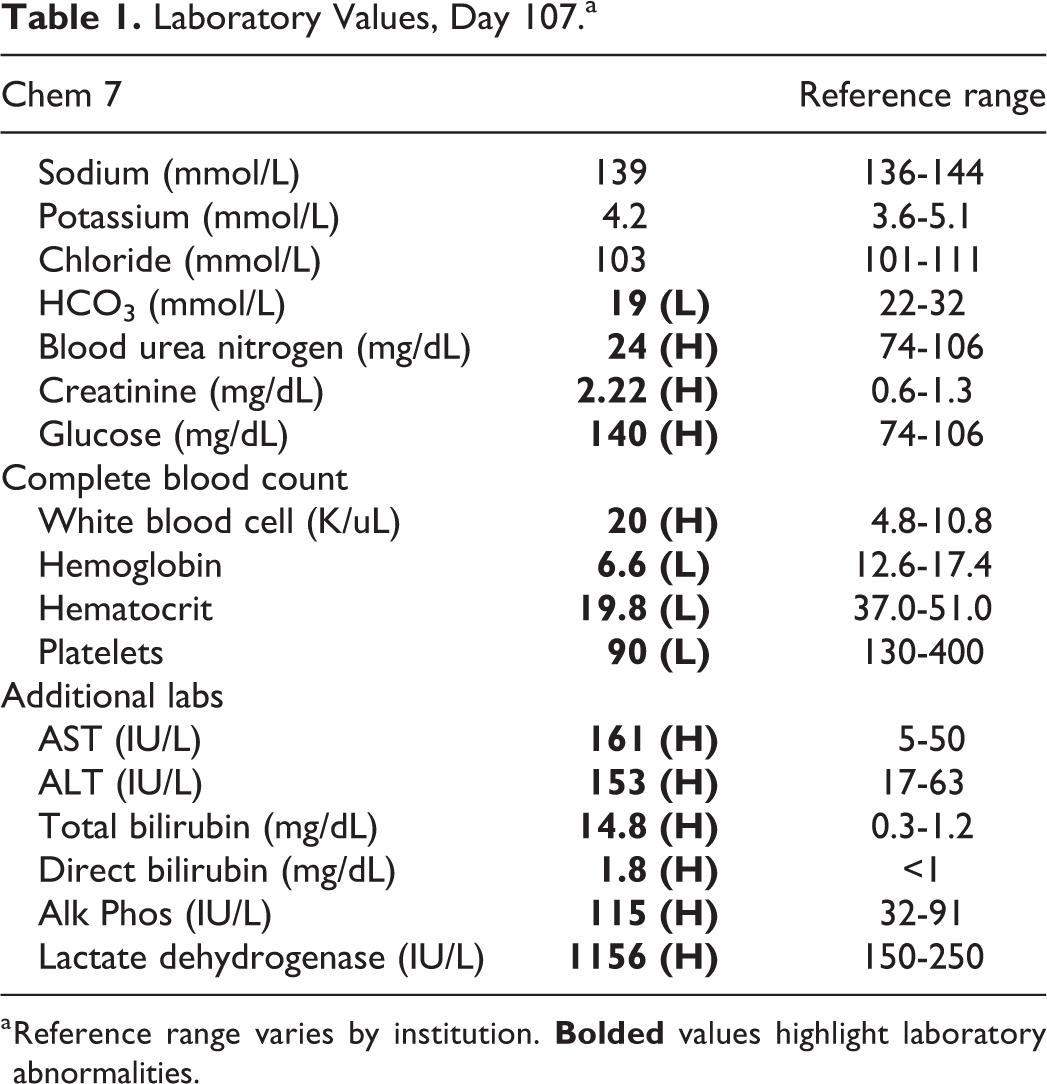
a Reference range varies by institution.
Questions/Discussion Points, Part 3
Given This Presentation and Constellation of Laboratory Findings, Can You Narrow Your Differential and Propose Possible Underlying Etiologies?
Anemia in the setting of hyperproliferative reticulocytosis (15.3%; normal 0.5%-1.5%), up-trending LDH, indirect bilirubin, and an undetectable haptoglobin, is consistent with a diagnosis of hemolytic anemia, the lysis of red blood cells. 3 Even more specifically, the elevated indirect bilirubin and low haptoglobin indicated intravascular hemolysis. 3
What Are Causes of Hemolytic Anemia in Patients Who Have Received Transfusions?
