Abstract
Keywords
Primary Objective
Competency 2: Organ System Pathology; Topic HWC: Hematopathology—White Cell Disorders; Learning Goal 3: Classification of Leukemia and Lymphomas.
Patient Presentation
A 19-year-old woman presented to the emergency department complaining of fatigue and easy bruising over the past few weeks as well as fevers without chills or recent weight loss. She had no significant past medical history. She denied any personal or family history of cancer and was taking no medications. Physical examination revealed diffuse bruising of the arms, trunk, and legs without organomegaly or palpable lymphadenopathy.
Diagnostic Findings
A complete blood count (CBC) including the automated differential is provided in Table 1.
Complete Blood Count and Automated Differential Count.
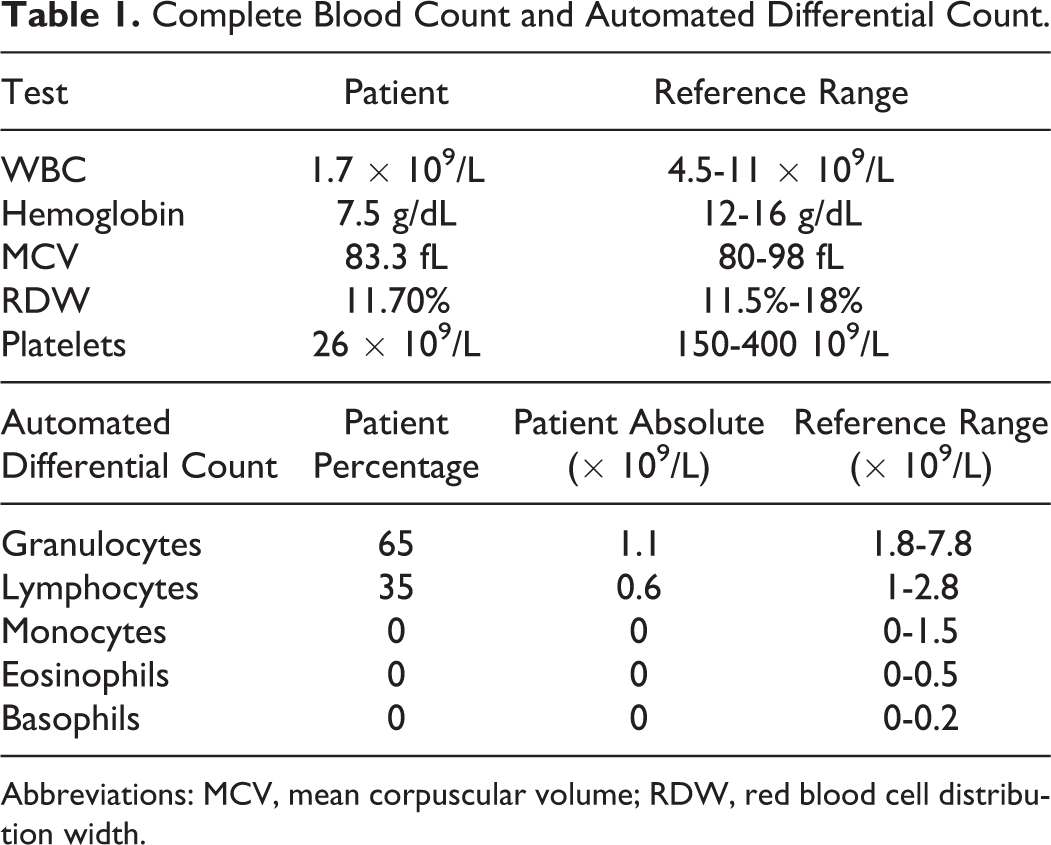
Abbreviations: MCV, mean corpuscular volume; RDW, red blood cell distribution width.
Questions/Discussion Points
What Is the Differential Diagnosis Based on the Clinical Findings and Complete Blood Count Data and What Should be the Next Step in Evaluating This Patient?
