Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Objective
Objective GT8.1: Dysphagia. Describe the pathophysiology and clinicopathological features of disorders presenting with dysphagia.
Competency 2: Organ System Pathology; Topic GT: Gastrointestinal Tract; Learning Goal 8: Mechanical Disorders of Bowel
Patient Presentation
An 18-year-old male presents to his primary care physician with a 6-month history of dysphagia to solid foods and retrosternal discomfort that has been unresponsive to antacids. Additionally, he reports that after eating, he has the sensation that food is “stuck” in his throat. He has a history of asthma and atopic dermatitis. He uses an albuterol inhaler as needed and takes no other medications. Physical examination is notable for mild eczema on both hands, mild upper abdominal tenderness, and an absence of thrush, ulcers, or lesions in the oral cavity.
Diagnostic Findings
Upper endoscopy is performed, revealing pallor and longitudinal furrowing within the esophageal mucosa. Additionally, concentric rings are seen within the esophagus in a pattern that mimics the cartilage rings of the trachea. During the endoscopy, biopsies are taken from the proximal, mid, and distal esophagus. The stomach and duodenum appear unremarkable.
Questions/Discussion Points
What Disorders Can Present With Dysphagia in Adults?
In evaluating a patient with dysphagia, it is important to distinguish between oropharyngeal and esophageal dysphagia. Oropharyngeal dysphagia results from abnormalities in initiating a swallow reflex, with an aberrant transfer of the food bolus from the pharynx into the esophagus. 2 This condition typically presents in older patients, with symptoms such as coughing or choking up food during swallowing, nasopharyngeal regurgitation, or aspiration. The many etiologies of oropharyngeal dysphagia can be separated into the following categories: peripheral/central nervous system abnormalities, neurodegenerative disease, muscular/neuromuscular disease, local/structural lesions, and drug induced. 3
Contrastingly, esophageal dysphagia refers to difficulty swallowing after initiating a swallow reflex, characterized by the sensation that food or liquid is obstructed during passage into the stomach. For patients with suspected esophageal dysphagia, it is important to take a thorough history about severity, progression of dysphagia, and the specific foods that produce symptoms. For example, dysphagia to solids and progressive symptoms can be caused by peptic stricture or esophageal strictures. 4 Additionally, esophageal neoplasms, metastases, or lymphoma in the surrounding lymph nodes typically present with dysphagia to solids that progresses to liquids. Dysphagia to solids with intermittent symptoms could be indicative of eosinophilic esophagitis (EoE) or esophageal webs/rings. Dysphagia to liquids and solids is typically due to an esophageal motility disorder such as achalasia or diffuse esophageal spasm. 4 Both infectious esophagitis and medication-induced esophagitis can present with esophageal dysphagia and odynophagia. 4 In contrast to dysphagia, odynophagia refers to pain with swallowing. It is typically associated with esophageal mucosal lesions and is especially prominent in infectious esophagitis. Lastly, because odynophagia occurs with swallowing, it is an important diagnostic clue for pathology in the esophagus.
Define Esophagitis. What Are the Different Subtypes?
Esophagitis is inflammation of the esophagus. It can be split into several subtypes such as infectious esophagitis (cytomegalovirus, herpes simplex virus, Candida), medication-induced esophagitis, EoE, and reflux esophagitis. Inflammation in the esophagus can result in esophageal narrowing, fibrosis, and scarring. This in turn can cause difficulty swallowing or dysphagia. As a result, dysphagia is a common presenting symptom in patients with esophagitis.
Describe the Histological Features of the Esophageal Biopsies in This Patient (Figures 1 and 2)
Histological examination of the esophageal biopsies from all 3 sites reveals an abundance of eosinophils throughout the esophagus, exceeding 100 eosinophils/high-power field in areas. Other histopathological findings include extracellular eosinophilic granules, suggestive of degranulation, and subepithelial fibrosis. 5 The subepithelial fibrosis in the lamina propria is a consequence of tissue remodeling, likely secondary to the inflammation. Additionally, reactive and regenerative changes in the epithelium are present.
