Abstract
Rationale:
The differential diagnosis for a patient with high-anion-gap metabolic acidosis (HAGMA) is broad; lactic acidosis is an important entity to screen for and treat. An elevated serum lactate is often used as a marker of inadequate tissue perfusion in critically ill patients but can also be indicative of decreased lactate utilization or poor hepatic clearance. Investigating for the underlying cause such as diabetic ketoacidosis, malignancy, or culprit medications is essential to establish the diagnosis and treatment plan.
Presenting concerns of the patient:
A 60-year-old man with a history of substance use and end-stage kidney disease treated with hemodialysis presented to hospital with confusion, altered level of consciousness, and hypothermia. Initial laboratory investigations were significant for a severe HAGMA with elevated serum lactate and β-hydroxybutyrate levels, but toxicology screen was negative, and there was no clear underlying precipitant. Urgent hemodialysis was arranged to mitigate his severe acidosis.
Diagnoses:
He had an initial single dialysis treatment for 4 hours, with posthemodialysis labs showing significant improvement in his acidosis, serum lactate level, and clinical status (cognition, hypothermia). Given this rapid resolution, a sample from his predialysis blood work was sent for analysis of plasma metformin and returned significantly elevated at 60 mcg/mL (therapeutic range 1-2 mcg/mL).
Interventions and outcomes:
On careful medication reconciliation in the dialysis unit, the patient stated he had never heard of the medication metformin, and there was no record of a filled prescription at his pharmacy. Given his living situation with shared accommodations, it was presumed that he had taken medications that were prescribed to a roommate. Several of his other medications including his antihypertensives were subsequently given after dialysis on dialysis days to improve adherence.
Teaching points:
Maintain a broad differential diagnosis for patients presenting with a clinical syndrome consistent with an acute toxicity even if no culprit medications are identifiable on history, especially in patients with a suggestive social history.
Anion-gap metabolic acidosis (AGMA) is common in hospitalized patients but sometimes requires further history and/or confirmatory testing to elucidate the root cause underlying typical causes of AGMA such as lactic acidosis or ketoacidosis.
The main treatment of metformin toxicity is resuscitation and supportive care; however, metformin’s biochemical properties make it readily dialyzable via either diffusion or convection.
The Extracorporeal Treatments In Poisoning group recommends hemodialysis for metformin toxicity when there is a serum lactate >20 mmol/L, a blood pH <7.0, a failure of standard therapy, end-organ damage (hepatic or renal insufficiency), or a decreased level of consciousness.
Introduction
Metabolic acidosis is a common finding in hospitalized patients and generally classified by its association with a high or normal anion gap (AG). Within the high-anion-gap metabolic acidosis (HAGMA) category, there is a broad differential diagnosis including lactic acidosis, kidney failure, ketoacidosis, and several medication or drug toxicities. Here, we discuss a patient with a history of substance use and end-stage kidney disease (ESKD) treated with dialysis who presented with an altered level of consciousness. He was found to have a severe HAGMA with elevated serum lactate level; etiology unclear. The patient was treated with hemodialysis (HD) and dramatically improved. Later confirmatory testing revealed that his serum metformin level on presentation was extremely elevated despite no personal history of diabetes mellitus and no documentation in the provincial database of receipt of this contraindicated medication. We discuss the approach to metabolic acidosis and lactic acidosis, with a focus on the evaluation and management of a patient with metformin toxicity.
Patient Presentation
A 60-year-old man was brought to our hospital with confusion, altered level of consciousness, and hypothermia.
His past medical history was significant for ESKD secondary to presumed ischemic nephropathy treated with intermittent HD 3 times per week; other medical conditions included treated hepatitis C (completed course of sofosbuvir/velpatasvir/voxilaprevir in 2021), hypertension, and dyslipidemia. He was nondiabetic. He had an admission to the local psychiatric hospital remotely for delusions. He had no past surgical history. Home medication list included atorvastatin, amlodipine, pantoprazole, ezetimibe, pregabalin, and perindopril.
He lived in an apartment in a boarding home with a shared bathroom. The apartment complex was noted by paramedics to be poorly maintained. He was an active smoker of about 10 cigarettes per day and 1 to 2 joints of marijuana per day. He consumed 1 to 2 alcoholic beverages per week. He had a history of crack cocaine and heroin use as per his son.