Review of the CBC and automated differential provided by the hematology analyzer reveals pancytopenia. The differential diagnosis of pancytopenia is broad and includes nutritional deficiencies, medication effect, other toxins, acute or chronic infections, autoimmune diseases, infiltrative bone marrow processes, hypersplenism, and primary bone marrow disorders, including aplastic anemia, myelodysplastic syndrome, leukemia, and paroxysmal nocturnal hemoglobinuria. Given the patient’s age and physical exam findings, a peripheral smear review with manual differential is an important initial step in narrowing the differential diagnosis. The manual differential count is performed by having a trained laboratory person visually review the stained blood smear, count 100 cells, and assess for cellular morphology, count confirmation, and artifacts, such as platelet clumps.
How Does the Peripheral Smear Findings in This Case Help Narrow the Differential?
The manual differential is provided in Table 2 and images from the peripheral smear are shown in Figure 1. The manual differential confirms pancytopenia and includes circulating nucleated red blood cells and immature cells/blasts consistent with a leukoerythroblastic smear. Smear review reveals the leukocytes present to be abnormal in appearance with “figure eight”-shaped or folded nuclei (Figure 1A), numerous cytoplasmic granules, and few forms with Auer rods (Figure 1B). Auer rods represent fused primary granules. The presence of Auer rods is diagnostic of a neoplastic blast with myeloid differentiation and is important to distinguish from lymphoblasts due to different treatment regimens in acute myeloid leukemia (AML) or other myeloid neoplasms and acute lymphoblastic leukemia.
Manual Differential Count.
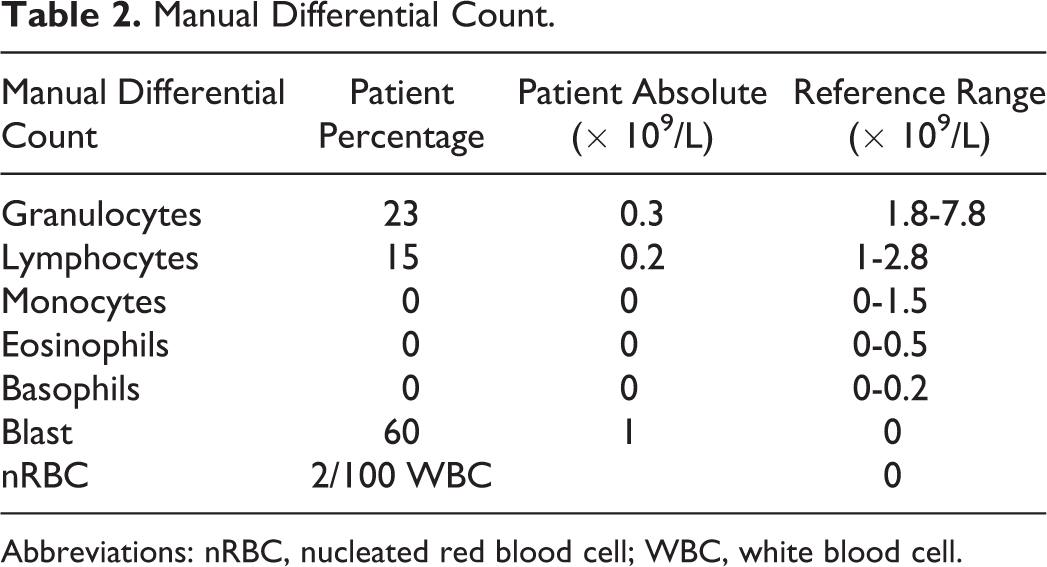
Abbreviations: nRBC, nucleated red blood cell; WBC, white blood cell.
