Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Objective
CHEM1.7: Toxicology. Determine the value of testing for drugs and toxins accounting for the routes of administration, distribution and metabolism of the agent of interest, including the specimen source, the analytes to be detected given the medical questions, and the timing constraints for specimen collection.
Competency 3: Diagnostic Medicine and Therapeutic Pathology; Topic Chemistry (CHEM): Learning Goal 1: Pathogenesis, Diagnosis, and Treatment of Common Disorders
Patient Presentation
A 70-year-old man is brought to the emergency department (ED) by his wife and brother-in-law for confusion, slurred speech, and altered mental status that started approximately 2 hours before presentation to the ED. The patient’s mental status had been normal earlier in the day. He had been depressed for several months following the recent deaths of his brother and father and has talked about life not being worth living. The patient and his wife had been in the house together the whole day except for a period approximately 6 hours prior to presentation to the ED when the patient went into the garage to retrieve some supplies. The wife did not observe what happened in the garage but noticed that her husband changed shirts after returning. The husband resisted going to the ED but finally relented after the wife called her brother for assistance.
Upon arrival in the ED, the patient was confused and appeared intoxicated, having difficulty following basic questions and commands from the medical team. Vital signs were temperature 36.3°C, blood pressure 128/78 mm Hg, heart rate 68 beats/min, respiratory rate 14 breaths/min, and O2 saturation 97% on room air. Screening physical examination before intubation showed no major abnormalities aside from depressed neurological status. Neurologic examination did not reveal any focal findings. The patient was not talkative and provided very little information. He did not admit to any recent use of drugs or alcohol. His chronic medications include hydrochlorothiazide for hypertension, a statin for hypercholesterolemia, and daily low-dose aspirin. The wife was not aware of any other medications in the house except acetaminophen, aspirin, and various cough and cold over-the-counter products. The patient had no prior history of myocardial infarction, stroke, thromboembolism, alcoholism, or renal issues.
Diagnostic Findings
The laboratory studies performed on the patient are summarized in Table 1.
Laboratory Test Results.
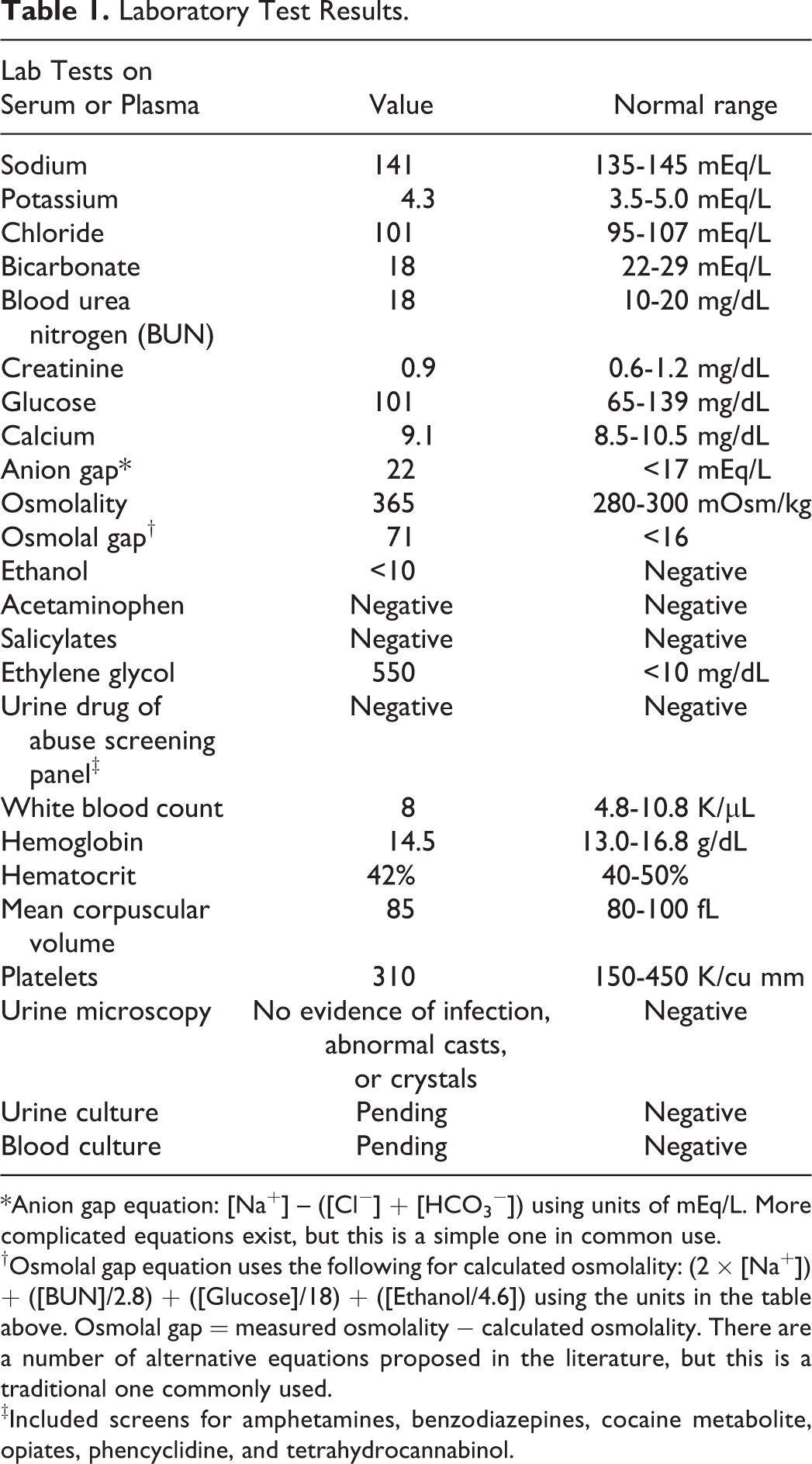
*Anion gap equation: [Na+] – ([Cl−] + [HCO3 −]) using units of mEq/L. More complicated equations exist, but this is a simple one in common use.
†Osmolal gap equation uses the following for calculated osmolality: (2 × [Na+]) + ([BUN]/2.8) + ([Glucose]/18) + ([Ethanol/4.6]) using the units in the table above. Osmolal gap = measured osmolality − calculated osmolality. There are a number of alternative equations proposed in the literature, but this is a traditional one commonly used.