Clinical Findings
On initial presentation, he was tachypneic with a respiratory rate of 30 breaths per minute and hypothermic with a temperature of 31.5°C. His heart rate was 72, and blood pressure, 104/55 mm Hg (significantly lower than his baseline significant systolic hypertension on out-patient dialysis with systolic blood pressure ~160-200 mm Hg).
He was extremely agitated, restless, and hyperactive. He was not oriented to person, place, or time and was speaking in mostly incoherent sentences. Clinical examination was not consistent with any focal signs of infection—he did not have any nuchal rigidity or photophobia; examination of his heart, lungs, and abdomen was unremarkable; and there was no evidence of skin or soft tissue infection. He did not have any focal neurological deficits although was not cooperative with neurological examination.
Collateral was obtained from his son who indicated that the patient had mentioned feeling depressed although had never specifically expressed any suicidal ideation. He had a history of occasionally missing dialysis sessions although his last missed session was 2 weeks before presentation.
Diagnostic Focus and Assessment
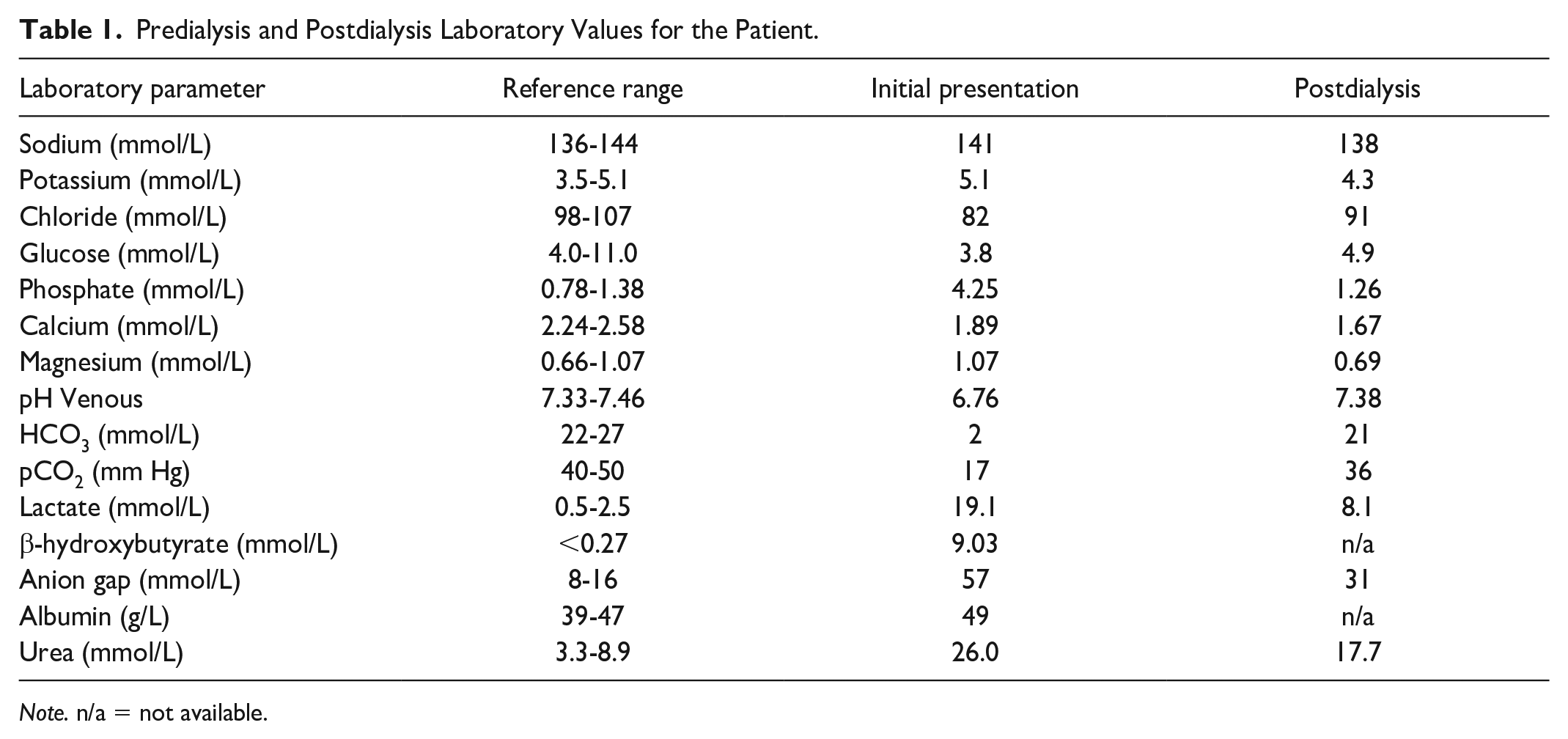
Initial laboratory investigations are summarized in Table 1.