Hemolytic anemia in recently transfused patients has a wide differential including acute hemolytic transfusion reactions (AHTRs), delayed hemolytic transfusion reactions (DHTRs), and hemolytic anemia secondary to transfusion-transmitted infection (TTI). 3 -5 Acute hemolytic transfusion reactions can range from mild to life-threatening and occur within 24 hours of transfusion. 3 Acute hemolytic transfusion reactions are caused by the activation of the complement cascade in the presence of antigen: antibody complexes, leading to intravascular hemolysis. 3 Transfusion of ABO-incompatible PRBCs causes most AHTRs, estimated to occur in 1:38 000 to 1:100 000 transfusions. 3 An AHTR was unlikely in this case given the chronicity of hemolysis, and that all PRBC units were confirmed to be ABO compatible.
Delayed hemolytic transfusion reactions occur 3 to 10 days following transfusion in patients who have previously been all immunized to non-ABO RBC antigens during previous transfusions or pregnancy. 4 Patients found to have alloantibodies should be transfused with PRBCs that are antigen-negative. 4 Titers of alloantibodies may wane over time, causing some patients to have negative antibody screens. 4 In these patients, the blood bank may not be aware of the presence of the alloantibody and could issue antigen-positive PRBC units. This reexposure would result in a rapid rise in antibody titer, leading to a DHTR. 4 Delayed hemolytic transfusion reactions cause predominantly extravascular hemolysis, with milder clinical signs and symptoms than AHTRs. 3,4 In this case, a DHTR was on the differential because of the newly discovered alloantibody and clinical timeline but refuted by the presence of severe intravascular hemolysis. Additionally, the PRBC units were all negative for the Kell antigen against which she was all immunized.
Progressive hemolytic anemia in an immunosuppressed, febrile patient was highly suspicious for an infectious etiology. Because she had received so many blood transfusions during her hospitalization, a TTI was considered. Babesiosis and malaria are 2 not able protozoan TTIs that can cause hemolytic anemia. 5
What Are the Infectious Disease Risks of Transfusion?
Viruses, bacteria, protozoa, and prions can all cause TTIs. 5 Pathogens that cause TTI must be blood-borne, survive product processing, be parentally infectious, and cause morbidity in a susceptible recipient pool. 5 Transfusion-transmitted viruses include the hepatitis viruses, HIV, CMV, parvovirus, and EBV. Hepatitis A and C are each transmitted in less than 1 in 1 million red cell transfusions, while the rate of HIV transmission is 1 in 1.5 to 2 million transfusions. 5 Bacterial TTIs are due to organisms from donor blood, skin, or environmental contamination. They are estimated to occur in 0.2 per 1 million transfusions. 5 Babesiosis and malaria are infections cause by protozoa that vary widely in incidence based on geographic location. 5 Incidence of babesiosis ranges from 1/604 to 1/100 000 infections per RBC unit transfused. 6 Transfusion-transmitted malaria is rare in the United States, occurring an estimated 0 to 0.18 cases per million units transfused or less. 7 Creutzfeldt-Jakob disease can be transfusion transmitted and has been observed in a limited number of cases, albeit exceedingly rarely. 5 Risk reduction may be achieved through careful donor recruitment, questioning and testing, along with aseptic phlebotomy technique. 5 Despite a variety of test methodologies used for many TTIs, there remains a subset of TTIs for which there is no universal donor testing in the United States (Table 2). 8 Both babesiosis and malaria fall into this category. 8
FDA Requirements for Blood Donor Infectious Disease Testing.a

a Data as of 2018 FDA requirements. 8
Diagnostic Findings, Part 4
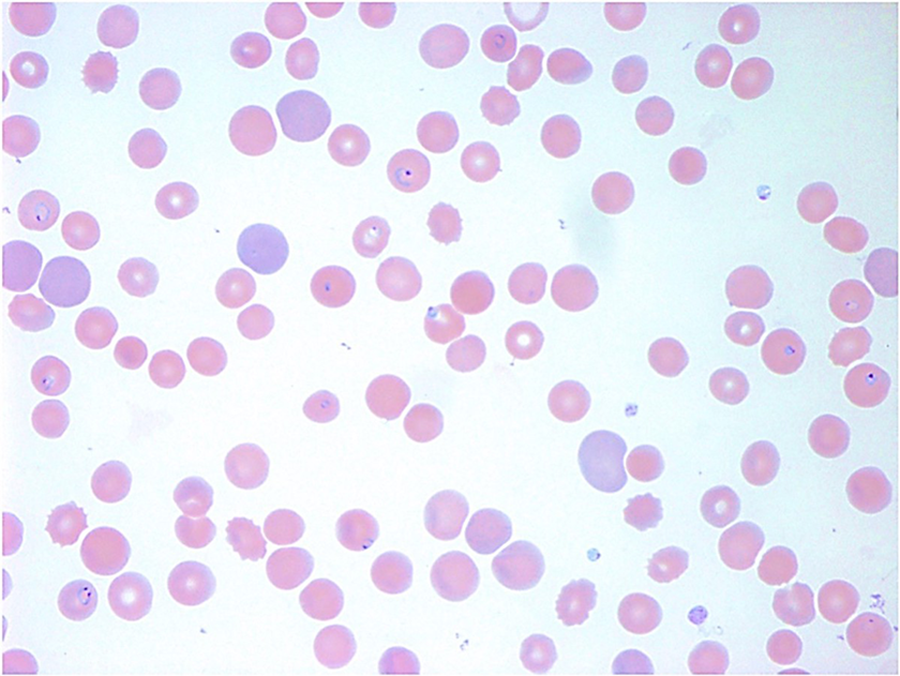
On hospital day 108, a blood smear was obtained, depicted in Figure 4.
