Abstract
Background
The American Urological Association (AUA)/Society of Urology Oncology (SUO) guidelines recommend a repeat transurethral resection of bladder tumor (TURBT) for high-risk, non-invasive (HR Ta) nonmuscle invasive bladder cancer (NMIBC) patients. The evidence base for this recommendation is weak (grade C) and fraught with methodological shortcomings, such as the lack of adjuvant intravesical Bacillus Calmette Guerin (BCG) and single-center study designs.
Objective
We sought to evaluate the effect of repeat TURBT on recurrence-free survival at a population level in HR Ta NMIBC patients who completed BCG induction therapy.
Methods
High-grade Ta NMIBC patients who underwent TURBT for a ≥5 cm tumor were identified within the SEER-Medicare database. All patients completed induction BCG and were stratified into two groups: repeat TURBT within eight weeks of initial TURBT and a group without repeat TURBT (control group). The primary endpoint was the 3-year high-risk recurrence rate.
Results
A cohort of 604 patients was identified, with 93 (15.4%) undergoing a repeat TURBT within eight weeks of initial TURBT and 511 (84.6%) without a repeat TURBT. Patient demographic and clinical characteristics were similar overall. No significant difference in the 3-year recurrence rate was noted (repeat TURBT: 20.4% vs. control group: 15.7%, p = 0.25). After adjusting for demographic and clinical characteristics, no association between repeat TURBT and 3-year high-risk recurrence was observed (HR (95% CI): 1.27 (0.76, 2.11); p = 0.36).
Conclusion
Although our study contains several major limitations, our results suggest that repeat TURBT in large volume HG Ta NMIBC treated with induction BCG therapy was not associated with improved high-risk recurrence-free survival.
Introduction
Nonmuscle invasive bladder cancer (NMIBC) is diagnosed in over 60,000 Americans every year. 1 High-risk (HR) NMIBC is characterized by the presence of carcinoma in situ (CIS), lamina propria invasion (T1), or large volume or multifocal high-grade (HG) non-invasive (Ta) disease. 2 Performing a complete visual resection and administering Bacillus Calmette-Guérin (BCG) induction and maintenance intravesical therapy is the standard of care treatment in HR NMIBC. 3 Adhering to these guidelines can prevent high-risk recurrence in approximately 60 to 70% of patients. 4 The National Comprehensive Cancer Network (NCCN) additionally recommends early repeat TURBT within six weeks of diagnosis if no muscle is present in the specimen with high-grade disease, for any large >3 cm or multifocal lesion, or for T1 disease.5,6 The European Association of Urology (EAU) similarly recommends a second TURBT within 6 weeks, but only in the setting of incomplete resection, the absence of detrusor muscle in high-risk patients, and in T1 patients. 7 Proceeding with repeat TURBT is a strong recommendation with Grade B evidence for patients with incomplete initial resection or HG T1 disease in the American Urological Association (AUA)/Society of Urology Oncology (SUO) guidelines. Notably, it is a moderate recommendation with grade C evidence strength for patients with HR HG Ta disease. 5
Multiple studies have demonstrated the potential diagnostic, therapeutic, and prognostic benefits of repeat TURBT in HR NMIBC.8–13 However, the literature on repeat TURBT is limited by several key drawbacks, including small sample sizes, lack of standardized BCG therapy, and the absence of comprehensive clinical outcome assessments. Furthermore, selection biases in patient populations and study designs can skew results, making it difficult to draw definitive conclusions. The morbidity associated with repeat TURBT, such as the risk of bladder perforation and urinary tract infections, is also a significant concern and must be weighed against the potential benefits. Additionally, the financial burden and healthcare costs associated with repeat TURBT are an important consideration, particularly given the estimated cost of bladder cancer care in the United States exceeds 5 billion dollars annually. 14 Moreover, some studies suggest delays in the receipt of adjuvant therapy, i.e., for repeat resection, could adversely impact patient outcomes.15,16 Addressing these gaps and biases in future research is essential to provide more precise estimates of the effect of repeat TURBT on oncologic outcomes in patients with HG Ta NMIBC.
In this context, we aimed to evaluate the oncologic impact of repeat TURBT in patients with large volume (>5 cm), HG Ta NMIBC who completed induction BCG therapy. We selected patients with tumors larger than 5 cm, as this group is most likely to benefit from a repeat TURBT. By utilizing the Surveillance, Epidemiology, and End Results (SEER)-Medicare database, we address several limitations in the existing literature, including controlling for the receipt of adjuvant BCG and enhancing generalizability. We are also able to characterize the frequency and timing of high-risk NMIBC recurrence, providing robust, real-world estimates of the effect of repeat TURBT in this setting. We aim to not only offer insights into the clinical utility of repeat TURBT in the community setting but also to generate discussion around the potential value of a randomized study to support the guideline recommendations.
