Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.
Keywords
Primary Objective
Objective GT3.1: Precursors to Bowel Carcinoma. Discuss the precursor lesions, risk factors, and hereditary cancer syndromes that lead to GI neoplasia.
Competency 2: Organ system pathology; Topic GT: Gastrointestinal Tract; Learning Goal 3: Gastrointestinal Neoplasia
Secondary Objective
Objective GT8.1: Dysphagia. Describe the pathophysiology and clinicopathological features of disorders presenting with dysphagia.
Competency 2: Organ system pathology; Topic GT: Gastrointestinal Tract; Learning Goal 8: Mechanical Disorders of Bowel
Patient Presentation
A 40-year-old white male with a 10-year history of acid reflux presents to his primary care provider with concern due to the increasing severity and frequency of his acid reflux symptoms. The patient describes these symptoms as a burning in his upper and middle chest, as well as the development of a sour taste in his mouth over the last several months. The patient’s history reflects that he has experienced the symptoms of acid reflux intermittently since his 20s, with more consistent symptoms over the last 10 years. The patient further describes his symptoms as occurring 30 minutes after eating and lasting up to 90 minutes, as well as occurring at night. His attempts to alleviate his symptoms include sleeping propped up on pillows and taking ranitidine daily and over-the-counter antacids as needed, but these measures are only providing temporary relief. At times, he states he has difficulty swallowing. The patient’s medical history is significant for weight gain over the last 4 years leading to a body mass index of 32.3 kg/m2. He has had no prior surgeries or other significant health concerns and no family history of malignancy. He states he does not use tobacco or drugs and that he consumes 1 to 2 glasses of wine on the weekend. He does not have any other constitutional symptoms such as fever, fatigue, chills, or pain and states he is not experiencing dysphagia or weight loss. The patient’s diet is notable for including fried food, chocolate, alcohol several times per week, 2 to 3 cups of coffee and orange juice every morning, and 4 diet sodas a day.
Diagnostic Findings: Part 1
The patient’s weight is 225 lbs. His height is 5 feet 10 inches. The patient’s vital signs showed a blood pressure of 127/76 mm Hg, a heart rate of 72 beats per minute, a respiratory rate of 16 breaths per minute, an oxygen saturation of 98% on room air, and a temperature of 98.4°F. The remainder of the examination was unremarkable.
Questions/Discussion Points, Part 1
What Is the Differential Diagnosis Based on the History and Physical Examination?
The differential diagnosis of increasing chest pain, worsening symptoms of acid reflux, and a sour taste include gastroesophageal reflux disease (GERD), esophagitis, esophageal webs, achalasia, esophageal motility disorder, Barrett esophagus, or gastritis. Other considerations include conditions that would explain the patient’s chest pain such as atherosclerotic heart disease.
What Is the Differential Diagnosis for Dysphagia?
Dysphagia refers to difficulty swallowing. Patients sometimes state they have the sensation of food being stuck in their throat or chest. The differential includes esophageal disorders and external esophageal disorders that compress the esophagus. Intrinsic esophageal disorders to consider include achalasia, esophageal webs and diverticulum, esophageal spasms, esophageal strictures, esophageal cancer, foreign bodies, GERD, scleroderma, and radiation therapy to the esophagus. External compression of the esophagus may be seen with large goiters, aortic aneurysm, laryngeal carcinoma, and lung cancer. Neurologic disorders including multiple sclerosis, amyotrophic lateral sclerosis, and myotonic dystrophy may lead to dysphagia.
Diagnostic Findings: Part 2
A complete blood count, comprehensive metabolic panel, and serum troponin level were normal. Esophagogastroduodenoscopy demonstrated narrow strips of salmon-colored mucosa extending from the gastroesophageal junction upward into the tan white squamous epithelium. A biopsy was performed (Figure 1).