Questions/Discussion Points, Part 4
Which Pathogens Have Ring Forms on Blood Smear and How Do You Differentiate Them?
Blood smear revealed intra- and extraerythrocytic pleomorphic and pyriform ring-shaped organisms. The parasitemia estimate was >10%, a critically high level. Babesia subspecies and Plasmodium subspecies have microscopic ring-shaped morphologies on blood smear, cause similar clinical presentations, and infect erythrocytes selectively. 9 -11
Babesia are characterized by small, pleomorphic intra-, and extraerythrocytic ring forms (Figure 4). 9,10 Maltese cross tetrads are pathognomonic for Babesia, but this form is rarely seen in human hosts. 10,12 Plasmodium, the cause of malaria, is located only within erythrocytes and is characterized by ring forms, schizonts, and gametocytes (Figure 5). 9,10 Plasmodium species can be further differentiated by the size of infected erythrocytes, parasitemia level, number of merozoites, and gametocyte type. 9 For example, the most pathogenic species, P falciparum, is associated with infection of erythrocytes in all stages of maturation and high parasitemia levels. 9
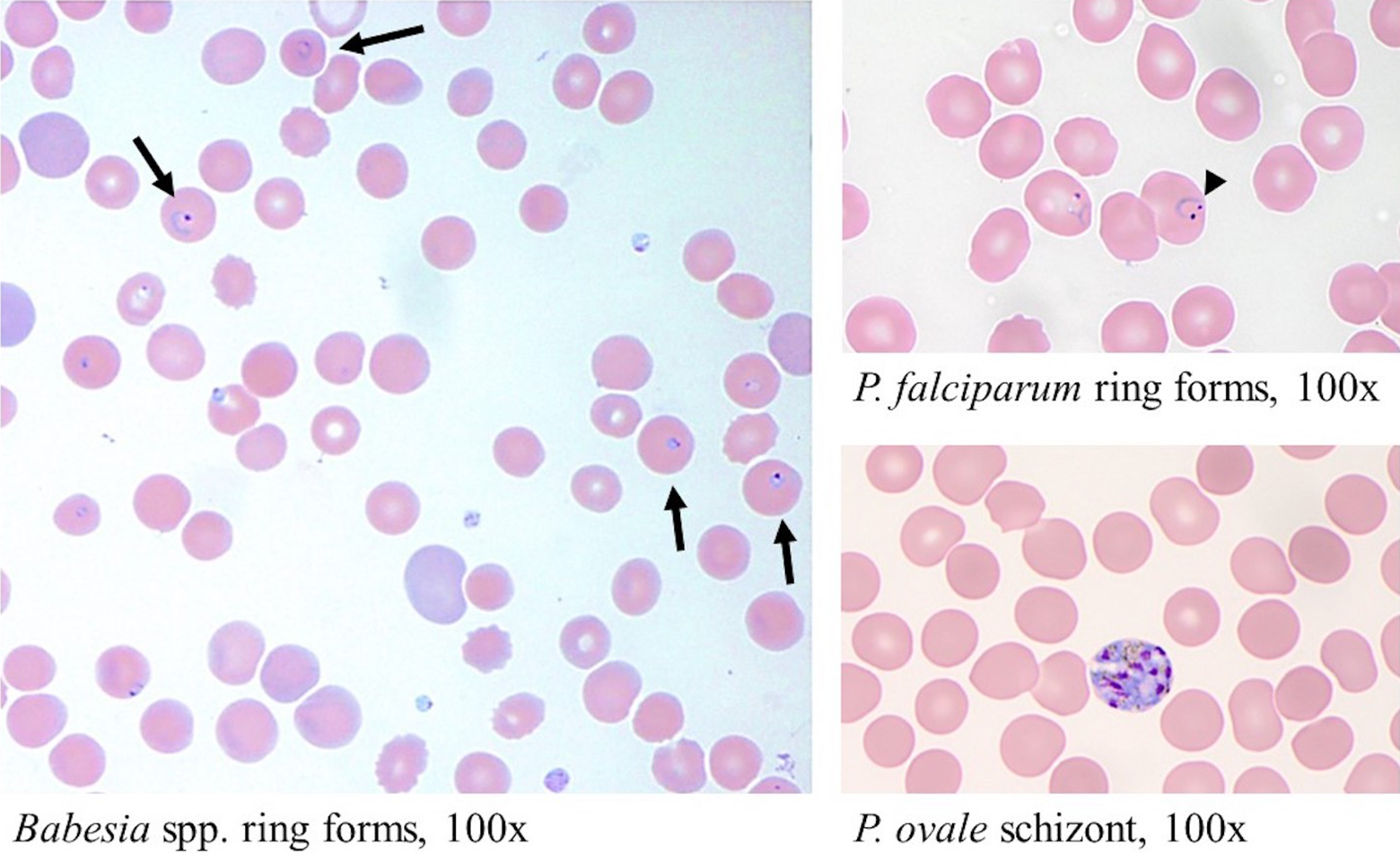
Thin blood smears of Plasmodium falciparum and Plasmodium ovale compared to Babesia subspecies Images all taken at 100×. Both Babesia subspecies and Plasmodium subspecies form ring forms. 9,10 Babesia ring forms are intracellular and extracellular, with characteristic ring morphology (arrows). 9 Plasmodium falciparum ring forms are located only within erythrocytes and are characterized by “headphone” morphology (arrow head). 9 Additionally, maturational forms of Plasmodium are often present and can be used to differentiate from Babesia, such as shown by the Plasmodium ovale schizont. 9,10 Malaria blood smear images were reproduced with permission from Dr Ashley Vogel, a hematopathology fellow at Thomas Jefferson University Hospital.
