Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Objective
Objective GT1.1: Congenital Disorders of the Gut. Discuss the clinicopathological features of tracheoesophageal fistula, pyloric stenosis, intestinal atresia, Meckel diverticulum, and Hirschsprung disease.
Competency 2: Organ System Pathology; Topic: (GT) Gastrointestinal Tract; Learning Goal 1: Embryology of the Gut.
Secondary Objective
Objective AU1.1: Value of the Autopsy. Provide examples demonstrating the value of the autopsy toward improvement in clinical diagnosis and management, quality control, medical education, research, and elucidation of “new” diseases.
Competency 3: Diagnostic Medicine and Therapeutic Pathology; Topic: (AU) Autopsy; Learning Goal 1: Value of the Autopsy and Obtaining Consent.
Patient Presentation
A 25-year-old G1P0 female, who is at 36 weeks’ gestation, presents to the obstetrics clinic for a routine prenatal appointment. She has been followed by her obstetrician regularly and hitherto the pregnancy course has been uncomplicated. During the current visit, however, fetal ultrasonography shows that the amniotic fluid index (AFI) is 32 cm with a maximal vertical pocket > 11 cm (normal AFI range: <24 cm or maximal vertical pocket < 8 cm). Additionally, dilated loops of bowel associated with increased peristalsis are also noted during the examination (Figure 1). The patient is then transferred to the hospital Labor and Delivery (L&D) department for further evaluation and close monitoring.
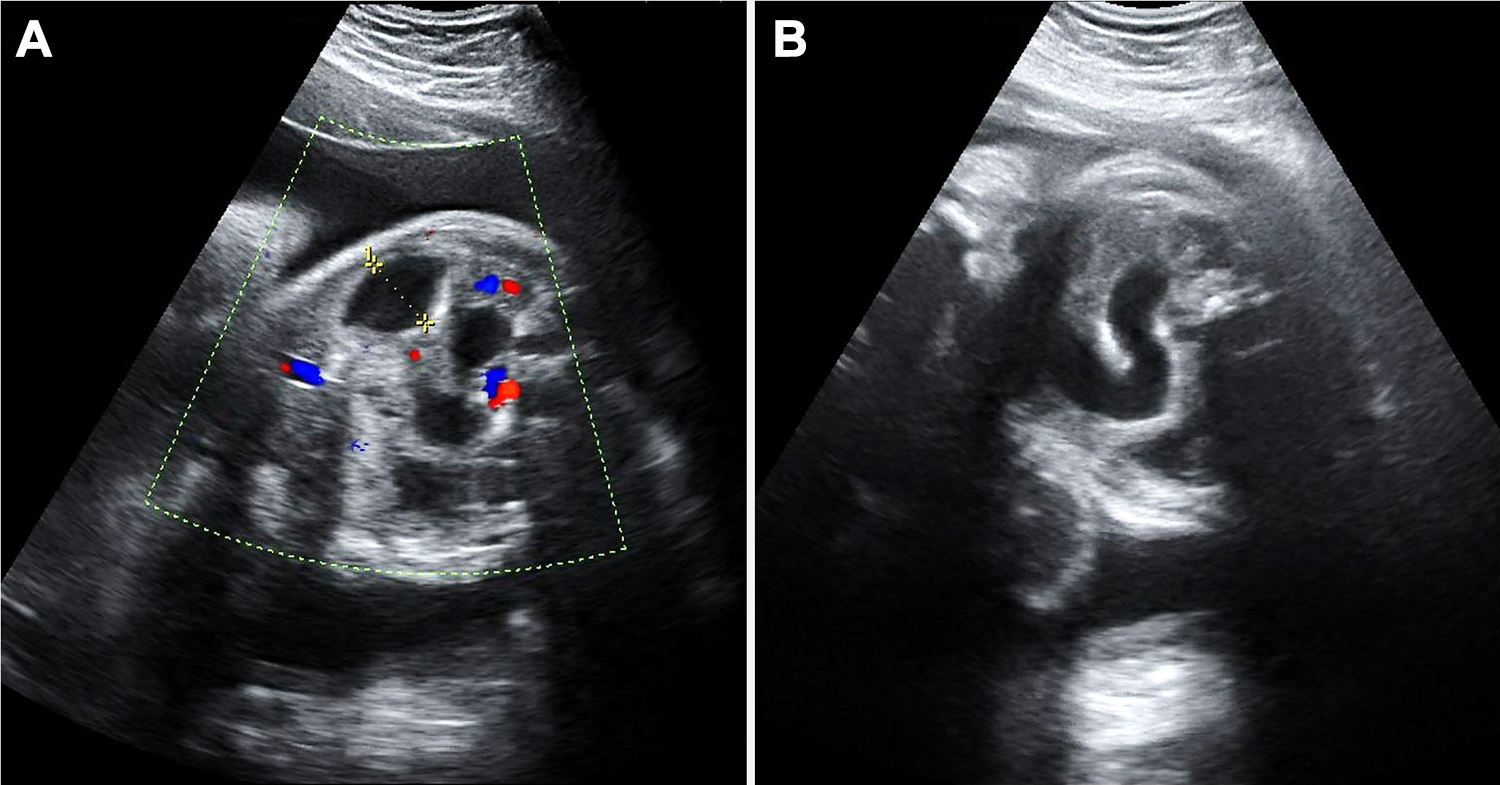
The ultrasonogram of the 36-week fetus. A clear, echogenic, dilated bowel loop is observed within the abdominal cavity (A), which is highly suggestive of intestinal obstruction. Additionally, dilated segment of the small bowel is also observed at different plane (B). The maximal dilation is measured (marked by the dotted line) to be 1.9 cm. It is also noted that abnormal peristalsis of the intestine is detected but no other overt congenital anomaly is identified during the examination.