‡Included screens for amphetamines, benzodiazepines, cocaine metabolite, opiates, phencyclidine, and tetrahydrocannabinol.
Questions/Discussion Points
Given the Clinical History, What Should Broadly Be Considered in the Differential Diagnosis?
The patient presentation can be summarized as delirium occurring on a background of major depression-like symptoms following the recent passing of 2 close relatives. The differential diagnosis of delirium in a 70-year-old man is broad and includes cardiovascular events, stroke, cerebral injury, metabolic disturbances (eg, hyponatremia), adverse effects of medications, infections, renal compromise, or intoxication or withdrawal from alcohol or other drugs. Multiple possibilities on the differential diagnosis (eg, stroke, myocardial infarction, and toxic ingestions) require urgent clinical attention, and thus, priority is placed on obtaining key diagnostic studies.
Given the Clinical Presentation, What Diagnostic Testing Is Indicated?
With the sparse history elicited from the patient and nonspecific physical examination findings, it is difficult to exclude most of the items on the differential diagnosis without diagnostic testing. Initial laboratory workup would include basic chemistries, urinalysis, complete blood count, serum ethanol, serum acetaminophen, serum salicylate, serum osmolality, troponin T, urine drug of abuse screening panel (or similar toxicology screen), urine cultures, and blood cultures. Most of these laboratory studies are likely available with fast turnaround time by the clinical laboratory or point-of-care testing associated with the ED. Urine and blood cultures will take time, although other laboratory studies (eg, urinalysis) may indirectly identify infection. Drug abuse/toxicology testing is important as common drugs or medications such as ethanol, benzodiazepines, and opioids can alter mental status and resulted in a clinical presentation similar to that seen in the patient. Additional possibilities for laboratory studies include arterial blood gas analysis. Diagnostic imaging (eg, computed tomography scan of the head) may also be indicated.
What Acid–Base Abnormality for the Patient Is Elucidated by the Laboratory Values? How Are Anion Gap and Osmolal Gap Calculated, and What Do the Values for This Patient Indicate?
The results of the basic metabolic panel are consistent with metabolic acidosis (low bicarbonate). This could be more definitely assessed with arterial blood gas analysis. Other notable laboratory findings in this patient are an elevated anion gap and markedly elevated serum osmolality. Anion gap is typically calculated by the serum/plasma sodium concentration minus the sum of the serum/plasma bicarbonate and chloride concentrations, with analytes measured in mEq/L (note: there are some variations in this basic formula such as incorporating potassium). The reference range for anion gap is approximately 8 to 16 mEq/L. An elevated anion gap indicates the presence of unmeasured anions such as lactate, glycolic acid, or formic acid.