Microscopy-based identification has limitations, despite being the mainstay of diagnosis for these parasites. Thick blood smears, a drop of blood on glass, are used to establish the presence of parasites. Thin blood smears, a drop of blood spread across a large portion of the slide, are used to calculate percent parasitemia and examine morphology. 11 Only ∼60% of Babesia infections are detected on blood smear. 12 Microscopy-based diagnosis is challenging because of the small parasite size, sparse parasitemia in the majority of infections, and low index of suspicion. 12 In this case, the diagnosis was missed on several blood smears that were found to have parasitemia on retrospective review. Once recognized, smears from this patient clearly demonstrated morphology consistent with Babesia. This illustrates the difficulty of making the diagnosis from blood smears alone without prior suspicion of infection.
What Other Tests Are Used to Diagnose Babesiosis?
Babesiosis can be diagnosed by detecting Babesia DNA in serum with nucleic acid testing (NAT), which is more sensitive than thick prep blood smears. 13 Additionally, serology testing detects antibodies in serum to Babesia and indicates current or past infection. 14
Describe How Multiple Disciplines Need to Work Collaboratively to Care for a Complex Patient Such as This Patient?
Early diagnosis and treatment of babesiosis often requires efforts from across the diagnostic laboratory and clinical specialties. This multidisciplinary case required involvement of the microbiology laboratory, hematopathology, gross pathology, and transfusion medicine. Additionally, it required consultation by infectious diseases, hematology, hepatology transplant surgery, and critical care physicians. Timely communication between disciplines allowed life-saving diagnosis and treatment for this patient.
Diagnostic Findings, Part 5
Subsequent Babesia microti NAT was positive, and malaria antigen screen was negative. Given the patient’s severe clinical decomposition and parasite burden, she underwent emergent red blood cell exchange transfusion. After exchange transfusion, parasitemia declined from >10% to 3.22% (Figure 6). The patient was started on atovaquone and azithromycin. She improved clinically and parasitemia became undetectable. The patient completed 6 weeks of antimicrobials, including 2 weeks after the first negative smear. A follow-up smear 3 months later remained negative for parasites.