Diagnostic Findings: Part 1
Numerous tests are performed during the patient’s stay in the L&D department. Her vital signs are stable, but her blood pressure is slightly elevated (146/85 mm Hg). Serum chemistry tests reveal that all electrolyte concentrations are within the reference range. Liver function tests are within the reference range and serum total bilirubin is not elevated. Complete blood count reveals slight anemia (hemoglobin 11.9 g/dL) but is otherwise unremarkable. Qualitative urinalysis shows decreased specific gravity and is negative for protein, glucose, blood, bilirubin, nitrites, and leukocyte esterase.
Repeated ultrasonography confirms polyhydramnios, dilated bowel segments with increased abnormal peristalsis (Figure 1), and decreased abdominal circumference (7th percentile). Biophysical profile of the fetus is all normal and scores 8/8. It is then decided that corticosteroid injection will be administered and outpatient follow-up with the maternal-fetal medicine specialist is scheduled for 2 days later.
Questions/Discussion: Part 1
What Are the Reasons for Sending the Mother to the Hospital?
The presence of dilated bowel is very concerning as it is highly suggestive of intestinal obstruction, resulting in the distension of the proximal bowel. There are multiple reasons, functional and mechanical, why fetal bowel obstruction occurs, such as congenital disorder, intestinal malrotation/volvulus, and even meconium plug syndrome. 2,3 Identifying the etiology, nonetheless, remains difficult due to the limited accuracy of ultrasound imaging. As such, this requires prompt attention from the neonatal specialists as well as pediatric surgeons and careful work-up is warranted in order to devise a proper treatment.
What Is the Significance of the Elevated Amniotic Fluid Index?
Amniotic fluid index is an indirect measurement, often by ultrasonography, of the volume of the amniotic fluid within the amniotic sac. While there is the steady production of amniotic fluid by the mother, the volume is maintained in a homeostasis as the fetus constantly swallows and digests the fluid. An increase in AFI is indicative of polyhydramnios and such findings should be addressed and evaluated to identify the underlying etiology. Although majority of cases of polyhydramnios are idiopathic, in this case, the intestinal obstruction is highly suspected to cause the elevated AFI. Furthermore, studies have shown that the severity of polyhydramnios directly correlates with the risk of prenatal abnormality as well as antepartum complications, such as the increased risk for premature delivery. 4 Considering these findings, intramuscular corticosteroid administration is given in anticipation of a possible preterm delivery.
Why Is Corticosteroid Given to the Mother?
Corticosteroids such as betamethasone and dexamethasone aid fetal lung maturation, and extensive studies have shown the benefits and favorable clinical outcomes when antenatal corticosteroids are administered to mothers who have a high risk of preterm labor. 5 This practice is endorsed by various professional societies, such as The American College of Obstetricians and Gynecologists and now it is considered as the standard of care. 6
Diagnostic Findings: Part 2
The 25-year-old mother, now at 36-week and 2-day gestation, comes to the maternal-fetal medicine center for assessment and evaluation. The mother denies any significant events occurring over the past 2 days, but she is anxious to know what is happening. During the checkup, however, it is found that there is an absence of fetal cardiac activity as well as loss of muscle tone; the diagnosis of intrauterine fetal demise (IUFD) is made. The heartbroken patient is transferred to the L&D department again for induction of labor. The delivery process is uneventful and yields a stillborn female fetus with attached, intact umbilical cord and placenta. Of note, the amniotic fluid is brown-green in color. In addition to performing genetic studies on the placental tissue, a full fetal autopsy examination is requested by the parents.