Predialysis and Postdialysis Laboratory Values for the Patient.
Note. n/a = not available.
They were most significant for a venous blood gas with pH 6.76, pCO2 17 mm Hg, HCO3 2 mmol/L, lactate 19 mmol/L, and serum electrolytes with sodium of 141 mmol/L, potassium 5.1 mmol/L, chloride 82 mmol/L, phosphate 4.25 mmol/L, albumin 49 g/L, β-hydroxybutyrate 9.03 mmol/L, glucose 3.8 mmol/L, and urea 26.0 mmol/L. The AG (corrected for a serum albumin of 49 g/L) was 53 mmol/L. A screen for toxic alcohols (ethylene glycol, methanol, isopropranol) and ethanol were negative. The osmol gap was 23 (serum osmolality 335 mOsmol/L).
Nasopharyngeal swab for the severe acute respiratory syndrome coronavirus 2 (causing COVID-19) was positive. Further infectious workup was negative—urinalysis showed no nitrites, leukocyte esterase, or crystals; no infiltrates were seen on chest X-ray; computerized tomography scan of his head was unremarkable; and blood cultures were negative.
The working differential diagnosis was septic shock, hypovolemic shock in the context of COVID infection, cardiogenic shock, or late presentation of toxic alcohols with the presence of metabolites (glycolate/formate) and disappearance of parent alcohols given the history of substance use and concerns for depression. His single missed session of dialysis 2 weeks before was not thought to be a contributor to his presentation.
Therapeutic Focus and Assessment
Although the differential diagnosis for his presentation remained broad and required further investigations, dialysis was urgently arranged for treatment of his extreme AG metabolic acidosis. He had an initial single dialysis treatment for 4 hours with a maximum blood flow of 350 mL/min via his central venous catheter and dialysate flow of 500 mL/min; his bicarbonate dialysate was 38 mEq/L; and there was no ultrafiltration. The filter was a Revaclear 300 dialyzer (Baxter International Inc., Deerfield, IL).
Post-HD labs (~10 hours after presenting labs) showed significant improvement to his metabolic acidosis and serum lactate level (see Table 1)—his pH normalized to 7.38, HCO3 increased to 21 mmol/L, and lactate decreased to 8.1 mmol/L.
At presentation, due to suspicion of potential occult sepsis as the driving etiology for his altered level of consciousness, he was also started on broad-spectrum intravenous antibiotics (piperacillin-tazobactam) and completed a 7-day course.
His clinical status rapidly improved with normalization of his respiratory rate and temperature and improvement in his cognition over the following 72 hours. He was admitted for a total of 9 days until he fully recovered to his baseline functional status, and he was subsequently discharged home with follow-up on dialysis.
Follow-up and Outcomes
Given his presentation with profound metabolic acidosis, elevation in serum lactate, and the rapid resolution of these abnormalities with one 4-hour treatment of HD sparked a re-review of his electronic medical record. Despite no history of diabetes mellitus, metformin appeared on one of his dialysis medication lists; a sample from his initial laboratory investigations (before dialysis) was sent to a reference laboratory (National Medical Services labs in Pennsylvania) for analysis of the plasma metformin level by high-performance liquid chromatography. This eventually resulted in a level of 60 mcg/mL (therapeutic range 1-2 mcg/mL). A repeat metformin level assessment was not performed after dialysis given there was no clear history of metformin toxicity, and the level sent from his presentation was pending.
The patient was subsequently followed up on a regular basis during his dialysis sessions. He stated that he had never heard of the medication metformin, and there was no history of him filling a prescription at his pharmacy. His home medications were reconciled in the dialysis unit, and he did not have metformin or antivirals among the medications he took at home. Given his living situation with shared accommodations, the presumption was that he had taken medications that were prescribed to a roommate.