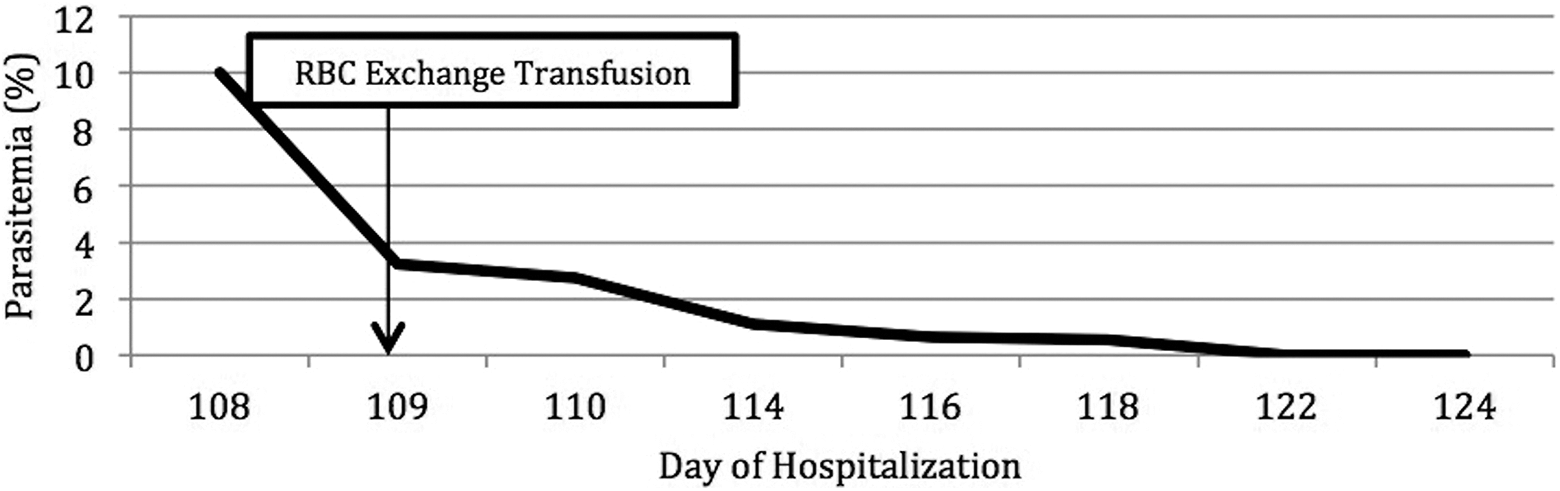
Percent parasitemia calculated from thin blood smears, obtained on the day of diagnosis (108), until 2 negative smears on days 122 and 124. RBC exchange transfusion occurred on day 109 and a subsequent smear demonstrated a dramatic parasitemia decline to 3.22%.
Questions/Discussion Points, Part 5
What Is Babesiosis?
Babesiosis is an infectious disease caused by Babesia, an intraerythrocytic protozoan. 10,15 Babesia species are endemic to the Northeast and upper Midwest regions of the United States. 10,16 Humans most often acquire infection from B microti when sporozoites are introduced through a bite from an infected Ixodes scapularis tick, but infection may also be transmitted via blood transfusion, organ transplantation, and transplacentally. 10,15 Sporozoites enter human erythrocytes where they undergo asexual reproduction and cause erythrocyte lyses. 10 Sporozoites include ring-shaped trophozoites and Maltese-cross shaped merozoites. 9,10
Infection can be asymptomatic but often causes noncyclic fevers, chills, sweats, headache, body aches, loss of appetite, nausea or fatigue, dark urine, and jaundice. 10 Symptomatic infection is termed babesiosis. 15 Babesiosis can be severe, with complications including profound hypotension, hemolytic anemia, thrombocytopenia, disseminated intravascular coagulation, multi-organ failure, and death. 10,13,15 Individuals with weakened immune systems, other serious health conditions, and the elderly have a particularly high risk of severe disease. 10
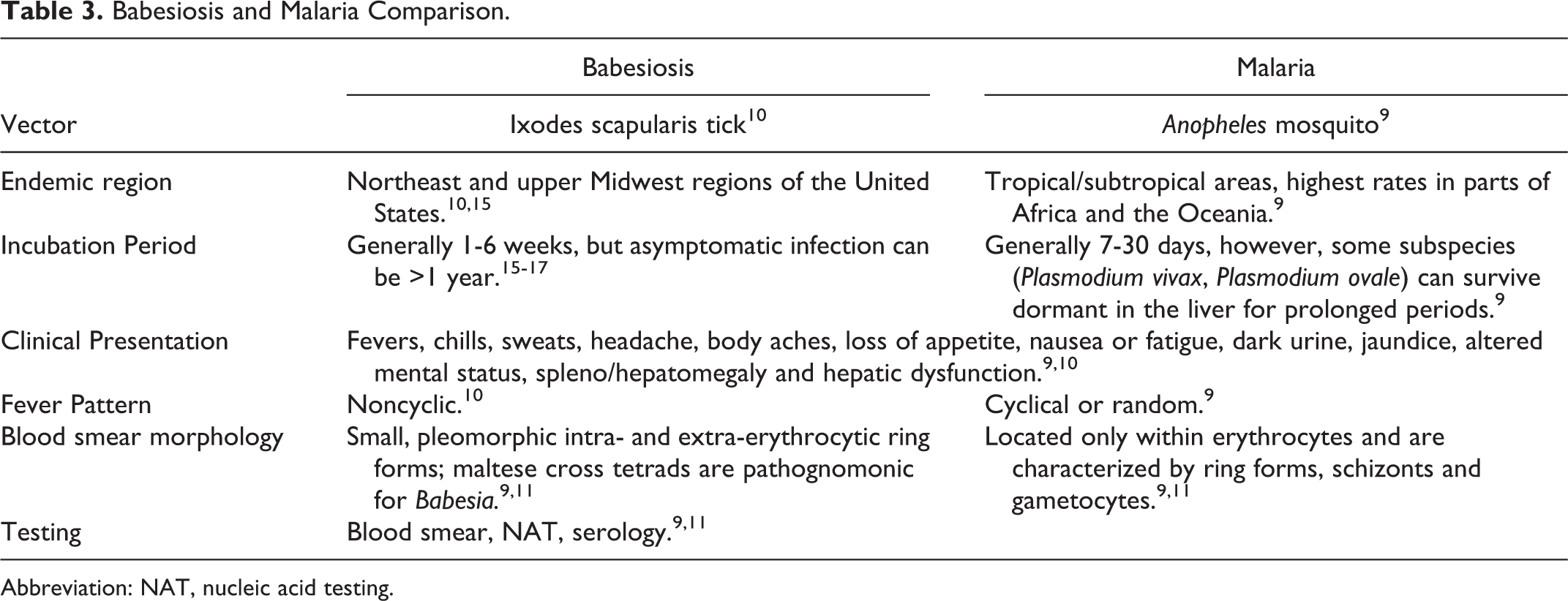
How Do the Life Cycles and Presentation of Babesiosis and Malaria Compare and Contrast?
