Abstract
Esophageal perforations may recur, and/or develop a mature fistulous tract to the pleura, despite prompt surgical management. Treatment of a chronic esophageal fistula is challenging and may require multiple reoperations. We describe the closure of a controlled, chronic, benign esophagopleural fistula using an Amplatzer occluder and sealed with a liquid copolymer after failed open repair. At 3 years postprocedure, the patient has no further recurrence or complication associated with the repair. Amplatzer plugs and occluders, designed for endovascular and cardiac procedures, are increasingly used off-label for the treatment of complex, recurrent, or otherwise difficult to manage bronchopulmonary, pleural, and esophageal fistulae.
Case Presentation
The patient is a 59-year-old woman, current smoker, otherwise healthy, who presented to an outside institution 1 year prior with a spontaneous esophageal perforation. Athough she did have a remote history of alcohol abuse, she denied any recent emesis or retching. She presented several days after the onset of symptoms, and consequently primary repair of the perforation was not attempted. The perforation was accessed via a right thoracotomy, treated with wide drainage, and healed over the subsequent 2 months with nutritional support and a prolonged hospital course.
Four months later, she presented with recurrent fever and chest pain. A barium esophagogram demonstrated extravasation of contrast into the right pleural space (Figure 1). Again, right thoracotomy was performed, and primary repair of the perforation was undertaken. However, the fistula recurred and failed to close over the following weeks, despite adequate drainage and nutritional support with jejunal tube feeding.
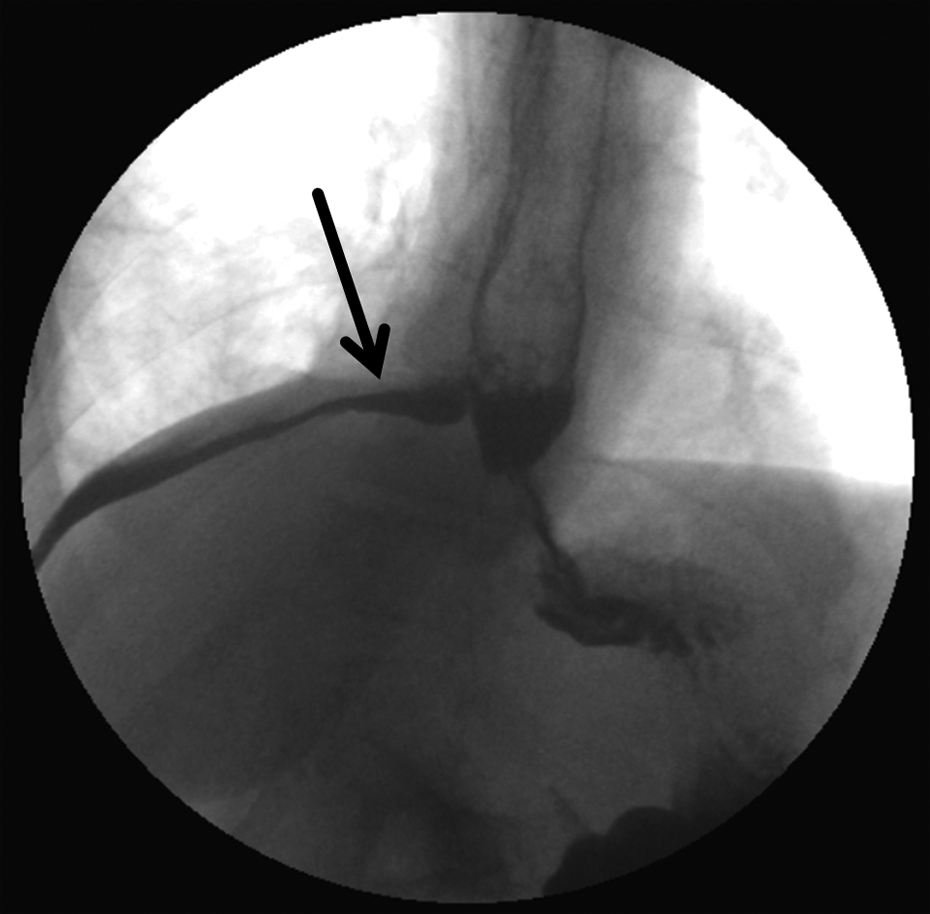
The esophagopleural fistula is demonstrated on barium swallow with free extravasation of oral contrast through a defect in the esophagus into the right pleural space tracking just above the diaphragm (arrow).