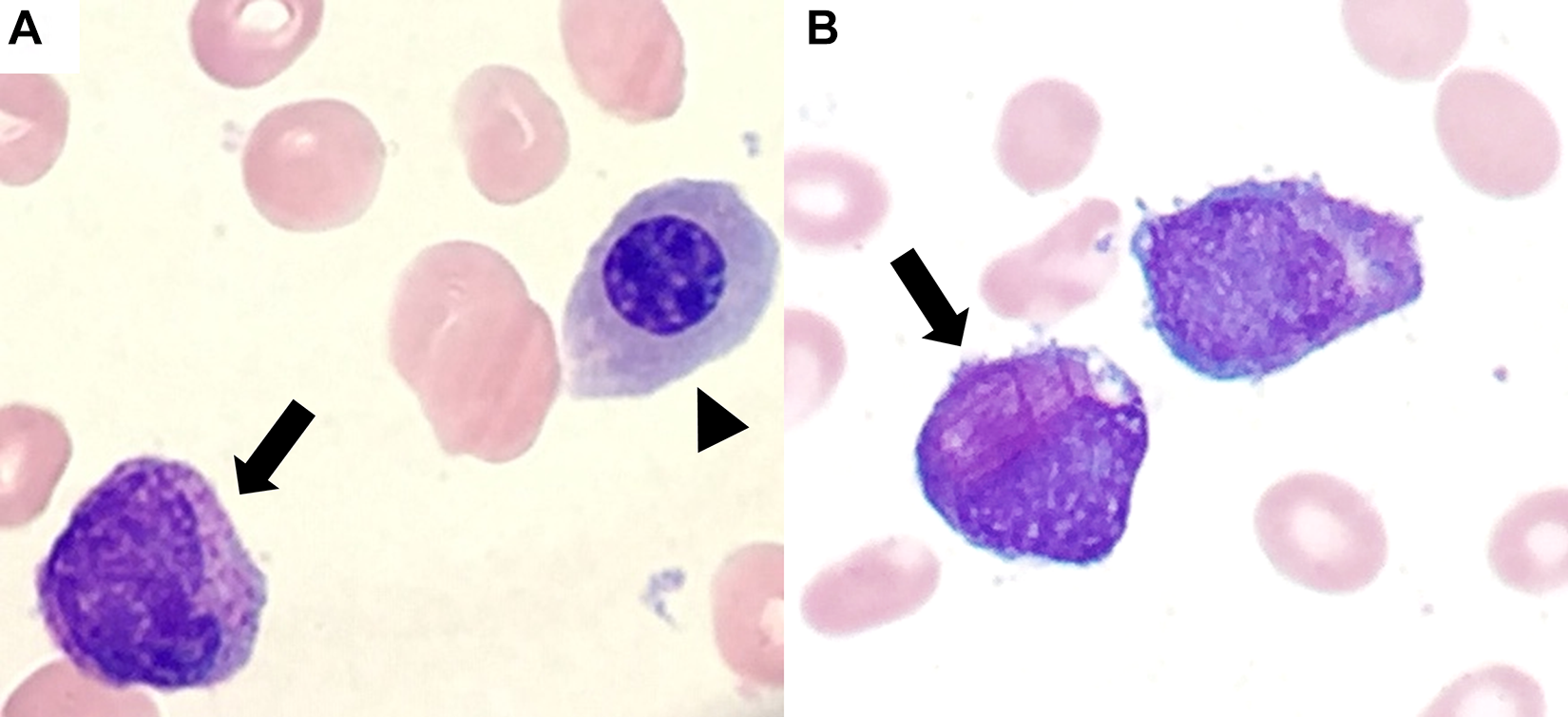
A, Leukoerythroblastic peripheral smear (×1000 magnification). Note the abnormal leukocyte with folded nucleus and numerous cytoplasmic granules (arrow) as well as the nucleated red blood cell (arrowhead). B, Leukocyte with numerous Auer rods (arrow) (×1000 magnification). Auer rods result from a fusion of primary granules and are diagnostic of a neoplasm with myeloid lineage.