Determination of serum osmolality by freezing point depression on an osmometer (“measured osmolality”) can allow for the calculation of the osmolal gap (OG). Determination of OG requires a “calculated osmolality” that is derived from equations that use the main endogenous contributors to serum osmolality such as sodium (typically multiplied by 2 to account for all the anions that counter the positive charge of sodium ions), urea nitrogen, and glucose (footnote in Table 1 uses a commonly used calculated osmolality equation). The OG is the measured osmolality minus the calculated osmolality, with most of the healthy population falling between −10 and +10. Suspicion of a pathophysiologic or toxic contribution to osmolality increases with higher OG. Values exceeding 30 have more specificity for toxic alcohol or glycol ingestion compared to other causes. 2,3 There are also equations that incorporate the estimated osmolal contribution of serum ethanol (if present). While there is debate in the literature on optimizing OG equations and thresholds for diagnosing toxic alcohol/glycol ingestions, OG can be a useful tool in management. 4,5 The differential diagnosis for an elevated OG includes toxic alcohols and glycols, diabetic ketoacidosis (presence of acetone and other ketones), alcoholic ketoacidosis, renal failure, mannitol administration, and shock. 2
What Is Most Likely Diagnosis Given the Clinical and Laboratory Findings?
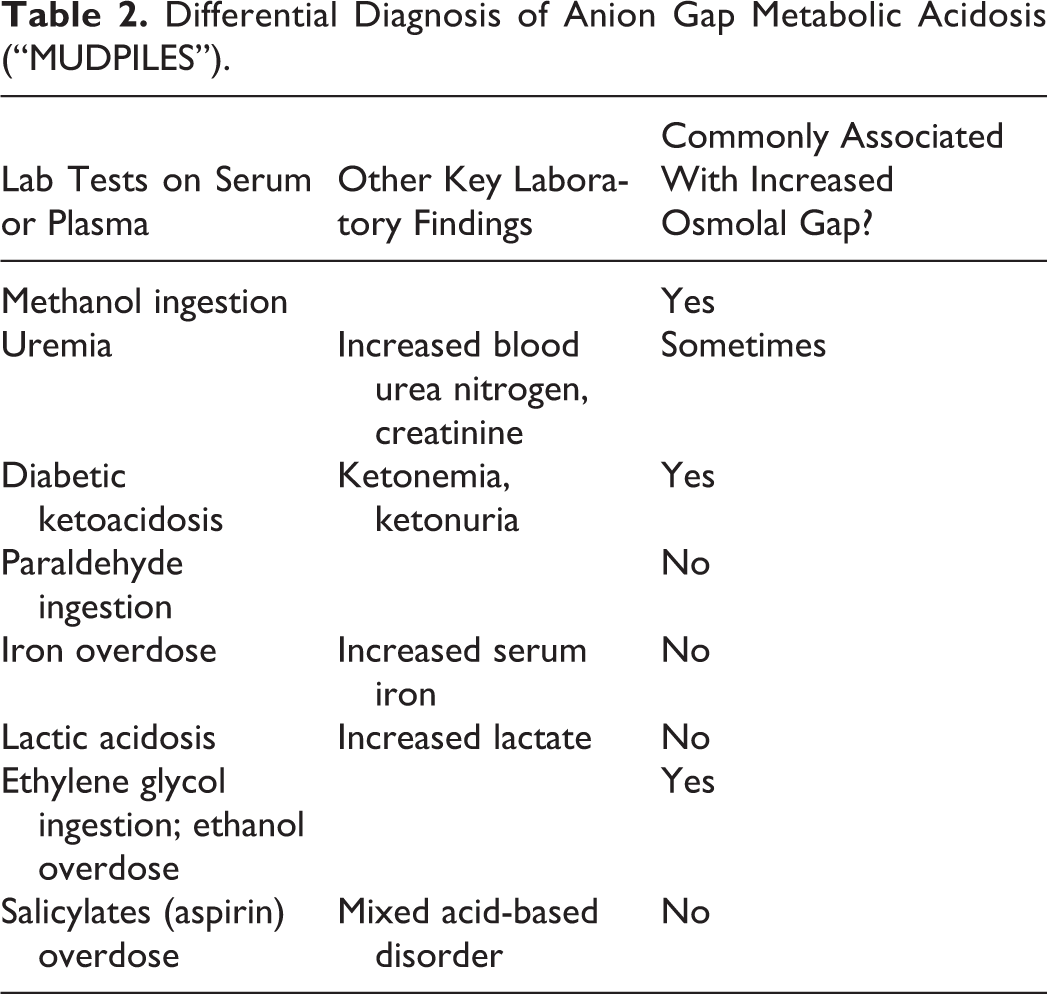
Common causes of high anion gap, metabolic acidosis (often summarized using the mnemonic “MUDPILES” with some variation in possibilities for each letter) include methanol ingestion, uremia (renal insufficiency/failure), diabetic ketoacidosis, paraldehyde (obsolete drug), iron poisoning, lactic acidosis, ethanol or ethylene glycol (EG) ingestion, and salicylate poisoning. 5,6 Table 2 summarizes causes of high anion gap, metabolic acidosis, also indicating which may be associated with increased OG. Some of the possibilities in Table 2 are less likely for the patient given the diagnostic testing results in Table 1—normal creatinine reduces probability of renal compromise, normal serum glucose reduces probability of diabetic ketoacidosis, and undetectable salicylates and ethanol would be unlikely if those drugs were the cause of the confusion and altered mental status. The high serum osmolality narrows the search further toward a toxic alcohol or glycol ingestion such as EG or methanol, 6 compounds that may have been in products in the patient’s garage. Ingestion of either of these would be life-threatening, with time-sensitive clinical options for therapy. Ingestion of isopropanol (“rubbing alcohol”) is also a possibility, as this alcohol can produce altered mental status and elevated osmolality. However, isopropanol does not commonly cause high anion gap, metabolic acidosis as it is predominantly metabolized to acetone.
