Abstract
Endocrine-disrupting chemicals (EDCs) are chemicals that disrupt the normal functioning of endocrine system hormones, leading to a range of adverse health effects in humans and wildlife. Exposure to EDCs is ubiquitous and occurs through contaminated food and water, air, consumer products, and transfer from parents to offspring. Effective regulation has been challenging due to a limited understanding of EDCs’ complex and nonlinear dose-response relationships, as well as difficulty in attributing specific health effects to individual EDC exposures in real-world scenarios. Current EDC policies face limitations in terms of the diversity and complexity of EDCs, the lack of comprehensive testing requirements, and the need for more robust regulatory frameworks that consider cumulative and mixture effects of EDCs. Understanding these aspects is crucial for developing effective and evidence-based EDC policies that can safeguard public health and the environment.
Keywords
Social Media Post
The latest EDC science highlights the need for stronger regulations to protect human health and the environment. From latent effects to regrettable substitution, safe alternatives and robust oversight can work toward a healthier future!
Key Points
Endocrine-disrupting chemicals (EDCs) disturb the endocrine system and pose a widespread human health problem.
EDCs are associated with reproductive disorders, developmental abnormalities, hormonal imbalances, metabolic disorders, and increased risk of certain cancers.
Distinctive properties of EDCs (such as nonlinear dose-response relationships and latency between exposure and observed health effects) complicate regulation.
Consensus on effective identification and regulation of EDCs remains elusive due to conflicts among governmental agencies, scientific bodies, and interest groups.
Action can be taken to improve EDC regulation by improving chemical testing requirements, promoting public awareness, encouraging federal-level initiatives, and supporting research to inform evidence-based policymaking.
Introduction
Among the many environmental factors that influence health and disease are a group of chemicals classified as endocrine-disrupting chemicals (EDCs), which perturb hormone-related processes such as hormone synthesis, metabolism, and transport (La Merrill et al., 2020; Zoeller et al., 2012). These chemicals are found in many media, including pesticides/herbicides, plastics, and personal care products (Ingerslev et al., 2003; Lambert & Skelly, 2016; Wilk et al., 2019). As such, exposure to EDCs is everywhere and occurs via ingestion, inhalation, and skin contact, making it impossible to avoid them completely (Schug et al., 2013). Scientific research has linked EDCs to reproductive and developmental abnormalities in humans and wildlife since the 1970s (Colborn et al., 1993). EDCs significantly threaten public health, and contemporary research connects them with diverse adverse endocrine-mediated health outcomes including certain cancers, neurological and cognitive impairments, metabolic syndromes, thyroid disorders, and even skeletal malformation (Boas et al., 2012; Gore et al., 2015; Modica et al., 2023; Xin et al., 2018). Additionally, vulnerable populations such as pregnant women, infants, and children are at higher risk of adverse health effects from exposure to EDCs (Gore et al., 2015), underscoring the critical periods of life when EDCs are of particular concern.
The impact of EDCs on public health is a complex and contentious issue that requires the implementation of effective policy to mitigate exposure risks. While progress has been made in identifying and regulating EDCs, regulation has been slow to materialize because of the scientific complexity of understanding EDCs, lobbying efforts, ineffective screening strategies, and limited long-term studies on the effects of EDCs on human health. With regard to lobbying, manufacturing groups spent $66 M in 2022 lobbying on legislation relating to chemical regulation and 54 interest groups filed lobbies against the failed PFAS Action Act of 2021 (OpenSecrets, 2022, 2021). EDCs are a global problem, requiring international effort: EDCs are transported in water, air, and migratory species of the food chain, thereby exposing even remote peoples and wildlife.
This review provides background on the properties of EDCs and the mechanisms by which EDCs impair health, as well as the policies in place for regulating EDCs, and provides acontemporary scientific perspective on potential ways to improve regulatory policies concerning EDCs.