The diagnosis of AML requires a blast percentage of ≥20% blasts dedicated to myeloid differentiation including myeloblasts, atypical/neoplastic promyelocytes, monoblasts/promonocytes, and/or megakaryoblasts in the peripheral blood or bone marrow. An exception to this rule is the presence of a translocation involving one of the core binding factors
Acute myeloid leukemia with t(8;21)(22q;q22.1);
Acute myeloid leukemia with a predominance of abnormal promyelocytes and a
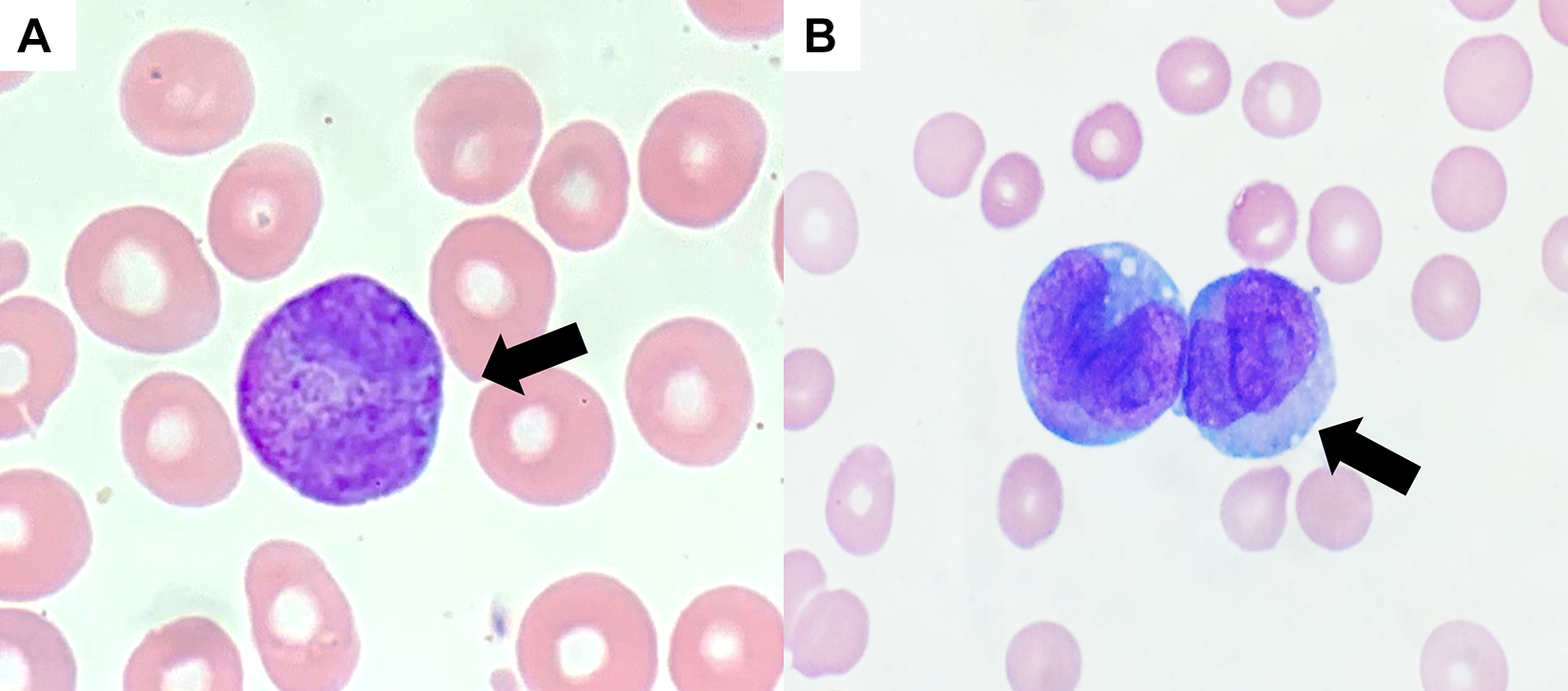
Acute promyelocytic leukemia (APL). A, Hypergranular APL (×1000 magnification). Note the abnormal promyelocytes with figure eight-shaped nucleus (arrow) and numerous cytoplasmic granules. In hypergranular APL, the granules can become so numerous that they obscure the nuclear features of the cell. B, Hypogranular APL (×1000 magnification). Note the absence of cytoplasmic granules on Wright Giemsa stain (arrow). The granules in these cases are submicroscopic and can be seen with a myeloperoxidase (MPO) stain.