The life cycles of Babesia subspecies and Plasmodium subspecies are similar in that they both are transmitted by vectors, have immature forms, infect red blood cells, form ring forms, and ultimately cause hemolysis leading to similarities in clinical presentation (Table 3). 9 -11,15 -17
Specifically in malaria, infected Anopheles mosquitos transmit malaria sporozoites into the blood stream. 9 Sporozoites then infect hepatocytes, where they grow and multiply into schizonts. 9 Eventually, the liver cells rupture and the parasite enters the blood stream, where it infects RBCs. In RBCs, the parasite forms trophozoites that mature into schizonts and gametocytes. 9 These forms also rupture RBCs, often cyclical pattern, leading to symptomatic malaria. 9 Gametocytes can be transmitted from an infected host to a new host via the Anopheles mosquito or via blood transfusion from an infected donor. 9
Babesiosis and Malaria Comparison.
Abbreviation: NAT, nucleic acid testing.
Like babesiosis, malaria can be characterized by symptoms of fever, chills, sweats, headache, body aches, loss of appetite, nausea, fatigue, dark urine and jaundice. 5,9 Malaria fevers, depending on the subspecies, can be cyclical or random. 5 Meanwhile in babesiosis fevers are non-cyclical. 9 The timing of fever depends on the Plasmodium subspecies. There are 5 Plasmodium subspecies, each with a slightly different fever pattern related to differences in life cycle timing. 5 P. falciparum causes random fevers, while P. knowlesi causes daily fevers. 9 Plasmodium vivax and Plasmodium ovale cause fevers every second day but can survive dormant for years in the liver. 9 Plasmodium malariae causes fever every third day. 9
How Is Babesia Transmitted?
