Abstract


Introduction:
This is a surgical video demonstrating the procedure of robotic rectopexy for rectal prolapse.1,3 A 16-year-old female presented with a chief concern of severe rectal prolapse, attributed to child abuse during her teenage years. The prolapse extended up to 2 feet during bowel movements and straining. On physical examination, significant rectal prolapse was confirmed even with minimal straining. Given the severity of prolapse, a decision was made to perform a robotic rectopexy using polypropylene mesh.
Materials and Methods:
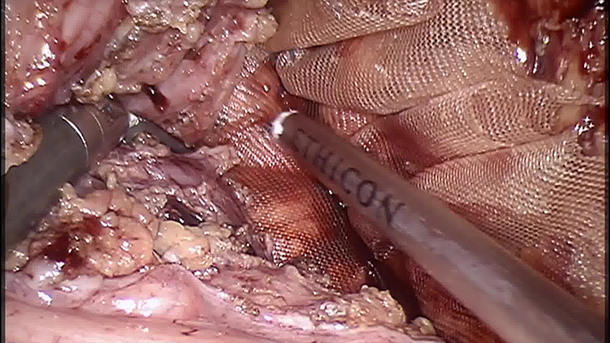
After standard surgical preparation, the patient was positioned in lithotomy. A 12 mm robotic trocar was placed through the umbilicus, and two 5 mm trocars were inserted in the right and left flanks. Tacking sutures were used to retract the fallopian tubes and ovaries. The redundant sigmoid colon was retracted laterally in reverse Trendelenburg position, exposing the pouch of Douglas. The peritoneum over the anterior rectum was incised bilaterally and inferiorly, fully mobilizing the rectum. A presacral plane was developed. Polypropylene mesh was anchored to the sacrum using Ethicon ProTack device and 2-0 Ethibond sutures, securing it to the rectum posteriorly and laterally. The peritoneum was closed with a running 3-0 Vicryl suture. Successful rectopexy was then confirmed via proctoscopy.
Results:
At the 4-week follow-up, the patient’s incisions were well-healed, and no evidence of rectal prolapse was observed. Long-term follow-up over 6 years demonstrated excellent outcomes, with no recurrence of prolapse.
Conclusions:
Robotic rectopexy with mesh provides a safe, effective, and durable solution for treating severe rectal prolapse. This technique allows for precise dissection, secure fixation, and preservation of critical anatomical structures, resulting in excellent long-term outcomes. In a retrospective review by Hiller et al. of four pediatric patients in whom this technique was utilized, there were no episodes of recurrent prolapse, with improvement of symptoms in two of the four patients. 2
Patient Consent Statement:
The authors have received and archived patient consent for video recording/publication in advance of video recording of the procedure.
Author Disclosure Statement:
There are no commercial associations that might create a conflict of interest in connection with this video. The authors have no conflicts of interest to disclose.
Runtime of video:
4 mins 45 secs.