What Additional Testing Should Be Performed to Confirm the Suspected Diagnosis?
Morphologically, the increased blasts with “figure eight”-shaped nuclei, numerous cytoplasmic granules, and Auer rods are suspicious for APL. To confirm this diagnosis, flow cytometric evaluation of the peripheral blood as well as rapid fluorescence in situ hybridization (FISH) to evaluate for
Flow cytometry and FISH can be performed rapidly on peripheral blood or bone marrow aspirate. Flow cytometry involves labeling cells with fluorescently tagged antibodies that are specific for hematolymphoid differentiation. These cells are then passed, single file, through a laser beam which detects the fluorescent label and separates the cells by antigen profile. In this case, the neoplastic cells were positive for CD117, CD33, CD13, and CD64 and mostly negative for CD34 and human leukocyte antigen-DR (HLA-DR) (Figure 3). The expression of CD33, CD13, and CD64 favor myeloid lineage, and this can be confirmed by MPO expression. Myeloperoxidase is an enzyme found in myeloid cells that aids in immune and inflammatory functions. The expression of CD117 is a marker of immaturity, and the hypergranular variant of APL is typically negative for other markers of immaturity including CD34 and HLA-DR. This is in contrast to the microgranular variant which typically expresses CD34 in conjunction with CD2. The flow cytometric findings are characteristic of AML and most consistent with hypergranular APL. The increased suspicion of APL in this case can be confirmed by FISH which uses DNA probes to recognize specific sequences located in a particular chromosomal region. These DNA clones are labeled with fluorescent dyes which bind to the desired chromosomal region and create a signal that is visualized under a fluorescent microscope. 5 Each gene is assigned a specific color probe, and within the cells being evaluated, there should be 2 signals for each gene (red and green, for example). When a translocation occurs, the 2 separate color probes fuse and a third color signal (yellow) is present due to the overlapping of the other 2 colors (Figure 4).
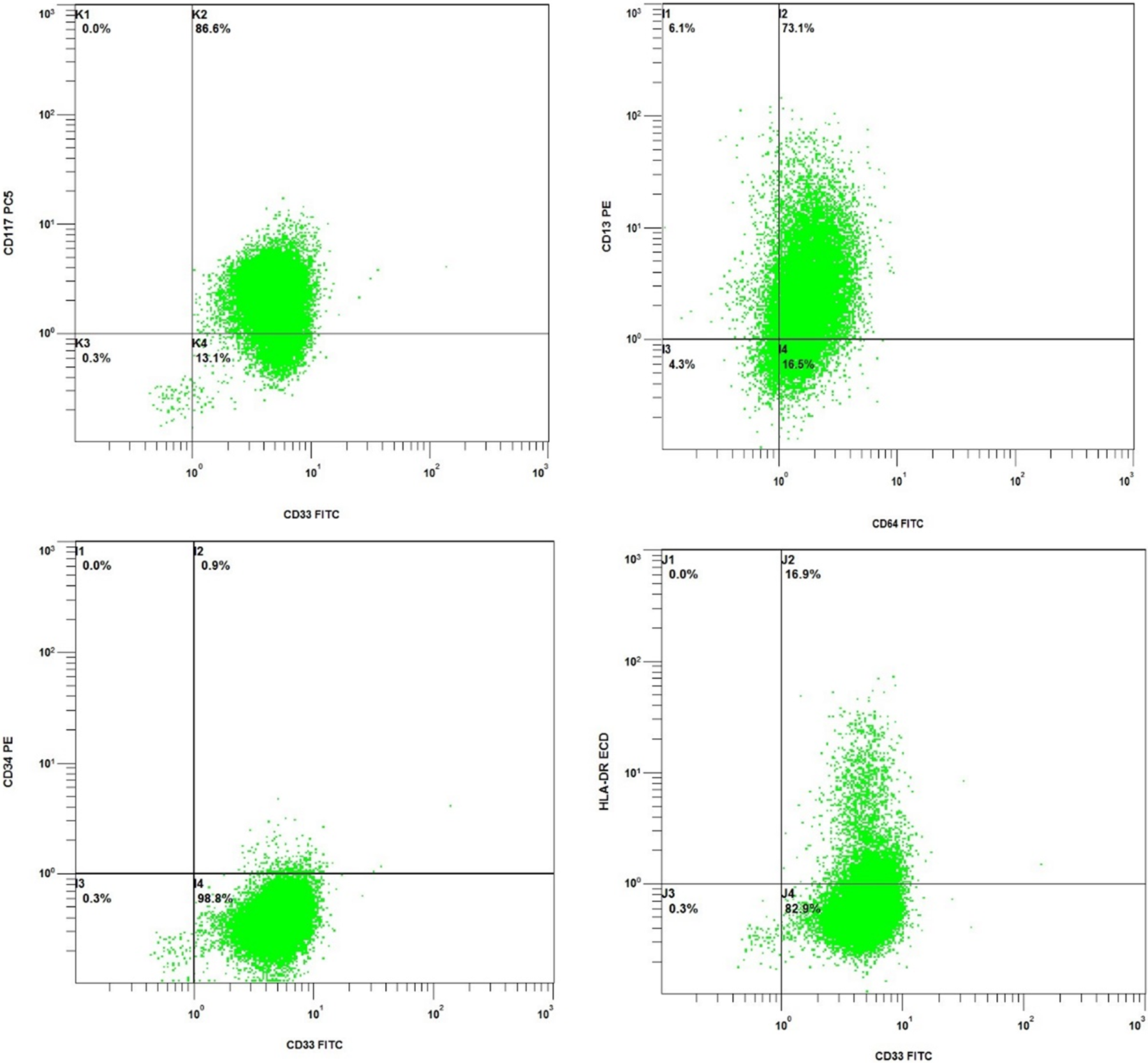
Flow cytometric phenotype of acute promyelocytic leukemia (APL). The neoplastic cells (green) are positive for CD117, CD33, CD13, and CD64 and mostly negative for CD34 and human leukocyte antigen-DR (HLA-DR).
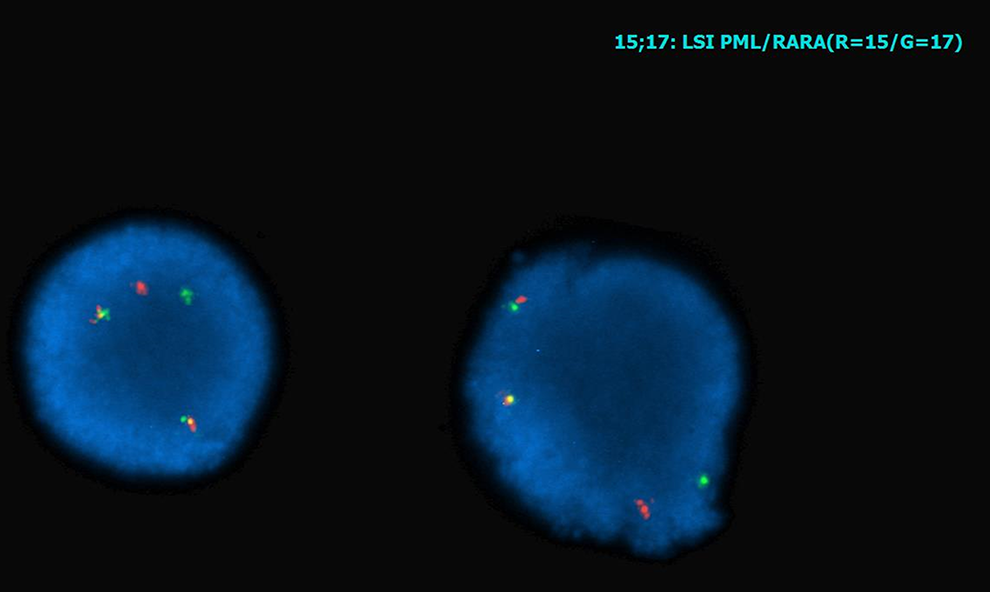
Fluorescence in situ hybridization (FISH) for
How Does the Fusion of These 2 Genes Result in Leukemia?