Common Classes of Endocrine-Disrupting Chemicals
EDCs are often classified by chemical structure and functional groups that have similar modes of action and biological effects. Common classes of EDCs include bisphenols, phthalates, polychlorinated biphenyls (PCBs), organochlorine and organophosphate pesticides, polybrominated diphenyl ethers (PBDEs), parabens, and per- and polyfluoroalkyl substances (PFAS) (Table 1). For the sake of brevity, this review focuses on effects most commonly studied, but EDCs often act by more than one pathway and have multiple effects, some of which may not have been evaluated for a particular chemical (La Merrill et al., 2020).
Common Classes of EDCs: Use and Health Outcomes.
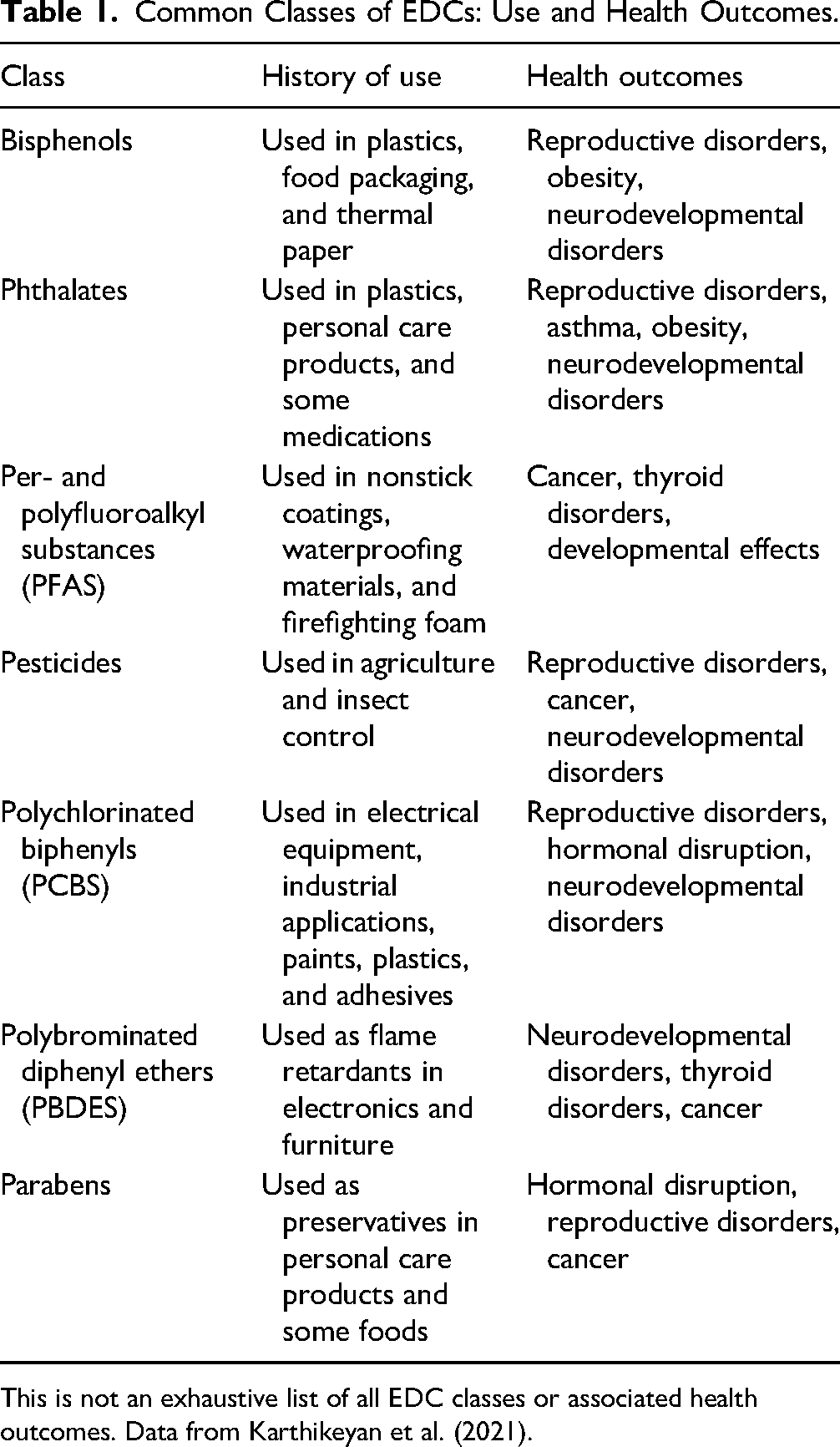
This is not an exhaustive list of all EDC classes or associated health outcomes. Data from Karthikeyan et al. (2021).