Discussion
Metabolic Acidosis
Metabolic acidosis is a common finding in hospitalized patients and generally classified by its association with a high or normal AG, with the AG calculated as follows: AG = Na+ − (Cl− + HCO3−). The former is termed HAGMA, and the latter, normal-anion-gap metabolic acidosis.
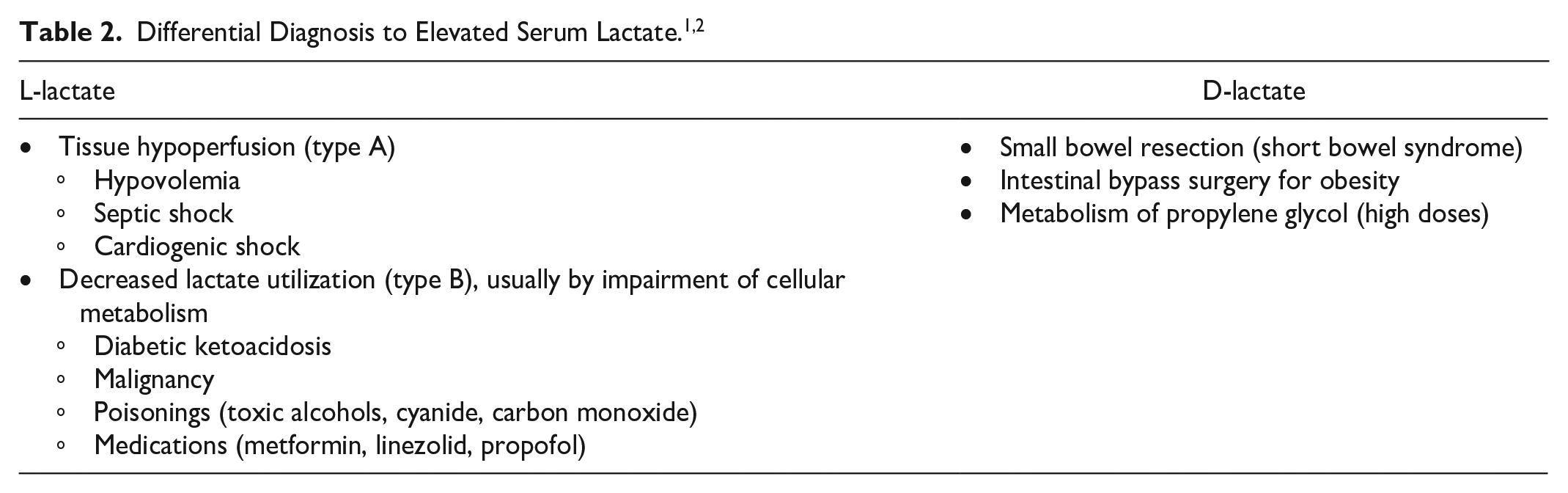
Numerous mnemonics exist for the differential diagnosis of HAGMA—the most contemporary is “GOLD MARK” as published by Mehta et al 3 to represent glycols (ethylene and propylene), oxoproline, L-lactate, D-lactate, methanol, aspirin, renal failure, and ketoacidosis.
Our patient had multiple biochemical derangements that were thought to be contributing to his presentation with a severe HAGMA. Lactic acidosis is well described to cause a disproportionate increase in the AG compared with the observed drop in serum bicarbonate—recent studies have suggested that it can be as high as 1.8:1. 4 This is likely due to Krebs cycle dysfunction and accumulation of the organic acid intermediaries. As such, the increase in serum lactate could potentially account for 34 mmol/K of the observed 57 mmol/L AG.
In nature, lactate exists in two isoforms: L-lactate and D-lactate (Table 2). The L-lactate form is by far the most abundant and significant and is the form that is specifically measured by lactate sensors in blood gas analyzers and by methods to measure lactate in the clinical laboratory. 5 Clinically, L-lactate is most commonly seen via increased production of lactate from anaerobic glycolysis because of reduced oxygen delivery to tissue cells and resultant tissue hypoxia; therefore, lactate is often used as a marker of inadequate tissue perfusion (secondary to sepsis, cardiogenic shock, etc.) in critically ill patients. This is often referred to as type A lactic acidosis. In type B lactic acidosis, there is inadequate hepatic clearance of lactate. Examples of this include liver disease, malignancy, poisonings (toxic alcohols, cyanide, carbon monoxide), and medications including paracetamol, linezolid, some antivirals, and metformin. 1

Timeline of Events.