Differential Diagnosis of Anion Gap Metabolic Acidosis (“MUDPILES”).
What Are Sources of Ethylene Glycol and Methanol?
Ethylene glycol is most commonly found in automobile antifreeze. 6 Ethylene glycol has a sweet taste, a factor that may lead to accidental ingestions by young children and pets. Methanol (methyl alcohol, “wood alcohol”) is found in a variety of products including windshield fluid and industrial cleaning supplies. Bootleg alcohol contaminated with methanol has resulted in outbreaks of methanol poisoning (eg, in Eastern Europe).
How Can You Make Diagnosis of Ethylene Glycol and Methanol Poisoning?
The definitive laboratory method for measurement of toxic alcohols and glycols in serum/plasma is gas chromatography (GC). 5,6 This method can detect and individually quantitate concentrations of EG, methanol, and other toxic alcohols such as isopropanol. Gas chromatography is also a reference method for measurement of serum/plasma or whole blood ethanol (especially in the law enforcement and forensic realms), although most clinical laboratories use automated enzymatic assays for routine measurement of ethanol. A significant limitation of GC is that the technique is labor-intensive and often not performed at clinical laboratories serving hospitals and medical centers, with the exception of some larger medical center laboratories. Consequently, GC may only be available at remote reference laboratories, with a turnaround time too slow for immediate clinical decision-making. In some cases, a hospital or medical center is close enough to a regional or reference laboratory performing GC to allow for suitable turnaround time.
The diagnosis of EG or methanol ingestion thus often depends on clinical history, symptoms, and laboratory testing to assess electrolytes, acid–base status, and serum osmolality. 5,6 A recent development has been the clinical use of enzymatic assays for measuring serum/plasma concentrations of EG. 7,8 These type of assays found earliest application in veterinary medicine, given that accidental ingestion of EG is a common poisoning of dogs and cats. The enzymatic assays have some limitations with specificity (eg, cross-reactivity with propylene glycol, a structurally related compound used as a diluent for poorly water-soluble intravenous medications like lorazepam) but have the advantage of low technical complexity and rapid turnaround time if run on automated chemistry analyzers.
What Organ Systems Can Be Involved With Ethylene Glycol and Methanol Ingestions?
Ethylene glycol and methanol on their own can produce profound central nervous system (CNS) depression similar to that seen with ethanol intoxication. 6 These CNS effects can be dangerous and life-threatening due to respiratory depression, risk of accidents, or injury to neurons. However, much of the toxicity of EG and methanol is caused by their metabolites.
Ethylene glycol is converted to glycolaldehyde by alcohol dehydrogenase (ADH), an enzyme that also catalyzes the first, rate-limiting step in the metabolism of methanol and ethanol. 5 Glycolaldehyde is subsequently converted to glycolic acid, then to glycoxylic acid, and finally to oxalic acid. Glycolic acid is cytotoxic and produces neurologic and cardiopulmonary symptoms. Glycolic acid primarily accounts for the high anion gap, metabolic acidosis of EG ingestions. The end conversion of EG to oxalic acid can result in calcium oxalate crystal formation in the renal tubules approximately 24 to 72 hours after EG ingestion, potentially resulting in acute and then permanent renal injury. The metabolism pathway of EG is summarized in Figure 1. Figure 2 shows an example of oxalate crystals in urine. Figure 3 shows an example microscopic image of oxalate crystals in the renal tubules.