Methods
Study cohort and design
After obtaining Institutional Review Board at Mayo Clinic in Arizona (protocol #21-011911), a study using the Surveillance Epidemiology and End Results (SEER)–Medicare database from the year 2000 to 2015 was performed. Patients aged 66 years or older based on Medicare exclusion criteria with American Joint Committee on Cancer (AJCC) HG Ta pathology on initial diagnosis with at least three years of subsequent Medicare enrollment data were identified (Figure 1). Additionally, all patients were required to have the Current Procedural Terminology (CPT) code 52240 (cystourethroscopy and transurethral resection of large bladder tumor >5 cm) associated with their initial resection noting the presence of large volume HG Ta disease. This cohort was selected to reduce heterogeneity among HG Ta tumors, ensuring the inclusion of patients with high-risk disease for whom repeat TURBT is most likely to be beneficial. 17 Induction BCG, defined as at least five sequential BCG treatments, started within four months of initial resection or repeat TURBT was completed in all patients. 18 Concomitant carcinoma in situ (CIS) or T1 disease, variant histology, or evidence of metastatic disease were exclusions. Medicare Part A, Part B, and non-HMO enrollment at the time of cancer diagnosis, one year prior to diagnosis, and three years post diagnosis were required for inclusion. Patients with End-Stage Renal Disease at the time of diagnosis or source of diagnosis as an autopsy report or death certificate were excluded. Patients with a non-cancer related death within 3-year post diagnosis were excluded. Patients with a prior or subsequent malignancy diagnosis beyond bladder cancer were excluded.
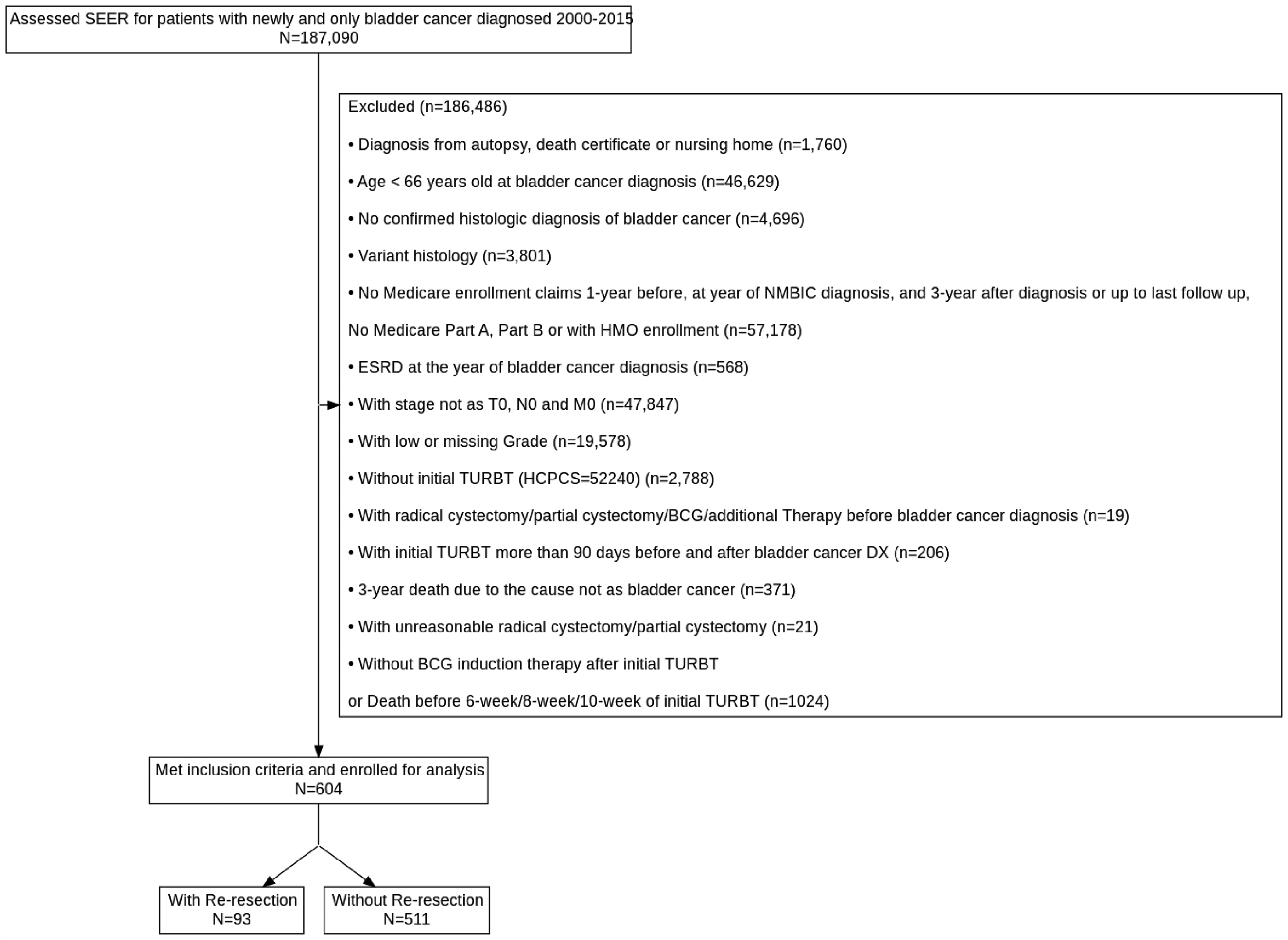
Flow chart for identifying high-grade, large volume Ta NMIBC with completion of induction BCG therapy.
The cohort of interest was divided into two groups: (1) the repeat TURBT group signified by completion of a re-resection (CPT 52204, 52214, 52224, 52234, 52235, 52240) within eight weeks of initial TURBT, and (2) the control group without a repeat TURBT. The primary outcome of interest was high-risk recurrence-free survival at 3 years. A high-risk recurrence was defined by a TURBT or cystoscopy biopsy and fulguration (CBF) followed by either additional induction therapy with BCG, mitomycin, or gemcitabine, or progression to partial or radical cystectomy. This definition of high-risk recurrence was utilized as no further pathologic data is provided by SEER-Medicare beyond initial diagnosis limiting the ability to apply conventional definitions.