Questions/Discussion: Part 2
What May Be the Cause of Intrauterine Fetal Demise Based on the Clinical Findings?
Extensive investigation is done to identify the etiology of the IUFD. While many IUFDs have no identifiable cause, infection, genetic abnormalities, and intrauterine growth restriction are among the most common etiologies. 7
The 3 most important findings related to the cause of death, in this case, are: (1) the dilated bowel, (2) the presence of the brown-green stain in the amniotic fluid, and (3) the polyhydramnios. It is still uncertain at this point if these 3 findings are associated or independent processes. One can speculate that the brown-green stain in the amniotic fluid is meconium, which subsequently leads to meconium aspiration in utero. Alternatively, it is plausible that there is an ascending infection from the birth canal causing chorioamnionitis and fetal death. Lastly, one can always argue that the dilated bowel may be a part of syndromic defects, such as Down syndrome, and the cause of death is due to a cardiac anomaly, which is unrelated to the clinical picture. All in all, a postmortem examination will be helpful in providing new information and hopefully, the cause of death can be identified.
Diagnostic Finding: Part 3
After proper consent form has been obtained from the parents, a full autopsy with the placental examination is performed. Brain and spinal cord are excluded in accordance with the parents’ wishes.
External Examination
Pertinent findings from external examination include a female fetus that is slightly underweight (2100 g; expected 2454 ± 294 g) with a slightly short crown-heel length (43.5 cm; expected: 47.5 ± 3.4 cm). Development of facial features, thorax, and extremities appear appropriate for the gestational age and anatomic defects are not identified. Of note, the abdominal circumference measures only 23.5 cm (expected 26.8 ± 2.9 cm), while the remaining body measurements, including the head and chest circumferences, shoulder width, and extremity length, are all within the reference range of a 36-week fetus.
Internal Examination