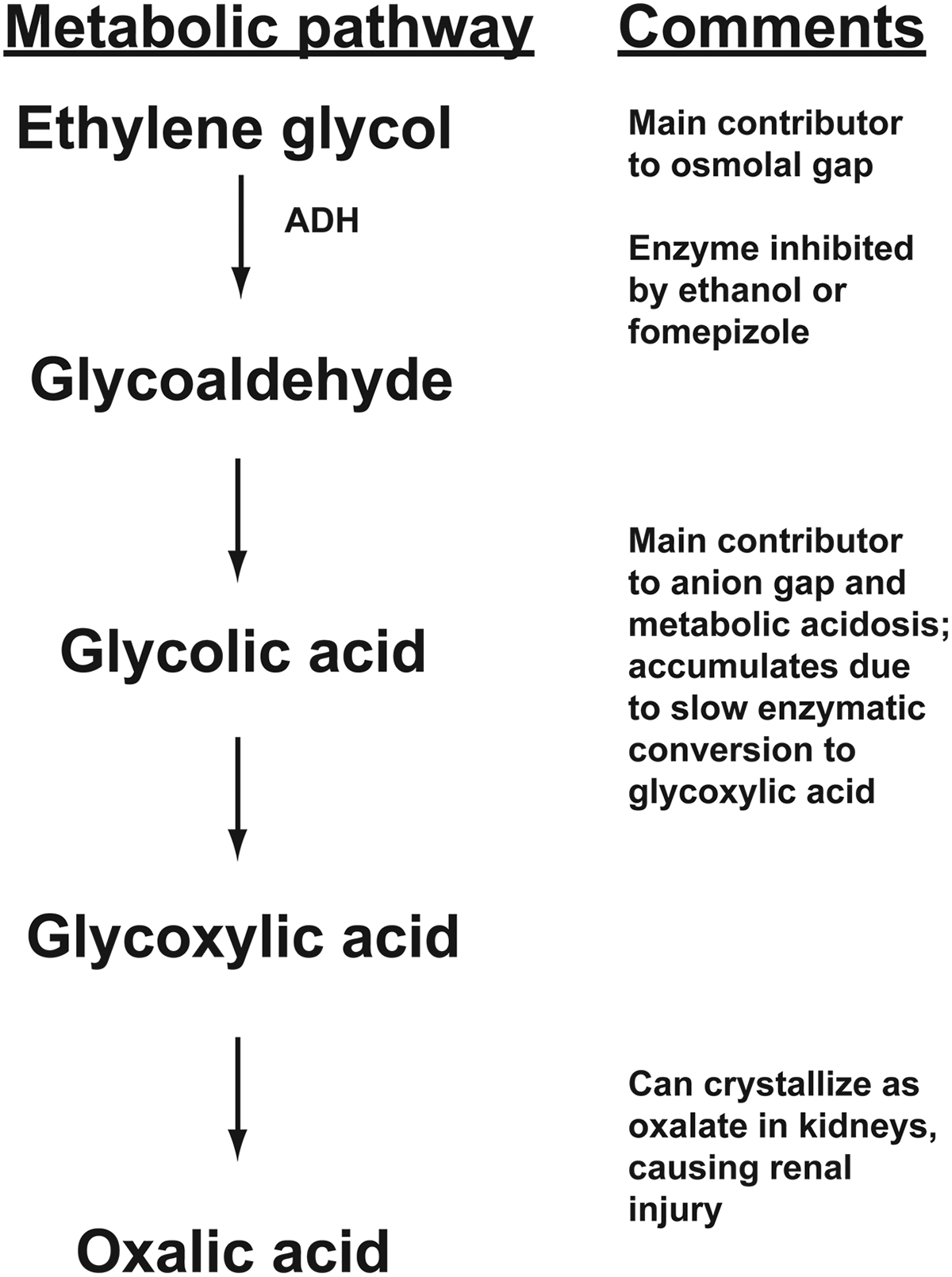
Metabolism pathway for ethylene glycol. ADH indicates alcohol dehydrogenase.