In addition to the aforementioned cohort, a sensitivity analysis evaluating high-risk recurrence at two additional time points for re-resection (6 or 10 weeks) was conducted. A sensitivity analysis of a cohort with or without BCG induction therapy was also performed comparing high-risk recurrence by repeat TURBT status regardless of BCG.
Statistical analysis
Patient demographic and clinical characteristics were evaluated using Wilcoxon rank sum test for continuous variables and a Chi-squared test for categorical variables. Variables impacting the 3-year high-risk recurrence rate after initial diagnosis were assessed using univariate and multivariable analyses. Variables included for the multivariable analysis were age at diagnosis, sex, race, region of patient location, Charlson comorbidity index, repeat TURBT status, and number of BCG treatment on 3-year follow up. A Fine and Gray's model was utilized for the analyses. Cumulative Incidence Function (CIF) curves were plotted using the Gray test to compare the 3-year recurrence of the initial diagnosis. Statistical analysis was conducted with SAS version 9.4 (SAS Institute Inc). All tests were 2-sided, and P values <.05 were considered significant.
Results
A cohort of 604 patients with high-grade, large-volume (>5 cm) Ta bladder cancer who had completed induction BCG therapy was identified (Figure 1). Of these patients, 93 (15.4%) underwent repeat TURBT within eight weeks of initial TURBT, and 511 (84.6%) did not. Patient demographic and clinical characteristics are outlined in Table 1. Both groups were similar regarding their baseline characteristics, with no significant difference in repeat TURBT status with respect to age, sex, race, marital status, rural-urban localization, or comorbidity index. Overall, median age was 76.0 (IQR 71.0, 81.0), with most patients being white (92.9%), males (77.6%), and residing in metropolitan counties (85.1%). The median Charlson Comorbidity Index score for the cohort was 0.0 (IQR 0.0, 1.0).
Descriptive analysis of patient characteristics by repeat TURBT status.

Wilcoxon rank sum p-value; bChi-Square p-value.
* Specific numbers fewer than 11 cannot be reported according to requirements from Surveillance, Epidemiology, and End Results.