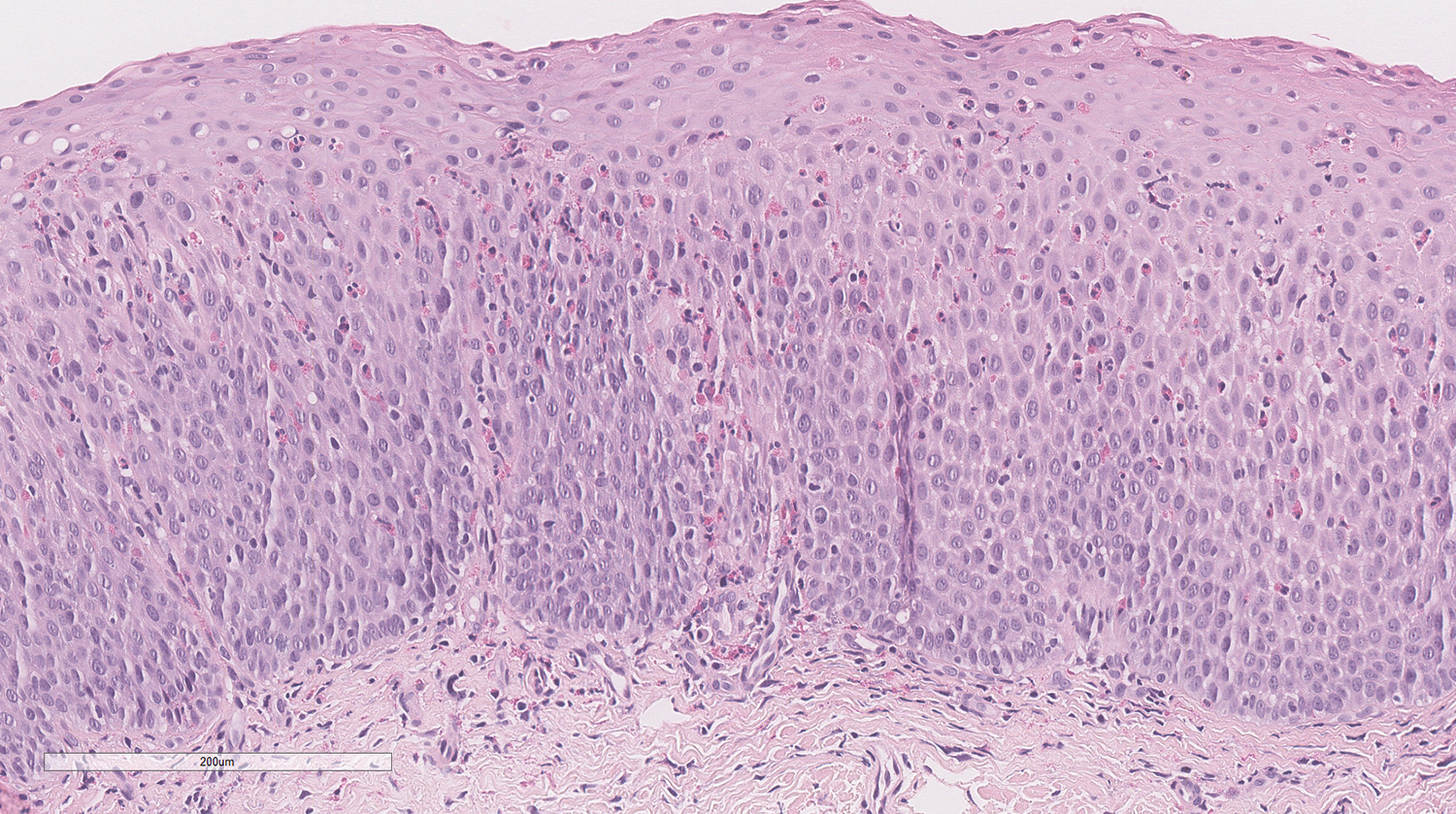
Eosinophilic esophagitis with numerous eosinophils infiltrating squamous mucosa and subepithelial fibrosis. Regenerative and reactive epithelial changes are present (hematoxylin and eosin, ×20. Bar = 200 μm).