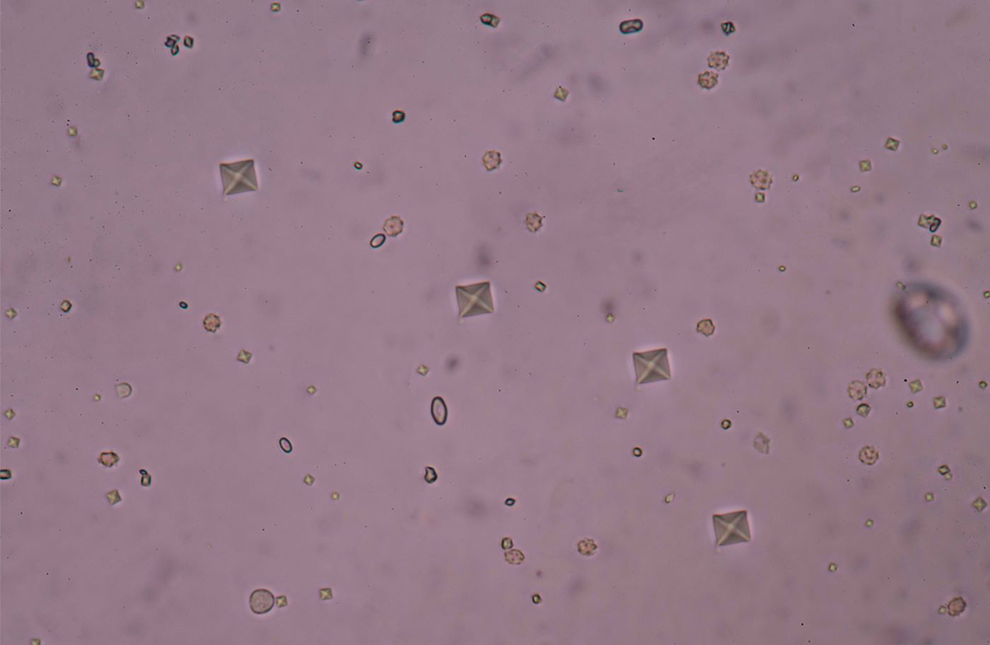
Example of oxalate crystals in urine (S. Kammayeengoen/Calcium oxalate crystal used under license from Shutterstock.com, https://www.shutterstock.com/image-photo/calcium-oxalate-crystal-667783018).
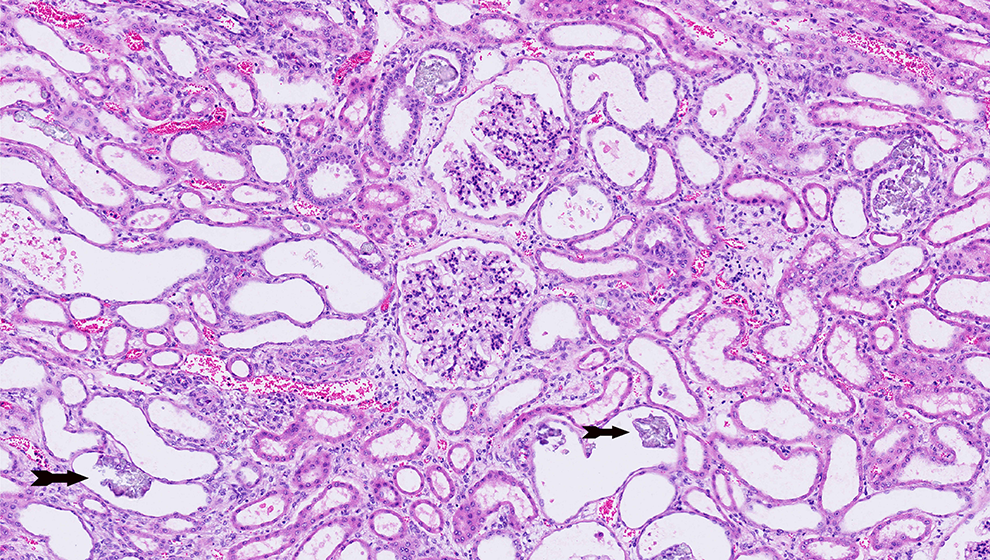
Microscopic image of oxalate crystal deposition in renal tubules. The arrows indicate crystals deposited in the renal tubules (Vetpathologist/Crystals in the kidney tubules. Oxalate crystals in the renal tubular lumen used under license from Shutterstock.com, https://www.shutterstock.com/image-photo/crystals-kidney-tubules-oxalate-renal-tubular-80813026).
Methanol is metabolized by a series of enzymatic reactions to formaldehyde and then to formic acid, a compound that can cause permanent injury to the optic nerve with subsequent blindness. 6 Similar to glycolic acid, formic acid produces high anion gap, metabolic acidosis. The metabolism pathway of methanol is summarized in Figure 4.