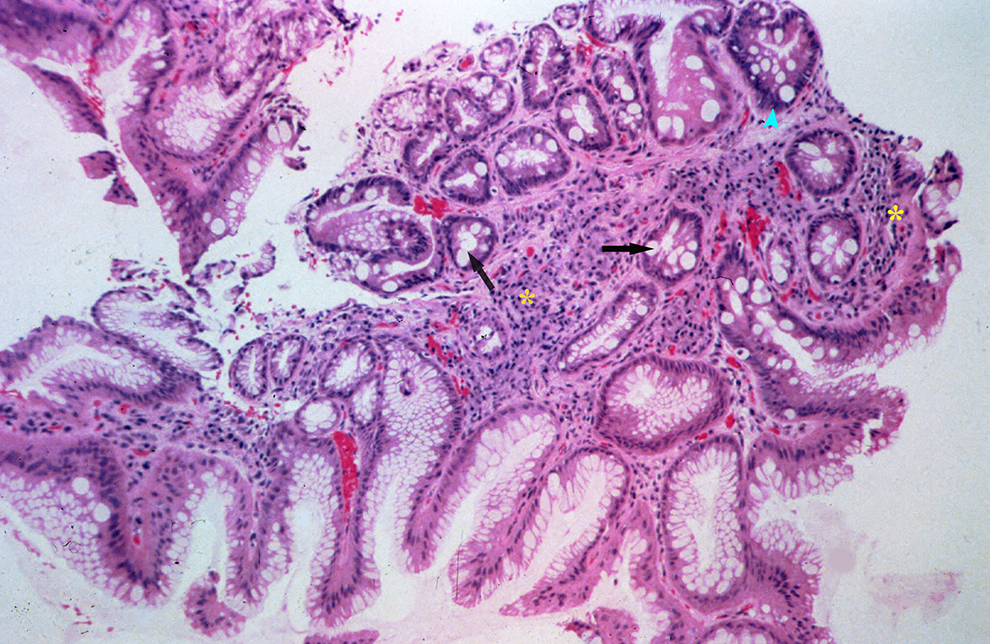
Esophageal mucosa with intestinal metaplasia. Columnar epithelium-forming glands with interspersed goblet cells (black arrows) are present. A chronic inflammatory infiltrate (asterisks) is present in the lamina propria. Hyperchromatic nuclei without loss or polarity are present in several glands (blue arrowhead; H&E, intermediate magnification). Note: This figure was obtained during the scope of US government employment for Dr Conran.
Questions/Discussion Points, Part 2
What Entities Are in the Differential Diagnosis Based on the Above Laboratory Studies?
Normal serum troponin levels allowed the differential diagnosis of myocardial infarction to be eliminated. Based on the normal laboratory values, the differential diagnoses of esophagitis, esophageal webs, achalasia, esophageal motility disorder, Barrett esophagus, and gastritis are still possibilities.
Describe the Biopsy Findings From the Esophagogastroduodenoscopy?
The biopsy reveals esophageal intestinal metaplasia characterized by intestinalized mucosa consisting of columnar and goblet cells. There is a prominent lymphocytic infiltrate in the laminal propria. Hyperchromatic nuclei without loss of polarity to the basement membrane are also present.
What Entities Are in the Differential Diagnosis Based on the Esophagogastroduodenoscopy and Biopsy?
The findings indicate a differential diagnosis of glandular metaplasia (Barrett esophagus) and severe GERD. Barrett esophagus is characterized by the replacement of normal nonkeratinized squamous epithelium with abnormal columnar epithelium and goblet cells. The salmon-colored tissue extending upward is representative of the intestinal columnar epithelium extending into the lighter colored, normal squamous epithelium of the esophagus. 1
What Is the Diagnosis Based on the Clinical Scenario?