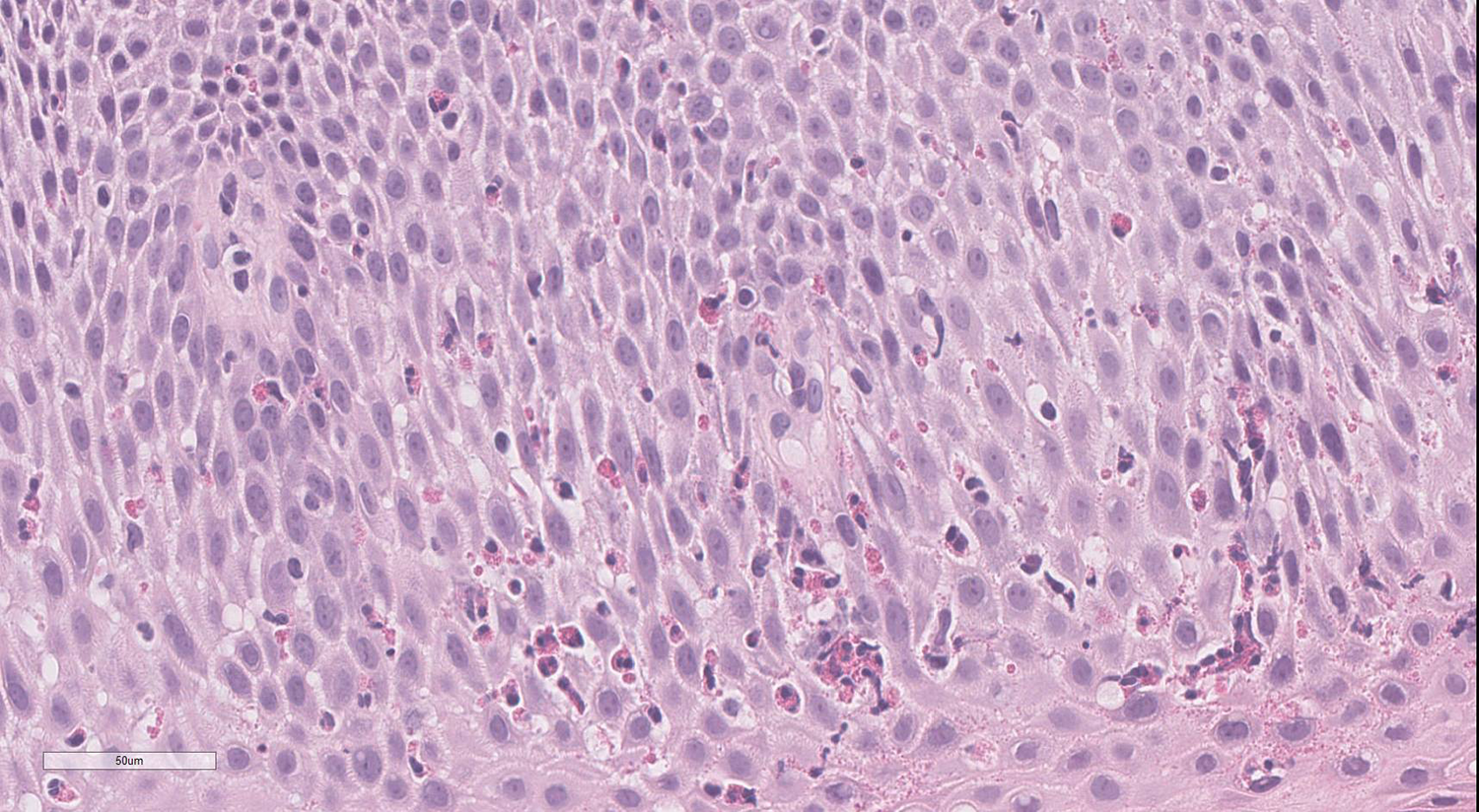
Eosinophilic esophagitis with numerous eosinophils infiltrating squamous mucosa (hematoxylin and eosin, ×40. Bar = 50 μm).
What Is the Differential Diagnosis for Esophageal Eosinophilia?
The presence of esophageal eosinophilia is not pathognomonic for a particular disease and can be seen in EoE, gastroesophageal reflux disease (GERD), hypereosinophilic syndrome, achalasia, drug hypersensitivity, connective tissue disease, food allergy, and parasitic infection. 5
It is important to take tissue samples from different portions of the esophagus. Gastroesophageal reflux disease is typically characterized by eosinophilia confined to the distal esophagus, while EoE manifests with eosinophilia in the proximal, mid, and distal esophagus. 6 Additionally, while not specific, GERD tends to have lower eosinophil counts in the distal esophagus when compared to EoE. 7
Based on the Clinical and Pathologic Features, What Is Your Diagnosis?
The patient’s diagnosis is EoE. The diagnosis of EoE is based upon 3 main criteria: symptoms of esophageal dysfunction, greater than 15 eosinophils per high-power field on esophageal biopsy, and exclusion of other disorders that could cause esophageal eosinophilia. 6 Of note, presence of comorbid atopic or allergic conditions should increase suspicion for EoE. Lastly, eosinophilic infiltration should be limited to the esophagus. 8
While there have been proposed classification systems that assess the morphological features of EoE, endoscopic findings are not required for diagnosis. Endoscopy may show linear furrows, stacked circular rings, changes in the subepithelial vascular pattern, whitish papules, strictures, or reduced esophageal diameter. 9 The sensitivity of these findings is low in EoE. The specificity is highest for linear furrow/strictures (95%), white plaques (94%), and rings (91%). 9 It is important to recognize that an absence of endoscopic findings does not rule out EoE. Microscopically, increased lymphocytes and mast cells are also often seen in the biopsy samples from patients with EoE. 8
What Are the Clinical Features of Eosinophilic Esophagitis?
Adults and teenagers with EoE are most likely to present with chronic symptoms such as retrosternal discomfort, dysphagia to solid foods, a history of food impaction, and dyspepsia that is not traditionally relieved with proton pump inhibitors (PPIs). 10 Additionally, patients may report changes in their eating habits; these include avoidance of foods that are difficult to swallow, eating smaller bites of foods, increased chewing for each bite of food, or vomiting after meals. Although rare, a complication of EoE in adults and children is spontaneous esophageal perforation. 10 Children with EoE are likely to present with failure to thrive, vomiting after meals, refusal of food, weight loss, and abdominal pain. 9
What Radiographic and Lab Findings May Be Observed With Eosinophilic Esophagitis?