The most remarkable finding from the organ system evaluation is that there is a severe gastrointestinal defect. The duodenum is disconnected from the jejunum and ends in a pouch-like structure. Both the proximal duodenum and the stomach are markedly distended and contain dark brown-green, finely granular and mucoid material. The distal small intestine is significantly shortened and coiled around a fibrous band, in an “apple-peel” fashion. Although aberrantly formed, the distal intestine is still patent and connects to the large intestine, which has a normal configuration and connects to the anus (Figure 2). The remainder of the examination is grossly unimpressive; all the internal organs are in their usual anatomic positions, with appropriate size and maturation. The fetal cardiovascular formation is anatomically correct, with open foramen ovale and patent ductus arteriosus.
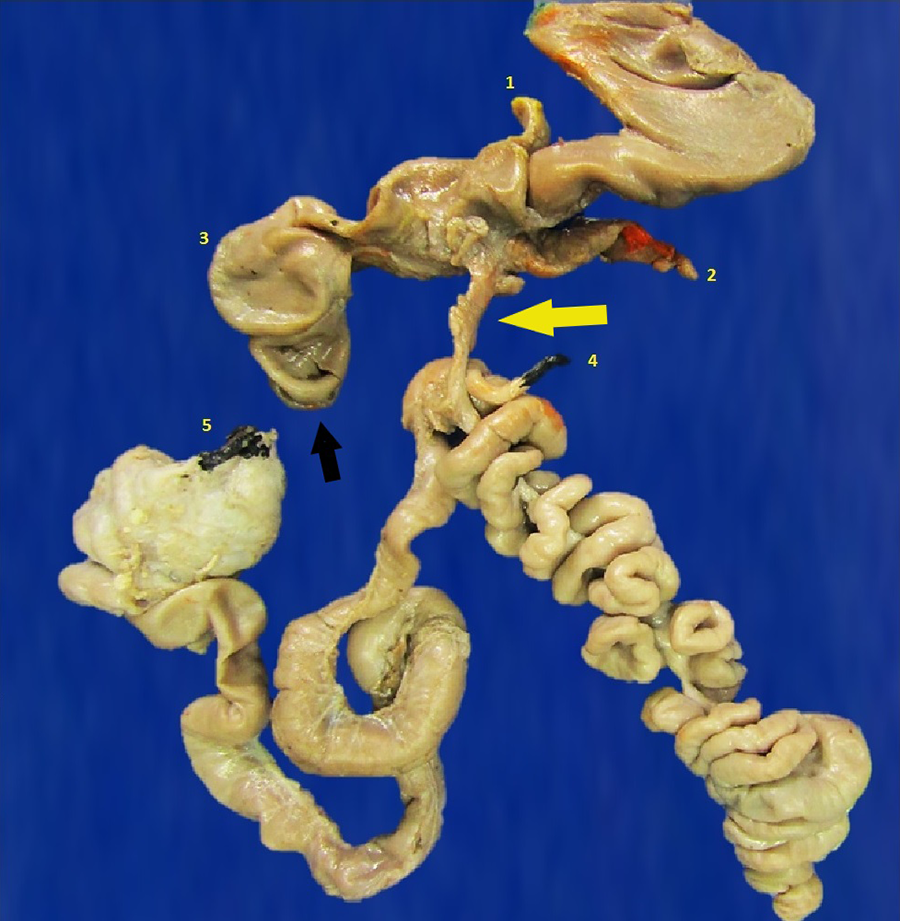
Fetal gastrointestinal system. This picture excellently illustrates the classic “apple-peel” configuration of jejunoileal atresia, type IIIb (see later text and Figure 6). There is a clear separation between the bowel, leading to marked dilatation of the proximal gastrointestinal tract and the coiling of the distal bowel around the fibrovascular core (yellow arrow). The structures in the diagram are labeled as follows: (1) gallbladder, (2) pancreas, (3) distal duodenum/proximal jejunum, (4) appendix, and (5) rectum. The black arrow points to the proximal end of atretic intestine.
Placental Examination
There is a singleton placenta with an eccentrically attached cord. The cord is intact, with appropriate length, and contains 3 vessels within. The weight and size of the placenta are appropriate for the gestational age and there are no obvious deformities. The amniotic membrane is intact and translucent, and its surface is slightly gray-green. The maternal surface is well-formed, and minimal blood clots are identified.
Histology/Microscopic Examination