Bisphenols are present in plastics, epoxy resins, and thermal paper; these compounds are classically studied for their estrogenic effects, although they can act via other mechanisms. Bisphenol A (BPA) represents one of the best-studied classes of EDCs and has a range of endocrine-mediated health effects (Rubin, 2011; Vandenberg et al., 2019). Due to widespread scientific and public concern over the mounting health issues associated with BPA, it has been banned for use in baby bottles and other child food-contact materials by the U.S. Food and Drug Administration (FDA) and restricted by many states. As such, BPA has been replaced in many consumer products with substitute bisphenols including bisphenol S (BPS) and bisphenol F (BPF). Mounting evidence suggests these replacement bisphenols are also estrogenic and exert similar or even more harmful health effects. This is not surprising considering the structural similarities among bisphenols and the unfortunate fact that often “like replaces like” when substituting chemicals in manufacturing.
Phthalates are plasticizers found in personal care products, in food packaging, and in medical tubing such as intravenous lines. They are commonly studied in the context of androgen-mediated developmental reproductive problems although they also have other mechanisms of action. In rats, phthalate syndrome occurs when a male fetus is exposed to high levels of phthalates and is characterized by consistent reproductive tract abnormalities (Foster, 2006; Welsh et al., 2008). Evidence exists for an analogous syndrome in humans wherein prenatal exposure to phthalates in utero is associated with reduced anogenital distance (AGD), which is linked to similar abnormalities as well as impaired semen quality and reduced fertility in adulthood (Swan, 2008). Parabens, also used in personal care products as antimicrobials, are estrogenic and linked to breast cancer, reproductive toxicity, and developmental disorders (Ali & Elgoly, 2013; Hager et al., 2022; Nowak et al., 2018).
Although now banned, PCBs were once widely used for industrial purposes and as flame retardants, among other uses (US EPA, 2015). PCBs resist degradation and therefore persist in the environment to this day, remaining a relevant source of EDC exposure for humans and wildlife due to their presence in soil, water, and air and accumulation up the food chain via biomagnification (Borja et al., 2005; Burreau et al., 2004). PBDEs replaced PCBs as flame retardants used in a range of consumer products; however, this substitution represents the introduction of a new source of endocrine disruption rather than reduction (Siddiqi et al., 2003).
Many pesticides have been banned or restricted due to their EDC-like effects. The most common pesticides linked to endocrine disruption are organochlorine pesticides (such as DDT and dieldrin), organophosphate pesticides (such as chlorpyrifos), and pyrethroid insecticides (McKinlay et al., 2008; Mnif et al., 2011; Ye & Liu, 2019). Although restricted, these pesticides are still available for use in agriculture and other industries, leading to ongoing human exposure.
PFAS are a class of synthetic chemicals that have been widely used in a range of consumer and industrial products, including firefighting foams, food packaging, and heat- or water-resistant coatings. Often referred to as “forever chemicals,” PFAS are highly persistent in the environment and have been detected in the blood of humans and wildlife worldwide (Cousins et al., 2020; Giesy & Kannan, 2001; Jian et al., 2018). PFAS interfere with the function of thyroid and sex hormones and may be associated with an increased risk of certain cancers (Coperchini et al., 2021; Rickard et al., 2022; Steenland & Winquist, 2021). In the United States, several high-profile cases of PFAS contamination of drinking water have led to state-specific restrictions or bans on PFAS for certain applications. Additionally, the U.S. Environmental Protection Agency (EPA) released a proposed National Primary Drinking Water Regulation for six types of PFAS in drinking water and is considering further regulation of these compounds (US EPA, 2016).