Radiographic and laboratory findings may support the diagnosis but are not part of the diagnostic criteria. Barium studies can be used to detail the length and diameter of esophageal strictures. Additionally, they can show esophageal luminal narrowing that is less evident on endoscopy. 10 Patients with EoE may have elevated IgE levels and peripheral eosinophilia, but neither is required for diagnosis. 11
What Is the Pathophysiology of Eosinophilic Esophagitis?
The pathophysiology of EoE is not entirely known. However, it has been postulated that a combination of host immune system dysregulation, genetic, and environmental factors contribute to the disease process.
Host immune system dysfunction is likely the predominant underlying mechanism for patients with EoE, as they are more likely to have conditions such as asthma, food allergies, allergic rhinitis, and atopic dermatitis. This is supported by the upregulation of adaptive immunity, through T helper type 2 cells, which stimulate cytokines such as interleukin 5 (IL-5) and IL-13. Of note, IL-5 is involved in the maturation of eosinophils in the bone marrow, the recruitment of mucosal esophageal eosinophils, and mediation of tissue remodeling. 12 Interleukin 13 is also implicated in other atopic diseases such as asthma and allergic rhinitis. It is involved in eosinophil chemotaxis, collagen deposition, goblet cell hyperplasia, and increasing smooth muscle contractility. 12 Additionally, IL-13 is involved in the endoscopically identified esophageal changes associated with EoE by driving eosinophil-activating cytokines (such as eotaxin 3), which promote collagen deposition and angiogenesis and induce tissue remodeling. 12 Lastly, while the role of mast cells in EoE is unknown, the condition is characterized by an increased number of mast cells in the esophagus. It has been suggested that mast cells may increase esophageal smooth muscle mass and remodeling via transforming growth factor β1. 12
What Is the Treatment and Prognosis of Eosinophilic Esophagitis?
The main treatments for EoE include medication or dietary modification. Therapy is aimed toward improvement in symptoms and reduction in esophageal inflammation. Additionally, clinicians may also administer therapy for prophylaxis of lamina propria fibrosis, which is believed to play a role in the development of dysphagia. Dietary changes can be used as initial therapy in both pediatric and adult EoE. The 3 strategies for dietary management include total elimination of all food allergens with elemental or amino acid–based formula, targeted elimination with allergy testing, or empirically removing the 6 most common foods that trigger EoE. 6 It is recommended that patients with dietary modifications undergo endoscopy with esophageal biopsies to assess response to each modification. The first-line pharmacologic therapy for EoE is 8 weeks of topical steroids. 6 Patients who do not have an improvement in symptoms or histology may require a longer course of steroids, higher dosage, dietary modification, or esophageal dilation. There is insufficient data to suggest use of mast cell stabilizers or leukotriene inhibitors in the treatment of EoE. 6 Several studies have shown that EoE is a chronic disease and that symptoms recur if medication or dietary management is stopped.
Teaching Points
Oropharyngeal dysphagia results from abnormalities in initiating the swallow reflex, whereas esophageal dysphagia refers to difficulty swallowing after initiating the swallow reflex. Dysphagia to solids and progressive symptoms can be caused by peptic stricture or esophageal strictures. Dysphagia to solids with intermittent symptoms can be indicative of EoE or esophageal webs/rings. Dysphagia to liquids and solids is typically due to an esophageal motility disorder such as achalasia or diffuse esophageal spasm. Infectious esophagitis and medication-induced esophagitis present with esophageal dysphagia and odynophagia. Esophageal eosinophilia is seen in several diseases and is not pathognomonic for a particular condition. The diagnostic criteria of EoE include symptoms of esophageal dysfunction, greater than 15 eosinophils per high-power field on esophageal biopsy, and exclusion of other disorders that could cause esophageal eosinophilia. Patients with EoE often have comorbid atopic or allergic conditions. Histologic features of EoE include at least 15 eosinophils per high-power field, eosinophils in the superficial layers of the epithelium, or accumulations of intraepithelial eosinophils that may form microabscesses, frequently accompanied by reactive/regenerative changes in the epithelium. Both IL-5 and IL-13 likely play a key role in the pathophysiology of EoE.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The article processing fee for this article was funded by an Open Access Award given by the Society of ‘67, which supports the mission of the Association of Pathology Chairs to produce the next generation of outstanding investigators and educational scholars in the field of pathology. This award helps to promote the publication of high-quality original scholarship in Academic Pathology by authors at an early stage of academic development.