No significant difference in the 3-year high-risk recurrence rate between the two groups was observed (repeat TURBT: 20.4% vs. control group: 15.7%, p = 0.25) (Table 2). Of patients with recurrence, the time to recurrence after initial TURBT was longer in the repeat TURBT group (repeat TURBT 47.0 weeks (IQR: 32.0, 71.0) vs. control group 31.0 weeks (21.0, 53.0), p = 0.04). After controlling for patient demographics and clinical characteristics, multivariable Cox regression showed no significant association between repeat TURBT and 3-year high-risk recurrence (HR (95% CI): 1.27 (0.76, 2.11); p = 0.368) (Figure 2). Patients with higher BCG treatment numbers were found to have a higher risk of recurrence (HR (95% CI): 1.10 (1.07, 1.13); p < 0.001) representing repeat induction courses for recurrent high-risk disease. Regional differences in recurrence were identified, as patients from the Northeast were more likely to have recurrence than patients from the West (HR (95% CI): 2.57 (1.53, 4.31); p = 0.01). A Cumulative Incidence Function with Gray test showed no significant difference in 3-year recurrence-free survival between the repeat TURBT and control groups (p = 0.30) (Figure 3).
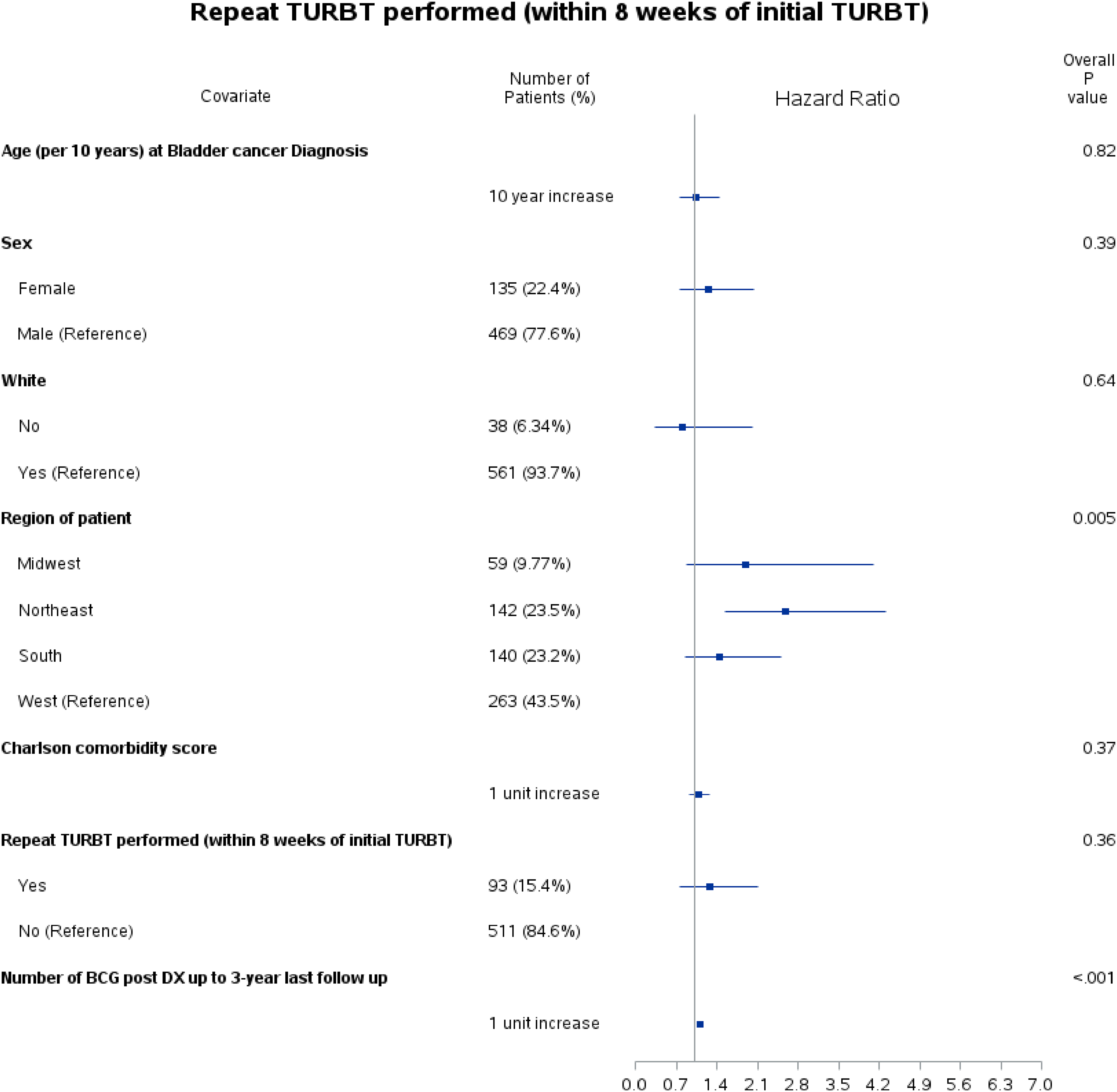
Multivariable analysis of 3-year recurrence.