The increasing severity and frequency of the patient’s GERD disease symptoms, along with a sour taste in his mouth, and the refractory nature of his symptoms to medications are consistent with the diagnosis of Barrett esophagus. 2 This diagnosis is supported by the endoscopy and biopsy findings, which shows intestinal metaplasia. Barrett esophagus is estimated to occur in up to 10% of patients with chronic GERD. 1
What Is Barrett Esophagus? How Does It Differ From Gastroesophageal Reflux Disease?
Barrett esophagus is a condition characterized by the development of intestinal metaplasia in the esophagus. Metaplastic columnar epithelium with goblet cells is present in the esophageal mucosa. 3 –5 Barrett esophagus is due to chronic inflammation that results from GERD. Gastroesophageal reflux disease, known as GERD, is a condition that is characterized by erythema of the mucosa of the esophagus, near the gastroesophageal junction. Typically, hyperemia is the only histological change seen in mild GERD. With more advanced GERD, hyperplasia and elongation of the papilla in the lamina propria along with scattered intramucosal eosinophils may be present. 1 Barrett esophagus and GERD share many clinical symptoms. Patients with either condition typically exhibit the symptoms of heartburn and difficulty swallowing, as well as possible chest pain and sour-tasting regurgitation. Classically, these symptoms are worse after a fatty meal. Gastroesophageal reflux disease and Barrett esophagus do not usually differ in clinical symptoms, with Barrett esophagus instead developing from a chronic course of GERD. 6 Diagnosis of Barrett esophagus is usually confirmed with endoscopy and multiple biopsies to differentiate the condition from both GERD and esophageal adenocarcinoma. 1 Studies suggest the transformation of nonkeratinized squamous epithelium into the intestinal epithelium is a protective mechanism to prevent the esophageal lining from being damaged by gastric acid. 3
Barrett esophagus is considered a premalignant condition, and therefore, frequent monitoring is recommended to detect the development of possible adenocarcinoma. 4 Esophageal adenocarcinoma occurs in 0.2% to 1% of individuals with Barrett esophagus. 1 Current research indicates that biopsies should be tested for the presence of a TP53 mutation that has been shown to be mutated in the vast majority of esophageal adenocarcinomas and a potential biomarker for the progression of Barrett esophagus to adenocarcinoma. 3 Barrett esophagus is differentiated from esophageal adenocarcinoma by the presence of invasion in esophageal adenocarcinoma, while mucosal dysplasia is the finding in Barrett esophagus. 6
What Are the Possible Complications of Barrett Esophagus?
The most concerning complication of Barrett esophagus is its progression to esophageal adenocarcinoma, though this complication only occurs in approximately 0.5% of patients with Barrett esophagus. 1 This complication is particularly concerning considering the 5-year survival rate is estimated to be only 25%. 1 This poor prognosis is attributed to the adenocarcinoma being diagnosed at a late stage. Symptoms of Barrett esophagus that have progressed to esophageal adenocarcinoma include dysphagia, weight loss, vomiting, and chest pain. These symptoms typically do not present until the adenocarcinoma has metastasized to local lymph nodes, which helps explain why it is not detected until late stage. 4
What Are Different Hereditary Syndromes Associated With Barrett Esophagus or Gastroesophageal Reflux Disease?
Due to the increasing worldwide occurrence of esophageal adenocarcinoma and the tendency for its late detection, research is currently working toward understanding any hereditary or other predisposing factors associated with its development. 4 As Barrett esophagus is considered a premalignant change that can be seen prior to the development of esophageal adenocarcinoma, research has been done to detect the prevalence of familial Barrett esophagus. One such study found that the occurrence of Barrett esophagus was 18% among patients who had a relative who was also diagnosed with Barrett esophagus, compared to the 2% risk of Barrett esophagus among the common population. 4 Studies also noted the increased risk of Barrett esophagus among patients with relatives diagnosed with esophageal adenocarcinoma. 4 These findings suggest that there is a possibility of underlying hereditary mutations playing a role in the development of Barrett esophagus, as well as progression into esophageal adenocarcinoma; however, it is important to note that these instances of familial Barrett esophagus are very rare. 4
How Is Barrett Esophagus and Its Complications Treated?