Small sections of tissues are taken from the various organs, such as the heart, lungs, liver, intestines, and so on for histologic evaluation. As a resultant of IUFD, most of the tissues obtained are moderately autolyzed (degenerated). Nonetheless, sections of the lung parenchyma demonstrate that there are widely dispersed, golden brown, radiant crystalline bodies throughout the alveolar spaces (Figure 3). Histologic examination of the “blind pouch” shows moderately autolyzed mucosa that is stained with brown-green pigments (Figure 4). Brunner’s glands are absent from submucosa, suggesting that this segment is not part of the duodenum (Figure 4). Furthermore, sections of the fibrous cord surrounded by the intestine reveal it to be a tortuous blood vessel (Figure 5). The remaining tissue sections from other organs are all unremarkable and without any significant histopathologic findings. Examination of the placental tissue is unremarkable. The umbilical cord is trivascular and is not inflamed. Decidual vasculopathy and acute inflammation are not observed. There is also no chorioamnionitis.
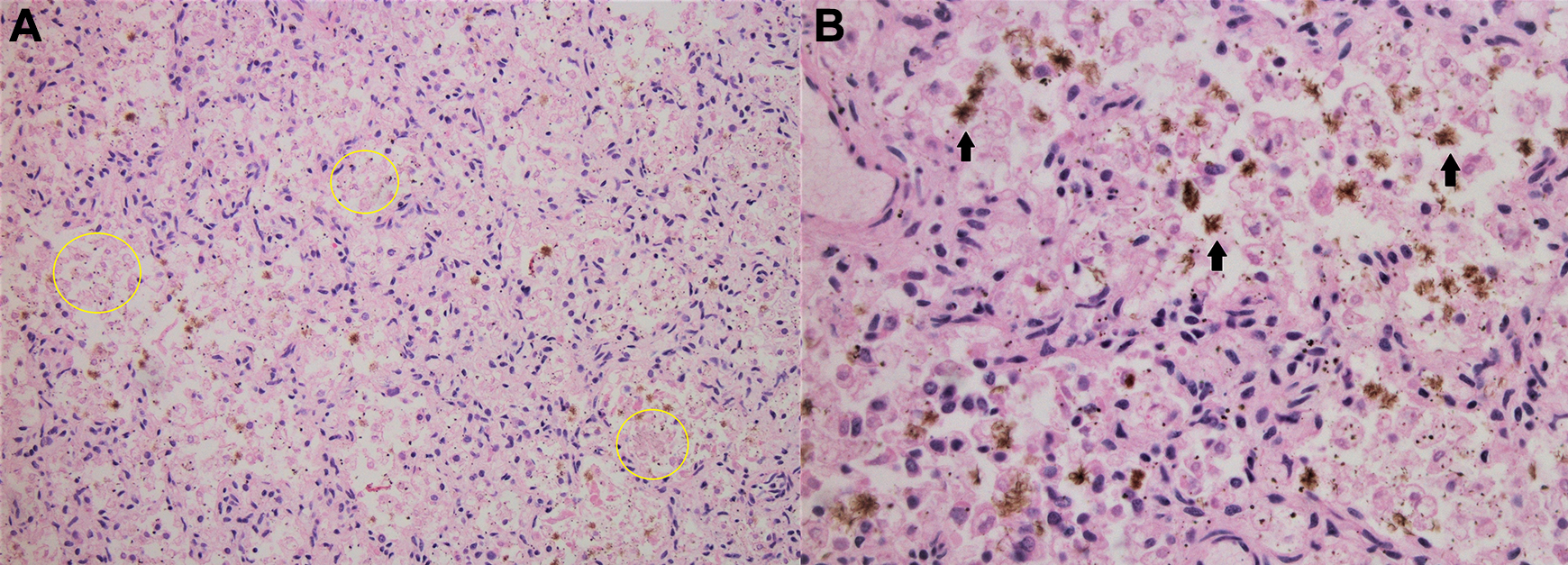
Representative sections from the lung (A-B). The pulmonary parenchyma shows airspaces rimmed by mildly autolyzed alveolar septa (yellow circle). One can appreciate that there are abundant brown-green granular bodies finely dispersed within the alveolar spaces formed by the maturing pneumocytes (A). High magnification of the brown-green granules (black arrow; B). It is more plausible that these pigments come from the bilious content of the upper gastrointestinal tract rather than meconium aspiration because the latter tends to be a conglomeration of eosinophilic granular materials often accompanied with degenerative squamous cells from the fetal skin as well as acute inflammation.
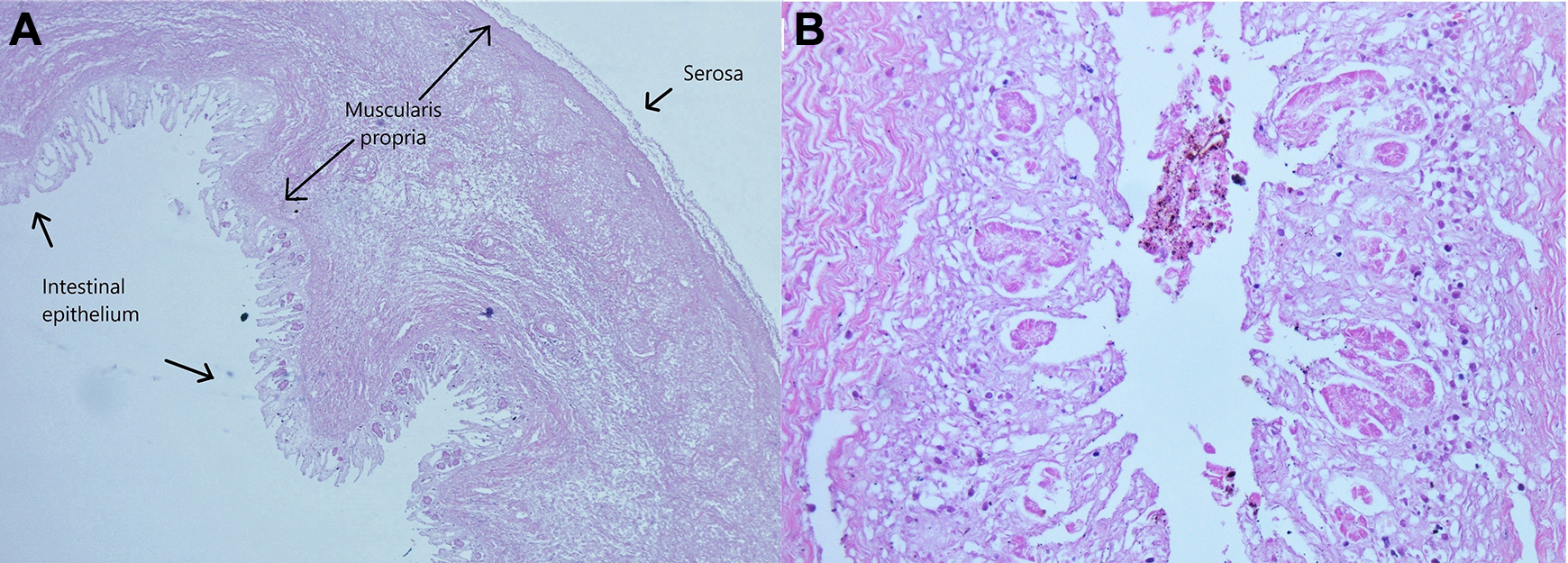
Representative sections from the intestinal “blind pouch” (A-B). This micrograph shows a moderately autolyzed segment of bowel (A) with sparse granular pigments found on the mucosal surface (B). These deposits are believed to be bile pigments drained from the biliary tracts. Note that Brunner’s glands are not found within the submucosa of the intestinal lining, suggesting that this pouch-like structure is not duodenum.