Biological Mechanisms of Endocrine Disruption
The endocrine system comprises dozens of glands, each producing and releasing one or more hormones into the bloodstream. Hormones fall into several structural categories; of these, steroid and thyroid hormones are lipophilic and signal through intracellular nuclear receptors. This categorization is relevant to EDCs because in many cases these chemicals mimic or block endogenous (“natural”) hormones. The best-studied EDCs, which are lipophilic and share structural similarities with estradiol, have been a major focus of research that explores their effects through nuclear hormone receptors (NHRs), particularly the estrogen, androgen, and thyroid receptors (Combarnous & Nguyen, 2019; le Maire et al., 2010).
NHRs are heterogeneously distributed through different tissues throughout the body; their functions vary depending on the specific tissue wherein they bind to hormones to regulate the expression of target genes (Smith & O’Malley, 2004). For example, the estrogen receptor is found in the uterus, bone, and brain, among other organs, and its activation or suppression exerts effects unique to each tissue (Martinkovich et al., 2014; Wend et al., 2012). Similarly, the androgen receptor, thyroid hormone receptor, and other receptors also exhibit widespread distribution and exert tissue-specific effects.
The effects of endogenous hormones (i.e., people's naturally occurring hormones) can occur at extremely low doses (Vandenberg, 2014). This implies that even small exposures to EDCs, which mimic or disrupt natural hormones, can alter gene expression and cellular function; this depends on the
Furthermore, hormone actions do not always occur in a linear dose-response manner; in fact,
Moreover, the
It has been argued that EDCs may bind NHRs with lower affinity than endogenous hormones, leading to the conclusion that the risk of certain EDCs is minimal at environmentally relevant doses because it is not high enough to competitively bind the NHR (e.g., Autrup et al., 2020; Castelain & Castelain, 2012; Okubo et al., 2001). This argument lacks validity when considering that physiological hormone-receptor interactions may not align with standard toxicity testing and that certain life stages are at increased susceptibility to endocrine disruption (discussed below).
EDCs and the Developmental Origins of Health and Disease (DOHaD)
A fundamental concept in endocrinology is that during certain developmental stages of life there may be no exposure to a particular hormone (Zoeller et al., 2012). EDC researchers have recognized this, especially in the context of early life (fetus, infant). Under these circumstances,
Evidence for the DOHaD hypothesis in humans came from the case of the pharmaceutical drug diethylstilbestrol (DES), prescribed for decades in the middle of the 20th century (Newbold, 2008). While causality of an exposure and an adverse outcome can be proven in controlled laboratory animal studies, this is not possible (or ethical) in humans. DES, structurally similar to natural estradiol, was prescribed as an antimiscarriage drug (later shown not efficacious). The effects of DES on the developing fetuses were later associated with a higher incidence of reproductive tract structural abnormalities and even rare cancers in young women. The connection between DES and these disorders was not initially made because of the long latency between exposure (
The vulnerability of the fetus and infant (as well as other life stages such as puberty and menopause), together with the nonmonotonic dose-response curves described above, leads to the very real implication that
Current EDC Policy Landscape
The regulation of EDCs encompasses state, national, and international policies. Decades of policy attempts showed the challenges of regulating EDCs. Current EDC policy landscape highlights innovative initiatives at the U.S. state level. Comparing U.S. federal regulations with international organizations’ efforts in addressing EDC challenges, consider their implications for safeguarding public health and the environment against the detrimental impacts of EDCs.