The patient was subsequently transferred to our institution for further management. Given that the fistula was chronic and failed to close after attempted primary repair and prolonged drainage, we elected to place a self-expanding, fully covered metal stent (23 × 105 mm WallFlex; Boston Scientific, Natick, Massachusetts; Figure 2). However, the stent migrated into the stomach, despite securing the stent to the mucosa with clips. Of note, there was neither a stricture nor a mass distal to the perforation, upper endoscopic examination was otherwise normal to the duodenum, and there was no appreciable obstruction or holdup of contrast during barium esophagogram.

Upper endoscopy performed 2 months after repair and drainage demonstrating the esophageal opening of the esophagopleural fistula (black arrow, A), followed by placement of a self-expanding, covered metal stent traversing the esophageal perforation (B).
Consequently, we elected to attempt closure of the esophagopleural fistula with an Amplatzer endovascular ventricular septal defect (VSD) occluder (St Jude Medical, St Paul, Minnesota). This procedure was performed under general anesthesia in the operating room. Under endoscopic guidance, the fistulous tract was cannulated with a 0.89-mm angled hydrophilic Glidewire (Boston Scientific) advanced through the endoscope. A sizing balloon was used to determine the opening of the fistula to be 8 mm in diameter. An 8F delivery sheath (St Jude Medical) was then advanced through the tract, and a 10 mm Amplatzer VSD occluder was then deployed across the fistula using both fluoroscopy and direct endoscopic visualization. An ethylene vinyl alcohol copolymer embolic system (Onyx liquid embolic system; eV3 endovascular, Plymouth, Minnesota) was then delivered into the deployed Amplatzer device to provide immediate sealing (Figure 3). At the completion of the procedure, contrast injection into the esophagus demonstrated complete closure of the esophagopleural fistula.

Successful endoscopic closure of the esophagopleural fistula (A) using a 10-mm Amplatzer ventricular septal defect (VSD) closure device (B). Onyx copolymer liquid embolization was injected inside the VSD closure device after deployment to provide immediate sealing (C).
The patient was discharged home 1 week after endoscopic repair tolerating a regular soft diet. Serial barium esophagograms performed at 1, 2, and 9 weeks after endoscopic repair demonstrated the closure device in stable position with no extraluminal extravasation of contrast (Figure 4). At 3 years postrepair, the patient had no further recurrences or complications. She was tolerating a regular, unmodified diet with a stable body mass index of 22. She reported no respiratory symptoms such as chronic cough or recurrent pneumonia and denied any symptoms of dysphagia, odynophagia, or reflux.

Barium swallow contrast study after endoscopic repair of esophagopleural fistula demonstrating excellent sealing of the esophageal defect without any extravasation of contrast into the right pleural space. Before oral contrast bolus, the Amplatzer device can be seen in stable position in the distal esophageal wall (arrow, A). Unimpaired flow of oral contrast through the esophagus into the stomach with no extraluminal extravasation (B).
Discussion and Review of Literature
The management of esophageal perforations is often complex. Perforations may be associated with a variety of underlying diseases, such as achalasia or esophageal cancer. The perforation may occur secondary to an endoscopic procedure, in which case the diagnosis may be made promptly and treatment undertaken before mediastinitis or peritonitis develops. On the other hand, patients with esophageal perforations due to retching (Boerhaave syndrome) often present in a delayed fashion. Drug or alcohol use is often associated with this condition, and patients may not present for medical care for several days. Furthermore, these patients may be misdiagnosed with an aspiration pneumonia and empyema, and the correct diagnosis may not be established until conservative treatment fails. In these situations, successful treatment by direct surgical repair is difficult. These patients may be malnourished and in septic shock, and tissue inflammation and edema may contribute to breakdown of suture lines.
In the present case, the initial presentation of esophageal perforation occurred several days after the onset of symptoms, and primary repair was not attempted. The fistula recurred, despite drainage, and, as expected, a subsequent attempt to directly repair the fistula also failed. At the time of transfer to our institution, a mature fistulous tract had formed between the esophagus and the pleural space. Given the chronic nature of the fistula, and that two prior thoracotomies were unsuccessful, we felt that any additional attempt to directly repair the fistula surgically would likely fail.
The surgical options that remain in this situation are limited. Avoiding esophagectomy for benign disease, proximal diversion with a cervical esophagostomy has been described. 1,2 Alternatively, an open thoracostomy window could be performed, with the hope that the fistula would close similar to bronchopleural fistulae following pulmonary resection. 1,2 However, both of these options carry a significant impact on the patient’s quality of life with the need for prolonged nursing care and the possibility that the fistula may persist, despite aggressive measures.