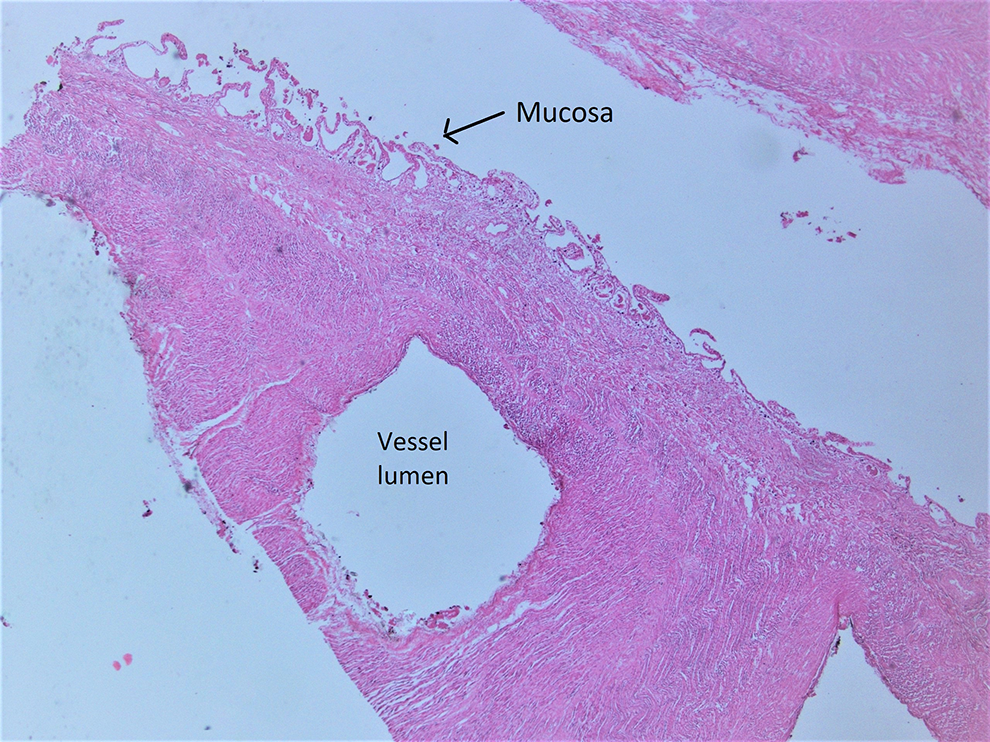
This is the longitudinal section of the fibrovascular cord encircled by the distal small intestine. It consists of tortuous blood vessels, resulting in the “donut” configuration when sectioned longitudinally. Note that the outer surface of the blood vessel is coated with autolyzed intestinal mucosa (arrow), indicating that the small bowel is fused with the blood vessels for direct nutritional supply. This is a hallmark feature of JIA, type IIIb and differentiates this entity from intestinal strangulation. JIA indicates jejunoileal atresia.
DNA Microarray Testing Result
Placental tissue is submitted for single nucleotide polymorphism microarray analysis. The array profile is consistent with a normal female genomic profile with no diagnostic copy number changes.
Questions/Discussion: Part 3
What Is the Diagnosis of the Gastrointestinal Anomaly?
Although one may be tempted to establish the diagnosis of duodenal atresia, the proper term for the observed anomaly is jejunoileal atresia (JIA), 8 since it is involving the proximal jejunum. Particularly, this case is categorized as the type IIIb subtype of JIA (Figure 6 and Table 1). 9 As the name “apple peel” suggests, this term applies when there is a malformation of the small bowel, leading to parts of the jejunum or ileum being obstructed or atretic, with the distal bowel coiling around the remaining intact blood vessel.