EDC Regulation in the United States
In the United States, states have the power to regulate chemicals within their boundaries, allowing for faster and more stringent legislation compared to the federal government (Kassotis & Trasande, 2021). This addresses the slow response of federal agencies in identifying and addressing EDC risks; however, it also creates 50 local regulatory bodies rather than a centralized decision-making body. Some state policies have shifted focus from restricting single chemicals to addressing chemical classes that have intrinsic hazards and promoting “green chemistry” practices that minimize hazardous substances in design and production (Belliveau, 2011; Dunn, 2012; Schifano et al., 2009). Prioritizing chemical classes for regulation based on potential exposure and hazard categories is more streamlined than the costly and time-consuming individual chemical assessments required by the U.S. federal government (Schwarzman & Wilson, 2009), and regulations that encourage green chemistry are proactive instead of reactive. State-led regulations are advantageous in considering regional needs, incorporating effective international approaches, and being more responsive to concerns (Kiss, 2013; Schifano et al., 2009).
Unfortunately, relying on state regulation as a response to federal inaction has limitations. While states can enforce more rigorous safety testing, they lack the resources and comprehensive data on chemical toxicity and endocrine activity of industrial chemicals, including the safety of potential alternatives (den Braver-Sewradj et al., 2020; Kassotis et al., 2020; La Merrill et al., 2020; Vandenberg et al., 2017). Inconsistent coordination between and within states leads to regional variations in regulations and disproportionate exposure for vulnerable populations (James-Todd et al., 2012; Ruiz et al., 2017). States with EDC-heavy economic interests may have less stringent EDC regulations, as these industries may have significant economic and political influence in the state. For example, Texas is home to several oil refineries, chemical plants, and other industries that produce EDCs and has notably weaker environmental regulations compared to other states (Berman & Bui, 2001; Willyard, 2019). On the other hand, states like Washington, Minnesota, and California have adopted proactive approaches, focusing on chemical classes and promoting green chemistry (Kassotis & Trasande, 2021; Wilson et al., 2006).
At the federal level, the U.S. EPA has the authority to require manufacturers to provide information on chemicals in commerce and to restrict or ban chemicals that pose an unreasonable risk to human health or the environment. Manufacturers are responsible for safety testing chemicals before they are marketed, and for sharing that information with the EPA. The Lautenberg Act of the Toxic Substances Control Act (TSCA) requires the EPA to conduct safety reviews of all new chemicals but has been criticized for substantial limitations such as (1) grandfathering in known or suspected EDCs under the previous law, (2) allowing companies to keep chemical information confidential and withhold that information from the public, and (3) current risk assessments under the amended TSCA deviate from best practices (McPartland et al., 2022). U.S. federal regulation is further limited by a lack of data on the health effects of many chemicals and chemical mixtures, limited resources and funding for regulatory agencies, and a political climate that may prioritize industry interests over public health concerns (Krimsky, 2017; Villanueva et al., 2014; Zoeller et al., 2012). As of 2023, there are over 60,000 chemicals in commerce with 33 undergoing risk assessments by the U.S. EPA, and only one has reached the point of formal suggestions for regulation in commerce (Kassotis & Trasande, 2021; US EPA, 2023).
EDC Regulation Internationally
International policy on EDCs is primarily governed by the United Nations Environment Programme (UNEP) and the World Health Organization (WHO) under the direction of the International Programme on Chemical Safety (IPCS). The IPCS works toward synchronizing chemical safety concerns, conducting global evaluations of chemical risks, developing consensus papers on risk assessment procedures, and addressing emerging environmental health concerns worldwide. These efforts have resulted in a consensus statement from the European Commission on EDCs, covering their definition, sources of uncertainty, scientific principles for identification, and research needs (Solecki et al., 2017; Zoeller et al., 2019).