Babesiosis is considered an emerging infectious disease. For example, in New York, more than 4600 cases of babesiosis have been reported, since 1986 with 3500 of those reported between 2006 and 2015. 18 The increased incidence may be partially attributable to increased awareness of the disease but is also due to increasing deer and tick populations along with increased human habitat in endemic areas. 13,18 Seroprevalence in endemic areas ranges from 0.5% to 1.6%, indicating a high rate of asymptomatic and self-limited infection in the general population. 19
The first known United States case of transfusion-transmitted babesiosis (TTB) was described in 1979, and now >200 cases have been identified. 15 The incidence of TTB is estimated to be 1.1 cases per million of red blood cell units distributed. 20 Most cases of TTB occur when tick-acquired infection is greatest, June to November. However, infection can occur at any time because asymptomatic infection can last >1 year and the incubation period after a transfusion is generally 1 to 6 weeks but has been documented up to 6 months. 15 -17 Transfusion-transmitted babesiosis is not limited to endemic areas because of blood donor travel and blood product transfer across geographic regions. Most TTB is acquired through PRBCs; however, there have been a few cases of TTB acquired from the small amount of residual erythrocytes in platelet units. 16
Post-solid organ transplant, Babesia infection is rare, and all known cases have been related to transfusions that occurred in the peri-transplant period. 21 To date, there are no known cases of Babesia transmitted via liver transplant documented in the literature.
How Did This Patient Acquire Babesiosis?
The source of Babesia infection in this case was likely transfusion related; however, it could have also been from the transplanted liver or reactivation of a previous asymptomatic infection. Before the patient developed fevers, she had received 35 units of PRBCs, 1 unit of platelets, and 2 units of plasma (Figure 1). Infection from the transplanted liver is unlikely given the lack of evidence of solid organ transplant-transmitted infection in the literature. Further, if the infection had been from an infected donor liver, a symptomatic infection would have most likely occurred within 6 weeks posttransplant during the typical incubation time, not 3 months posttransplant, given that the incubation time is about 6 weeks. 16 Because she became symptomatic greater than 6 weeks into her hospitalization, a recently acquired tick-borne infection in the patient herself was also unlikely.
What Are Treatment Guidelines for Babesiosis?
Treatment of babesiosis depends on the severity of infection. 22 Asymptomatic patients should not receive antimicrobial therapy and should only be observed. 10 Immunocompetent patients with <4% parasitemia should be prescribed a 7- to 10-day course of azithromycin and atovaquone. 9,22 Patients with >4% parasitemia should be hospitalized, receive antimicrobials, and are candidates for RBC exchange transfusion. 10,23 Immunocompromised patients are at high risk of relapse. 9,21 In these patients, antimicrobial therapy should be prescribed for a minimum of 6 weeks and continued 2 weeks past a negative blood smear. 10,23
What Is Apheresis and What Are Indications for It?
Therapeutic apheresis describes a procedure in which a centrifuge separates blood to remove one or more components from a patient. 24 Therapeutic plasma exchange is performed for many patients with immune-mediated diseases including a spectrum of infections, autoimmune disease, and hematology oncology disorders. 25 Removed plasma that is discarded and 5% albumin or plasma products are used as replacement fluid. 25
Erythrocytapheresis, or RBC exchange transfusion, is an apheresis procedure in which patient erythrocytes are removed and replaced with healthy donor PRBC units. 24 In the United States, the vast majority of RBC exchange transfusions are performed in patients with sickle cell disease for primary or secondary stroke prophylaxis. 24 However, this procedure is considered first-line therapy for patients with severe babesiosis. 23,25 Cerebral malaria is sometimes treated with RBC exchange; this diagnosis is quite rare in the United States. 25
How Does Apheresis Treat Babesiosis?
Red blood cell exchange transfusion in severe babesiosis can prevent death from multi-organ dysfunction and is indicated if parasitemia is >10%, hemoglobin is <10 g/dL, or if there is evidence of pulmonary, liver, or renal impairment. 25 In this case, the patient met all criteria for exchange transfusion. Exchange transfusion is effective treatment because it rapidly decreases parasitemia, corrects anemia, and removes circulating toxic byproducts of cell lyses and inflammatory mediators. 23,6 Response to treatment is achieved by tracking parasitemia on peripheral smears. 23
How Can TTB Be Prevented?