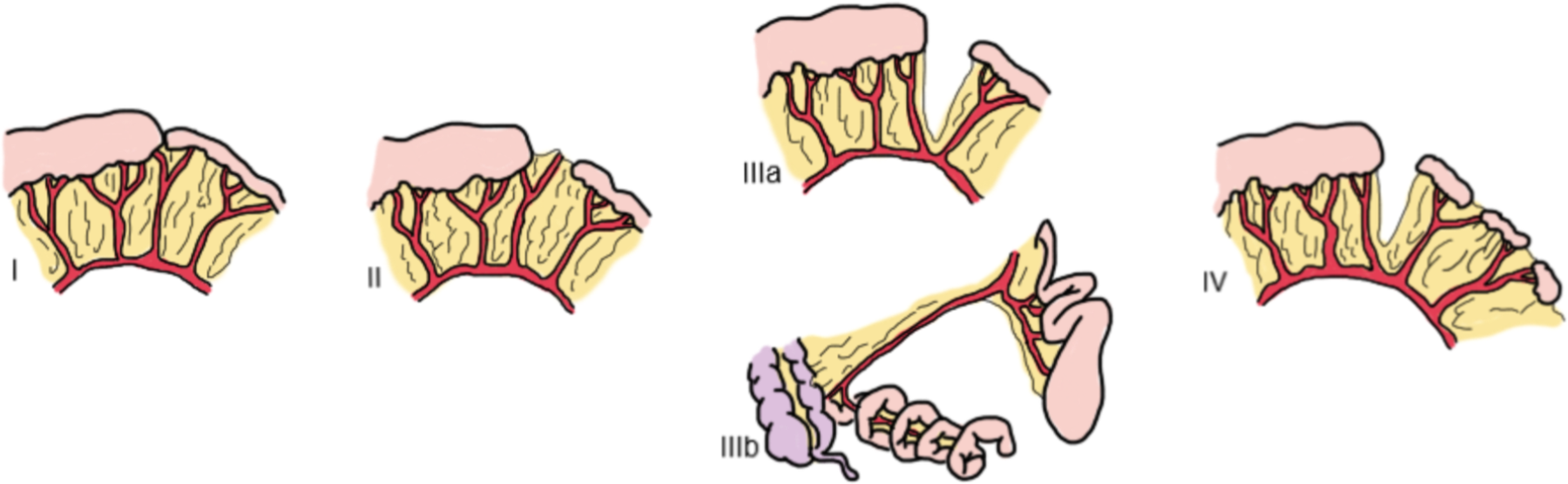
Grosfeld Classification of Jejunoileal Atresia.*

* Grosfeld et al 9 proposed this classification scheme for jejunoileal atresia based on the pathologic findings and anatomic defect. This classification is widely used by pediatric surgeons because it also guides the surgeons in choosing the proper surgical procedures for treating patients.
Epidemiologic study has shown that the reported incidence of jejunal or ileal atresia is estimated in 0.9 neonates per 10 000 births, accounting for up to 20% of all intestinal atresia. 10 Different from duodenal atresia, JIA is not commonly associated with multiorgan anomalies. Some conditions that copresent with JIA in up to 10% of the reported cases include intrauterine growth restriction, gastroschisis, volvulus, and cystic fibrosis. 11,12
As the intestine is obstructed, JIA may present with polyhydramnios, dilated bowel, and increased bowel echogenicity. Clinically, the diagnosis of JIA can be very difficult, as prenatal ultrasound does not always detect this intestinal abnormality. Studies have shown that the sensitivity in detecting intestinal obstruction depends on the location of the lesion, with 50% sensitivity for duodenal obstruction, 40% sensitivity for jejunal/ileal obstruction, and 30% sensitivity for colonic obstruction. 13
It appears that there is a constant retrograde flow of bilious content into the proximal digestive tract, which explains the abnormal peristalsis detected by ultrasonography and bilious-stained amniotic fluid. In addition, normally intrauterine fetal expression takes place to allow lung development and maturation, but in this instance, it also results in introduction of bile-stained amniotic fluids into the lung.
What Is the Pathogenesis of Jejunoileal Atresia?
Multiple theories have been proposed to explain the etiology of JIA. Currently, the well-accepted one is the vascular accident theory, which is based on the pioneer work of Louw and Barnard. 8 While experimenting with in utero canine models, Louw and Barnard were able to reproduce almost all types of JIA, through ligating various mesenteric vessels. They then postulated that intestinal ischemia during the gestational period can lead to malformation/involution of the intestine such as atresia. Their theory is further supported by case reports of JIA where vascular compromise was found to be the cause of the defect.
Of note, one particular subtype of JIA, namely, Type 4, is associated with a genetic defect and is almost always fatal. Hereditary multiple intestinal atresia is an autosomal recessive disorder that presents with multiple atretic segments of the small bowel and is associated with immunodeficiency. 11 Due to its unique configuration, the term “sausage on the string” has been coined to describe such morphology. Although it has not been completely elucidated, the current understanding of the pathogenesis is a mutation of the tetratricopeptide repeat domain-7A gene (TTC7A), which is believed to have an important role in proper development and preservation of gastrointestinal epithelium. 14
How Is Jejunoileal Atresia Different from Duodenal Atresia?