A subset of cases will demonstrate variant translocation partners with the
Common Translocation Partners in APL With Variant
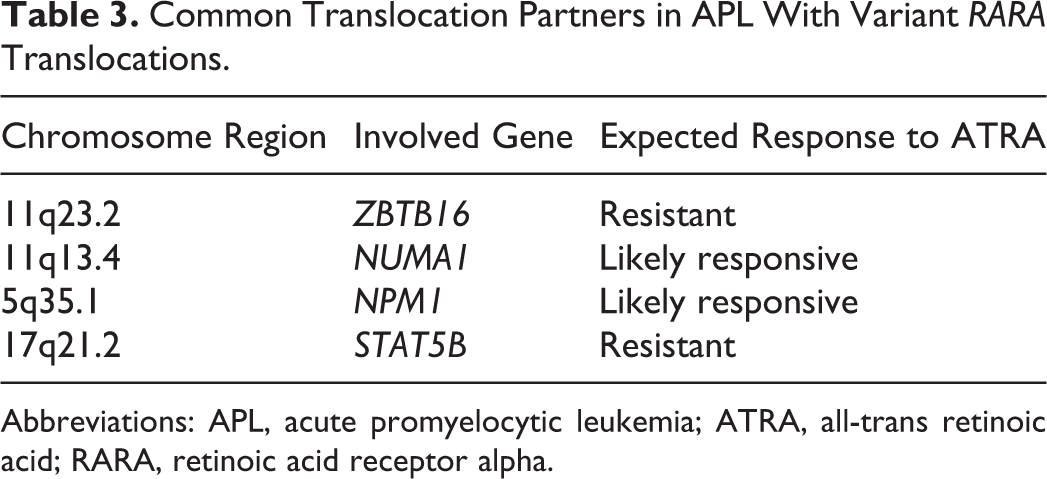
Abbreviations: APL, acute promyelocytic leukemia; ATRA, all-trans retinoic acid; RARA, retinoic acid receptor alpha.
What Is the Preferred Treatment Regimen for Acute Promyelocytic Leukemia? Are There Findings That Help Predict the Clinical Course or Treatment Response?
Rapid diagnosis and initiation of treatment of APL is critical due to the high risk of early death related to DIC. The successful treatment of APL with all-trans retinoic acid (ATRA) and arsenic trioxide is well-documented and is one of the first examples of differentiation therapy.
6
The
Two of the known variant translocations that are resistant to ATRA therapy, thus rendering patients more prone to developing DIC, include t(11;17)(q23;21);
Teaching Points
Acute myeloid leukemias have distinct morphologic features based on their genetic aberrations. Acute promyelocytic leukemia is a specific subtype of AML with a characteristic t(15;17)(q22;q12); Acute promyelocytic leukemia is characterized by atypical promyelocytes that can be hypergranular with “figure eight” nuclei, abundant cytoplasmic granules, and bundles of Auer rods, or hypogranular with similar nuclear shape but indistinct cytoplasmic granules. Rapid diagnosis by flow cytometry and FISH is essential due to the common association with DIC. There is an excellent clinical response to therapy with ATRA and arsenic trioxide which act as differentiating agents, and when used in combination with standard induction chemotherapy in high risk patients can achieve complete remission.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