In addition to familial mutations, research is being conducted to determine what somatic mutations, or biomarkers, indicate a patient with Barrett esophagus is at high risk for developing adenocarcinoma versus low risk. 4 Such research is largely due to the growing initiative to practice preventative medicine and to diagnose the cases of Barrett esophagus that have malignant potential before they become malignant. This is especially important when considering the treatment plan for individual patients. The current standard of care for patients with Barrett esophagus is frequent endoscopy surveillance with biopsy. Multiple biopsies are performed at different levels to evaluate for areas of dysplasia. While this surveillance helps detect the progression to adenocarcinoma, its shortcomings include the associated time and cost of the procedure for both the patient and health-care system, as well as the potential for a spot biopsy to miss an area of dysplastic tissue. 4 Patients whose biopsies contain dysplasia are treated with radiofrequency ablation. 4 Research is currently being conducted with hope of establishing biomarkers that would have the potential to not only predict which patients are likely to go on to develop adenocarcinoma but also prevent patients without malignant potential from undergoing unnecessary procedures. 4
Studies have shown that 2 potential pathways exist for the progression of GERD to Barrett esophagus and, subsequently, to esophageal adenocarcinoma. The traditional pathway is characterized by an early mutation resulting in TP53 inactivation, followed by p16 inactivation, and finally, an activation of the ERBB2 oncogene. 3 An alternate pathway involves a TP53 mutation followed by a whole-genome doubling that leads to genomic instability and oncogene activation, 3,7 It has been suggested that the genome-doubling pathway allows the development of adenocarcinoma from Barrett esophagus to occur much more rapidly than the traditional pathway and helps explain why surveillance biopsy is not always able to detect the progression of metaplasia to dysplasia. 3,7
What Other Risk Factors Exist?
Gastroesophageal reflux disease and its progression to Barrett esophagus have well-established risk factors unrelated to hereditary conditions. Factors that place patients at risk for GERD include obesity, medications, and/or conditions that result in reduced tone of the lower esophageal sphincter, hiatal hernias, pregnancy, conditions that impact connective tissues, such as scleroderma, and smoking. 8 Diet is another important consideration for the development of GERD, as studies have shown that certain foods can trigger and aggravate GERD symptoms. 8 As Barrett esophagus is a progression of GERD, the previously mentioned GERD risk factors would also predispose an individual to developing Barrett esophagus. In addition, Barrett esophagus has been shown to be more prevalent in males, those with Caucasian ancestry, those with central obesity, smoking, advanced age, and those with a family history. 1,9
Teaching Points
Barrett esophagus should be considered in patients presenting with worsening symptoms of GERD, sour regurgitation, and symptoms refractory to treatment (Table 1). Patients with acid reflux and Barrett esophagus may at times present with dysphagia due to stricture formation. Other mechanisms for dysphagia include obstruction of the esophageal lumen, esophageal motor disorders, neurologic disorders, and external esophageal compression. Diagnosis of Barrett esophagus is based on the histological findings of intestinal metaplasia in the esophageal mucosa. Gastroesophageal reflux disease is a precursor to the development of Barrett esophagus and is characterized by inappropriate relaxation of the lower esophageal sphincter that allows gastric contents to reflux back into the esophagus. A concerning complication of Barrett esophagus is the development of esophageal adenocarcinoma. It is thought that there may be hereditary factors involved in some cases of Barrett esophagus, though familial associations are very rare and the exact inherited mutations have yet to be elucidated. Two pathways of mutation have been established for the progression of Barrett esophagus to adenocarcinoma. The traditional pathway involves the mutation of TP53, p16, and subsequently the ERBB2 oncogene. The genome-doubling pathway begins with a p53 mutation and subsequent genome duplication.
Signs and Symptoms of Barrett Esophagus (1, 2, 8, 9).

Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