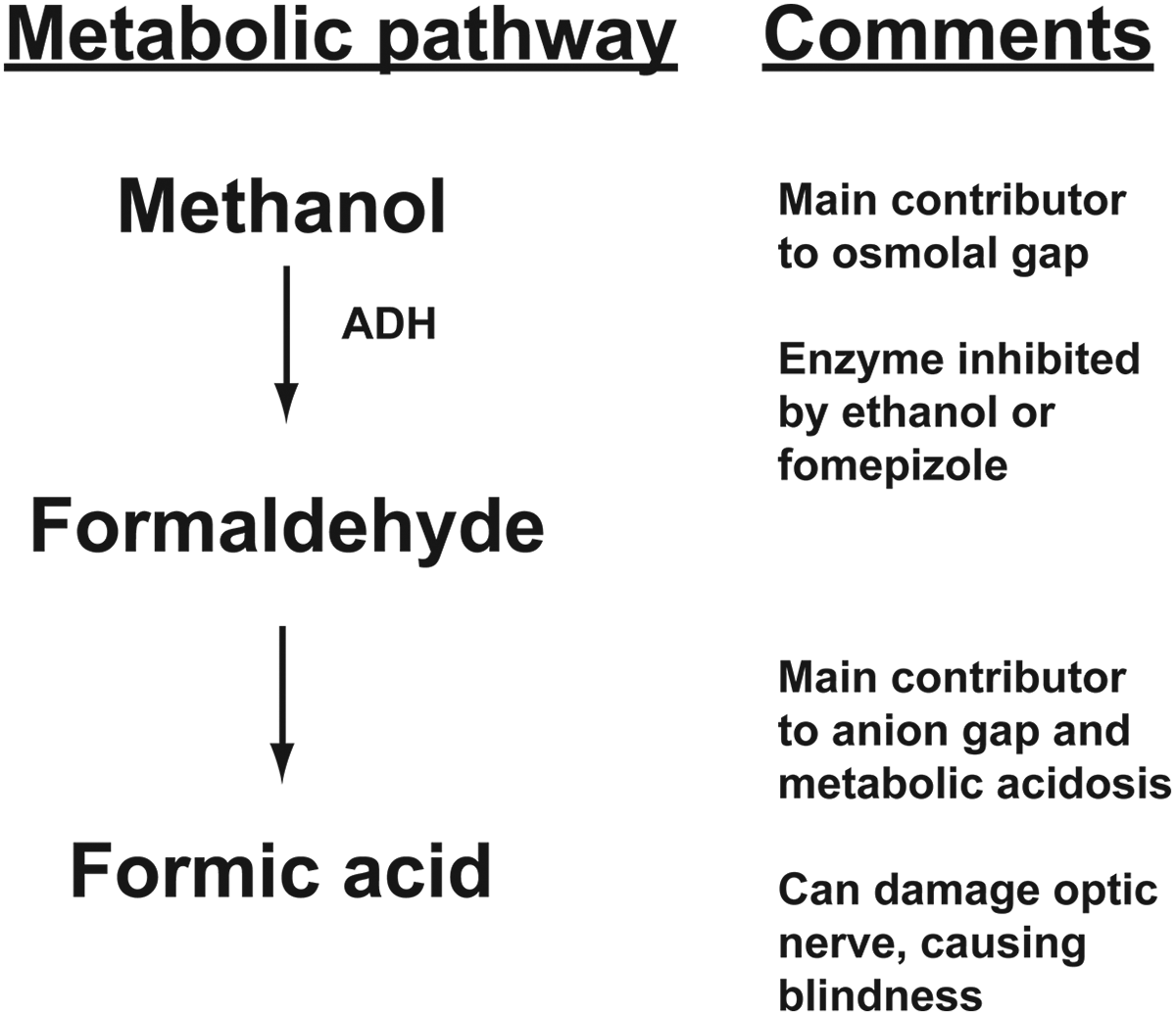
Metabolism pathway for methanol. ADH indicates alcohol dehydrogenase.
What Are Common Laboratory Findings in Ethylene Glycol and Methanol Ingestions?
Ingestion of either EG or methanol classically first produces an OG, followed later by a high anion gap, metabolic acidosis as the glycolic acid or formic acid metabolites accumulate. 5,6 The metabolic acidosis produced by ingestion of EG or methanol is mainly due to these metabolites. Thus, the pattern of results provides clues to the timing of ingestion. Laboratory workup performed within several hours of ingestion may reveal only an elevated OG. Without treatment, metabolic acidosis and anion gap then develop, with an intermediate period where there may be both an elevated OG and high anion gap, metabolic acidosis. If GC analysis is available, concentrations of EG and methanol may be directly determined as the gold standard measurement.
What Are Possible Treatments of Ethylene Glycol and Methanol Ingestions?