Under the EU's REACH regulation, companies must provide safety information on chemicals, and the EU has the authority to restrict or ban chemicals with significant risks (European Chemicals Agency, 2023). Additionally, the EU has banned several EDCs, including BPA and phthalates, in certain products such as baby bottles and toys. Strengths of international policy on EDCs include emphasis on collaboration and the pooling of resources and expertise from multiple countries and organizations. The IPCS database and guidelines for evaluating chemical safety provide a standardized approach to assessing the risks of EDCs (Vandenberg et al., 2016), and REACH regulation places a greater burden on companies to demonstrate the safety of their products, potentially incentivizing the development of safer alternatives. While the international policy on EDCs has its strengths, there is still room for improvement in the speed and comprehensiveness of the regulatory process.
Barriers to Effective EDC Regulation
Creating effective EDC regulatory policy is challenging for many reasons. EDCs have diverse effects on the endocrine system, and disease onset may emerge later in life or across generations, making cause-and-effect relationships difficult to establish. Moreover, nonlinear and dose-dependent effects of EDCs complicate the task of establishing safe exposure levels, which is typically the goal of risk assessment, as what may be considered a “safe” exposure level at one developmental stage of life can be extremely disruptive at another. This means there is not a one-size-fits-all answer. In the rare cases where EDCs are banned, restricted, or voluntarily removed from production, a disturbing trend has arisen wherein these chemicals are substituted with equally or more harmful compounds—hindering progress. Finally, the involvement of multiple stakeholders, including industry and public interest groups that have differing perspectives and priorities on the regulation of EDCs, makes the regulatory process more complex.
The Case of Regrettable Substitution
Perhaps one of the most glaring issues regarding the current inability of policy to protect human health in the face of increasing EDC burdens is the ease with which banned, restricted, or voluntarily removed compounds are replaced in production with structurally similar and at times
The case of regrettable substitution is mirrored in other examples such as PFAS, which are currently under intense public scrutiny as “forever chemicals” associated with aquatic toxicity and EDC-like action (Buck et al., 2011; Eggert et al., 2019; He et al., 2018; Kawabata et al., 2017; Lee et al., 2020). As PFAS are present in drinking water, there is a realistic concern about human health effects of this chemical; therefore, manufacturers have willingly phased out these chemicals even prior to state- and community-led bans and restrictions on their industrial use. As with BPA, however, new structurally similar PFAS are substituted that raise concerns about toxic and EDC-like potential (Mahoney et al., 2022). Regrettable substitutes occur throughout industries and reflect a concerning trend in U.S. health policy to minimize realistic risk in favor of industrial growth.
Conclusion
The body of scientific evidence supporting the health risks associated with EDCs is strong and growing stronger. Thousands of studies link EDC exposure to reproductive disorders, developmental abnormalities, hormonal imbalances, metabolic disorders, and increased risk of certain cancers (Boas et al., 2012; Gore et al., 2015; Modica et al., 2023; Xin et al., 2018). This evidence underscores the urgent need for policy action to address EDCs. When it comes to human health, the “innocent until proven guilty” approach to chemical manufacturing is an unacceptable policy that results in billions of dollars of associated healthcare costs and the immeasurable impairments of life quality for millions of humans (Attina et al., 2016). We’ve seen it again and again—DDT, PCBs, and now PFAS. Implementing robust federal-level regulations to restrict or ban the use of harmful EDCs, requiring stricter chemical testing requirements, and enhancing public awareness about the sources and risks of EDC exposure are crucial steps to protect human health and the environment. Currently, valuable years are lost generating evidence that any one chemical is harmful and ought to be regulated; as there are hundreds of known EDCs (and many chemicals simply haven’t been tested at all), generating the amount of health science data necessary for restrictions or all-out bans in the face of exponential substitution creates an insurmountable task for scientists in the current policy system. By prioritizing evidence-based decision-making and precautionary approaches, while incentivizing the use of safe alternatives, policymakers can mitigate the potential harm of EDCs and foster a healthier future for all. Failure to do so will ultimately result in increased morbidity and mortality.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Andrea C. Gore and Emily N. Hilz (grant numbers NIH 1R35 ES035024, NIH 1F32 ES034257).