Within this context, endoscopic closure was considered. Closure of the fistula was successful with the Amplatzer VSD device coupled with Onyx sealing, and the patient was discharged soon thereafter to her normal activities, with no significant dietary restrictions, and the patient had no further complications or recurrence at 3 years postprocedure.
The literature regarding endoscopic management of esophageal perforations and airway fistulae is primarily descriptive but increasingly common. A 2015, 12-year multicenter retrospective analysis of over 2500 patients in England examining the management and outcomes of esophageal perforation did note a significant increase in supportive management and a reduction in surgical management over time. 3 Case reports and small case series describing the use of stents, clips, and closure devices for esophageal perforations have all been documented. 4 -9 However, these series often include acute and chronic perforations from a wide variety of causes, including traumatic, malignant, iatrogenic, and spontaneous. Consequently, it is difficult to make broad conclusions from these series regarding the optimal management of esophageal perforation. A series of 9 patients with esophagopleural fistulas resulting from esophageal and lung malignancy, as well as a postoperative empyema for Boerhaave syndrome, reported technical success in 8 of the 9 patients treated with covered, expandable metallic stents. However the rate of fistula persistence or reopening was high (occurring in 5 patients, nearly 56%). 10
The first case report describing the successful use of an Amplatzer occluder to close an esophagopleural fistula was reported in 2010. 11 In that case, the device was implanted 7 days following surgical resection of an esophageal diverticulum, and a liquid polymer was also used to provide immediate sealing. In 2013, a second case report described a patient who developed an esophagopleural fistula following a left pneumonectomy. 12 However, the device failed due to migration, and the fistula was subsequently successfully treated surgically. Of note, a sealing polymer was not used concomitantly with the Amplatzer occluder in that case. Published the same year, however, was a second successful report of an Amplatzer device, in this case a vascular plug was used to treat a benign esophageal fistula which occurred 13 months postesophagectomy between the gastric conduit neoesophagus and the right upper lobe bronchus. 13 The authors report a 12-month successful follow-up during which time the patient remained asymptomatic with resolution of her recurrent right-sided pneumonia.
The use of an Amplatzer device to close a bronchial fistula was first reported in 2006 in the case of a persistent, nonmalignant esophagorespiratoy fistula. 15 Similarly, Amplatzer vascular plugs and occluders for bronchopleural and tracheopleural fistulae following pneumonectomy or lobectomy has been described in multiple cases. 16 -20 In a series of 10 patients with 11 postlung resection bronchopleural fistulae published in 2011, successful repair was reported in 9 of the 10 patients with a median follow-up of 9 months. 21 A second series of 5 patients treated by the same group reported successful Amplatzer closure in all bronchopleural fistulae, also with 9-month mean follow-up. 22 Similar to our use of a liquid embolic system used in conjunction with the Amplatzer device for an esophagopleural fistula, the use of autologous blood and glue embolization, 23 Bioglue (CryoLife Inc. Kennesaw, Georgia) surgical adhesive, 24 cyanoacrylate glue, and Onxy liquid embolic system 25 have all been used in conjunction with Amplatzer devices in the endobronchial closure of postlung resection bronchopleural fistulae.
The long-term efficacy of endobronchial Amplatzer occluders and vascular plugs was examined in a series of 31 patients with postlung-resection bronchopleural fistula, with a mean follow-up of 17.6 months. Immediate sealing was attained in all but 1 patient, no patient died due to fistula recurrence, and within 6 months of Amplatzer placement, all devices in living patients were covered with granulation tissue. 26
In summary, we present the second case report of successful closure of a native esophagopleural fistula using an Amplatzer device coupled with a sealing copolymer to support immediate and complete closure. The fistula was chronic and had recurred, despite previous attempts at open surgical treatment. When these devices are utilized for endovascular applications, endothelialization and thrombosis eventually lead to complete closure. Since complete endothelialization, or in this case epithelialization, is less likely to occur in this setting, we recommend that the use of an Amplatzer device be in conjunction with a sealing polymer in refractory cases of esophageal perforation. In the case of bronchopleural fistula repair with Amplatzer devices, granulation and incorporation of the devices have been demonstrated on subsequent bronchoscopy, and the use of concomitant sealing polymers has varied. Further experience is necessary to determine whether this approach should be considered in a broader range of patients when surgical correction is not feasible. A summary of reports using Amplatzer devices for esophagopleural and bronchopleural with success rates and mean follow-up can be found in Table 1.
Case reports and Case Series Using Various Amplatzer Devices for the Closure of Esophagopleural and Bronchopleural Fistulae With Reported Success Rates and Mean Follow-Up.

* B. Braun, Melsungen AG, Germany
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