The main therapeutic options for managing EG and methanol poisonings are inhibition of ADH and/or renal dialysis. 6,9,10 Fomepizole is a competitive antagonist of ADH marketed specifically for treatment of EG and methanol ingestions. Ethanol is a substrate of ADH that, if given in high enough doses, will inhibit ADH metabolism of EG or methanol. Fomepizole is generally simpler to administer and has the advantage over ethanol of not producing additional CNS depression; however, high cost of the drug may be a barrier for small hospitals that seldom treat ingestions. Ethylene glycol and methanol ingestions that are not diagnosed early enough, or that are severe based on ingestion of large amounts, often require hemodialysis to remove the parent compounds and their toxic metabolites.
An important concept is that the clinical benefit of ADH inhibitors is limited if significant metabolism of EG or methanol has already occurred. Laboratory findings help guide the treatment. A patient presenting with only an elevated OG and without metabolic acidosis or anion gap is likely to be at an early stage of ingestion and to respond well to therapy with ADH inhibitors. In contrast, the presence of metabolic acidosis and/or anion gap is consistent with a later stage of ingestion, with enough time that EG or methanol has already been significantly metabolized. Giving ADH inhibitors in a patient in a later stage of ingestion will prevent further conversion of EG or methanol to metabolites but will not remove the existing metabolites. In those cases, hemodialysis is used to clear the parent compounds and metabolites (eg, glycolic acid, formic acid).
With EG and methanol intoxication, prompt treatment significantly improves outcome and prevents end-organ damage such as renal injury from oxalate crystals. 6,9,10 One practical challenge with EG and methanol ingestions is that decisions on treatment often need to be made based on clinical history, physical examination findings, and indirect laboratory evidence (eg, OG, anion gap, metabolic acidosis) if specific analysis by GC or an EG enzymatic method is unavailable. Because the therapy is the same for EG and methanol poisonings, it is not necessarily crucial in the early clinical management to distinguish between these 2 compounds. The key step is to recognize when there is enough clinical suspicion to initiate therapy using ADH inhibition or hemodialysis. However, distinguishing between the 2 compounds is important given the differing organ toxicities between EG and methanol.
How Should the Patient Be Treated and Followed?
In this patient, confirmatory analysis using GC revealed a serum EG concentration of 550 mg/dL, a level far in excess of known toxic concentrations. The presence of a high anion gap, metabolic acidosis in the patient, but without the presence of oxalate crystals in the urine, was consistent with an intermediate stage of ingestion, with enough time for EG to be metabolized to glycolic acid (producing the metabolic acidosis and anion gap) but not long enough for oxalate crystals to be evident in the urine. In this setting, treatment with ADH inhibitors (fomepizole or ethanol) alone will not be sufficient. Instead, hemodialysis can be used to help clear EG and its metabolites and reduce possibility of end-organ damage due to oxalate crystal deposition in the kidneys. It is important to note that hemodialysis is indicated in this situation even in the absence of markers of renal compromise/failure such as increased blood urea nitrogen or creatinine. The goal of hemodialysis is to prevent irreversible kidney injury by limiting the ultimate metabolism of EG to oxalate.
The patient was therefore treated with hemodialysis to remove both EG and its metabolites. The patient’s mental status improved following the first hemodialysis session. Anion gap, OG, and metabolic acidosis resolved by 36 hours after presentation to the ED. The patient later admitted that he ingested a large volume of EG in the garage as a suicide attempt, with an estimated time of ingestion 6 hours before presentation to the ED. He was transferred to psychiatric care once stabilized medically.
Teaching Points
Ethylene glycol and methanol are toxic compounds that can produce significant CNS depression and organ damage. Both compounds are found in common household products, including EG in automobile antifreeze and methanol in windshield fluid.
A main organ toxicity of EG is damage to the kidneys, caused by precipitation of its metabolite oxalate in renal tubules.
Methanol can produce blindness due to damage of the optic nerve by its metabolite formic acid.
Common presenting symptoms and signs of EG are altered mental status, increased serum osmolality, elevated OG, high anion gap, and metabolic acidosis. Elevated OG is seen early after ingestion and is caused by the parent compounds, while a high anion gap, metabolic acidosis develops later due to metabolites such as glycolic acid (EG) and formic acid (methanol).
The 2 common treatment strategies are inhibition of ADH and hemodialysis. Fomepizole and ethanol are drugs that inhibit ADH and thereby limit conversion of EG and methanol to their toxic metabolites.
Definitive measurement of EG can be done using GC; however, this methodology is not widely available due to technical complexity. Enzymatic assays for EG can be performed on automated clinical chemistry analyzers but have limitations with specificity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