Although both disorders have a very similar presentation and the point of atresia is only different by few inches, etiologically duodenal atresia is very different from JIA. During the early stages of the embryonic development, the caudal portion of foregut elongates and joins with the rostral part of the midgut to form the primitive duodenum. The duodenal lumen is initially obstructed due to the proliferation of epithelial cells, but it will undergo reformation later to canalize the duodenum. Hence, it is believed that the failure of canalization or overproliferation of epithelial linings is a major cause of duodenal atresia. 15 Additionally, duodenal atresia occurs at sixth to eighth week of gestation, whereas JIA usually happens in the second or third trimester. 10,11 It is also noteworthy to mention that duodenal atresia is typically associated with Down syndrome, whereas JIA hitherto has not been associated with any congenital syndrome.
How Should the Cause of Death Be Stated?
Intrauterine fetal demise secondary to JIA, type IIIb in the setting of intrauterine growth restriction in a 36-week female fetus.
What Might Have Been the Mechanism Causing the Fetal Demise?
It is important to mention that intestinal atresia/agenesis alone should not lead to fetal demise. In fact, the literature documents the various types of intestinal atresia that are seen in live births and discusses their respective surgical interventions and clinical outcomes. 9,12 Therefore, some other events must have been triggered mechanistically to cause the fetus to die.
Over the last few decades, several working theories have been proposed to elucidate the mechanistic steps by which JIA leads to fetal death; among them, there are 2 plausible theories in this clinical picture. First, it is well documented in the literature that intrahepatic cholestasis of pregnancy, although benign for the mothers, increases the chance of perinatal adverse events, such as preterm birth, fetal stress, meconium-stained amniotic fluid, and IUFD. 16,17 In a meta-analysis study, 18 Ovadia et al were able to demonstrate that there is an increased risk of stillbirth when the total serum bile acid is high in the mother. Moreover, there is a great body of evidence in the current literature documenting the arrhythmogenic effect of serum bile acids, based on both in vitro studies of fetal murine hearts and long-term studies of patients with liver cirrhosis. In the current scenario, it is likely that the fetus was aspirating intestinal contents throughout the gestation and it is a reasonable assumption that some of the bile acids entered the fetal bloodstream. Consequently, a level of bile acid high enough to trigger a fetal heart arrhythmia was eventually reached, leading to IUFD.
The second postulate also revolves around cardiac dysfunction, but the cause is of a different mechanism. 19,20 In a retrospective case series reviewed by Brantberg et al where they were correlating duodenal atresia with adverse perinatal events, there was also a case of unexpected late-term IUFD with isolated duodenal atresia. Prior to fetal death, there were documented episodes of bradycardia detected via ultrasonography and Doppler studies. Brantberg et al hypothesize that the marked dilatation of the upper gastrointestinal tract could result in overactivity of vagal tone, leading to profound bradycardia and eventually asystole. Except for the difference in the atretic site, the case study described by Brantberg et al is analogous to the current case. Although there was no documented arrhythmia by the ultrasound clinician, it could still be a possible mechanism leading to fetal death.
What Do the Autopsy Findings Mean for the Clinicians and the Family?
In general, autopsies are performed with the intent and purpose of identifying or ascertaining the cause of death of the deceased. Here, we were able to help the physicians and the parents by providing a satisfactory explanation for how a rare anomaly led to a chain of events culminating in an IUFD. Not only does this information provide reassurance to the family that there was no wrongdoing in terms of prenatal care but also the clear explanation can bring closure. Finally, the autopsy findings can guide the clinicians when advising patients about future pregnancies. The current understanding of JIA is that it is primarily caused by the vascular accident and hence the risk for recurrence should not be a huge concern.
Teaching Points
Jejunoileal atresia is an uncommon congenital anomaly that can have a similar presentation as duodenal atresia. It is important to distinguish the 2 disorders because their diagnosis have different clinical implications and additional testing may be required in the setting of duodenal atresia.
Ischemic insult/vascular accident of the fetal intestinal tract is believed to be the etiology of JIA. It typically occurs in the second or third trimester, when insufficient blood supply results in intestinal involution and atresia.
Medical autopsy and placental examination are warranted in the context of fetal death, as postmortem examination may be helpful in providing answers to unexplained clinical questions and advice about future pregnancies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