The elevation in β-hydroxybutyric acid, related to his starvation ketosis, could account for another 9 mmol/L of his AG and the elevation in serum phosphate level for another 6 to 8 mmol/L. His markedly elevated β-hydroxybutyrate level was thought to be more likely secondary to starvation ketoacidosis than diabetic ketoacidosis given the patient’s low-normal glucose and no past history of diabetes.
Thus, overall, his metabolic acidosis was initially thought to be due to the combination of ketoacidosis (driven by starvation) and L-lactic acidosis, potentially driven by sepsis. He was treated with empiric antibiotics, and dialysis was arranged to mitigate his acidosis. Surprisingly, he had a dramatic improvement to his biochemical parameters with a single dialysis session (see Table 1), which led us to broaden our differential diagnosis. His toxicology screen was negative for toxic alcohols, but given his history of substance use and mental health challenges, a late presentation of a toxic alcohol was thought to be a remote possibility. There had also been uncertainty as to whether he was taking metformin as it had appeared on one of his out-patient dialysis medication lists, and so a serum metformin level test was requested from his presenting blood work and resulted 3 weeks later with a profoundly elevated level of 60 mcg/mL (therapeutic range 1-2 mcg/mL).
Metformin Toxicity
A meta-summary of metformin toxicity case reports was recently performed, and 242 individual cases were analyzed. 6 Metformin-associated lactic acidosis (MALA) was the most commonly reported toxicity, present in 92.6% of patients. A majority (68.6%) required kidney replacement therapy for toxin removal and correction of MALA.
The mainstay of treatment for metformin toxicity is resuscitation and supportive care. 7 However, metformin’s small size and limited protein binding confers free diffusibility through hemodialyzers (high extraction ratios) and hemofilters (high sieving coefficient), with the limiting factor for its extracorporeal elimination being its relatively large volume of distribution. 8 Therefore, there are guidelines developed by the multidisciplinary Extracorporeal Treatments In Poisoning group that recommend HD when there is a serum lactate level >20 mmol/L (and suggest HD for a lactate >15 mmol/L), a blood pH <7.0, a failure of standard therapy, end-organ damage (hepatic or renal insufficiency), or a decreased level of consciousness. 8 These guidelines also state that cessation of dialysis is indicated when lactate level is <3 mmol/L. In our patient, postdialysis lactate level was 8.1 mmol/L, but given that an elevated metformin level had not yet been proven, that his acidosis had resolved, and that alternate etiologies such as septic or cardiogenic shock remained on the differential, the lactate was further trended and had decreased to 2.8 mmol/L 10 hours later.
While metformin is seen as the best initial pharmacologic option in patients with type 2 diabetes, it must be used with caution in patients with chronic kidney disease (CKD). A systematic review of metformin in patients with type 2 diabetes and CKD found that although metformin is renally cleared, drug levels generally remain within the therapeutic range with lactate concentrations not substantially increased if used in patients with mild to moderate CKD (estimated glomerular filtration [eGFR] rates 30-60 mL/min per 1.73 m2) and with appropriate dose reductions. 9 However, its use is contraindicated for patients with eGFR <30 mL/min per 1.73 m2 and in patients on dialysis—MALA is therefore uncommon in patients with advanced CKD and ESKD. A recent study by Chen et al 10 studied the relationship between metformin use and advanced CKD (patients with eGFR <30 mL/min per 1.73 m2) in a retrospective, propensity-score matched cohort study. Using a linear regression model, they found that metformin significantly increased the risk of lactic acidosis (P = .02) as the eGFR declined in advanced CKD cases over a mean follow-up of over 600 days, even after confounder adjustment for age, sex, and comorbidities.
In summary, we discuss a unique case of MALA in a patient with ESKD on HD and no known history of diabetes or metformin ingestion. In this case, the lack of rapid access to serum metformin levels did not result in a negative outcome.
Footnotes
Ethics Approval and Consent to Participate
Written informed consent was obtained from the patient for publication of this case report.
Consent for Publication
All authors provided consent for publication.
Availability of Data and Materials
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.