As demonstrated by this case, TTB can be severe and life-threatening. Pathogen inactivation technology has been Food and Drug Administration (FDA) approved for some types of platelet units. 26 This technology prevents replication of any genetic material present in the bag, representing an agnostic approach to eliminating transmission of any identified or unidentified pathogen that may be present. 26 However, the inactivation technology cannot be used for PRBC units at this time, leaving Babesia screening at the forefront of TTB prevention. 27
Despite the ability to screen for the parasite, historically, requirements only included asking blood donors if they ever had babesiosis. 28 Studies in endemic areas have shown that NAT and serology testing significantly reduce TTB, with one study estimating a reduction of 3.39 cases/100 000 PRBC transfusions. 29 However, this decrease comes at increased cost. Endemic area screening costs on average US$760 000/quality of life adjusted year. 30 Despite the cost associated with testing, prevention is paramount given the increased incidence and severity of TTB.
In May 2019, the FDA published updated recommendations regarding Babesia, with an intent to require blood collection centers to be in compliance by May 2020. 31 These recommendations include year-round screening for Babesia with NAT for all blood donors in 15 Babesia endemic states, unless pathogen reduction is performed. 31 Blood collection facilities outside of Babesia endemic states are not required to test for the parasite but must ask all donors if they have ever tested positive for Babesia. 31 All donors with a positive NAT must be deferred for a minimum of 2 years after the last positive test. 31
Teaching Points
The safety of transfusions has improved greatly over time; however, TTI persists as a transfusion risk. Viruses, bacteria, protozoa, and prions can all cause TTIs. Transfusion-transmitted viruses include the hepatitis viruses, HIV, CMV, parvovirus, and EBV. 5 Babesiosis and malaria are protozoa transmitted infections that very in incidence based on location. 5 Incidence of babesiosis ranges from 1/604 to 1/100 000 infections per RBC unit transfused. 6 Transfusion-transmitted malaria is rare in the United States, occurring an estimated 0 to 0.18 cases per million units transfused or less. 7
TTI risk reduction can be achieved through careful donor recruitment, questioning and testing, along with aseptic phlebotomy technique. Despite a variety of test methodologies used for many TTIs, there no universal donor testing in the United States for babesiosis and malaria.
Babesiosis and malaria have similarities in presentation and both should be in the differential of a patient with fevers, hemolytic anemia, and history of transfusion. These parasites can be differentiated by their life cycles and morphology on peripheral blood smear.
Microscopy-based identification is used to diagnosis babesiosis and malaria. Thick blood smears, a drop of blood on glass, establish the presence of parasites. Thin blood smears, a drop of blood spread across a large portion of a slide, are used to calculate percent parasitemia and examine morphology. 11
Babesiosis is an infectious disease caused by the protozoan Babesia. 10,15 Babesia species are endemic to the Northeast and upper Midwest regions of the United States and are transmitted by the I scapularis tick. 10,15,16 Babesia sporozoites enter human erythrocytes where they undergo asexual reproduction and cause erythrocyte lyses. 10 Immature forms include intra- and extracellular ring-shaped trophozoites as well as Maltese-cross shaped merozoites, all of which can be observed on blood smear. 9,10
Malaria is an infectious disease caused by the protozoan Plasmodium, transferred to humans by the Anopheles mosquito vector. 9 The parasite infects hepatocytes and erythrocytes. Malaria fevers, depending on the subspecies, can be cyclical or random and depend on RBC rupture. 5 In RBCs, the parasite forms trophozoites that mature into schizonts and gametocytes. 9 Schizonts are unique to Malaria and can be found on blood smear. Plasmodium ring forms, also observed on blood smear, are strictly intracellular and have classic “headphone morphology.”
Therapeutic apheresis describes a procedure in which a centrifuge separates blood to remove one or more components from a patient. 24 Therapeutic plasma exchange is performed for a wide range of patients. Red blood cell exchange transfusion in severe babesiosis can prevent death from multi-organ dysfunction.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dykes was a fourth year medical student at Sidney Kimmel Medical College, Thomas Jefferson University when this case was initially submitted, now she is an internal medicine intern at Georgetown University. Peedin is an assistant professor in the department of Pathology, Anatomy, and Cell Biology at Thomas Jefferson University Hospital.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article processing fee for this article was funded by an Open Access Award given by the Society of ‘67, which supports the mission of the Association of Pathology Chairs to produce the next generation of outstanding investigators and educational scholars in the field of pathology. This award helps to promote the publication of high-quality original scholarship in Academic Pathology by authors at an early stage of academic development.