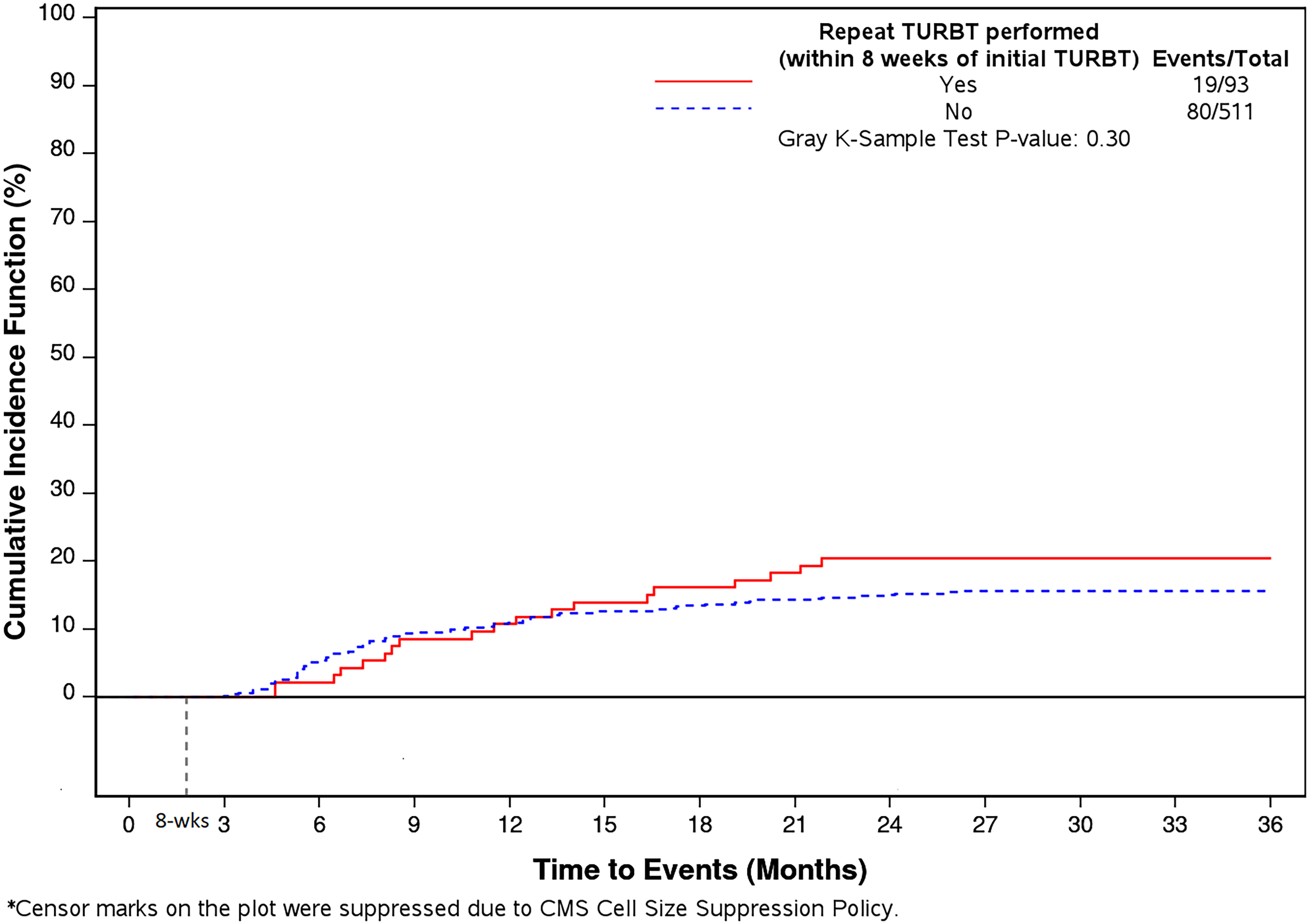
Cumulative incidence function curve of 3-year recurrence by repeat TURBT status.
Treatment and outcome profiles of patients by repeat TURBT status.
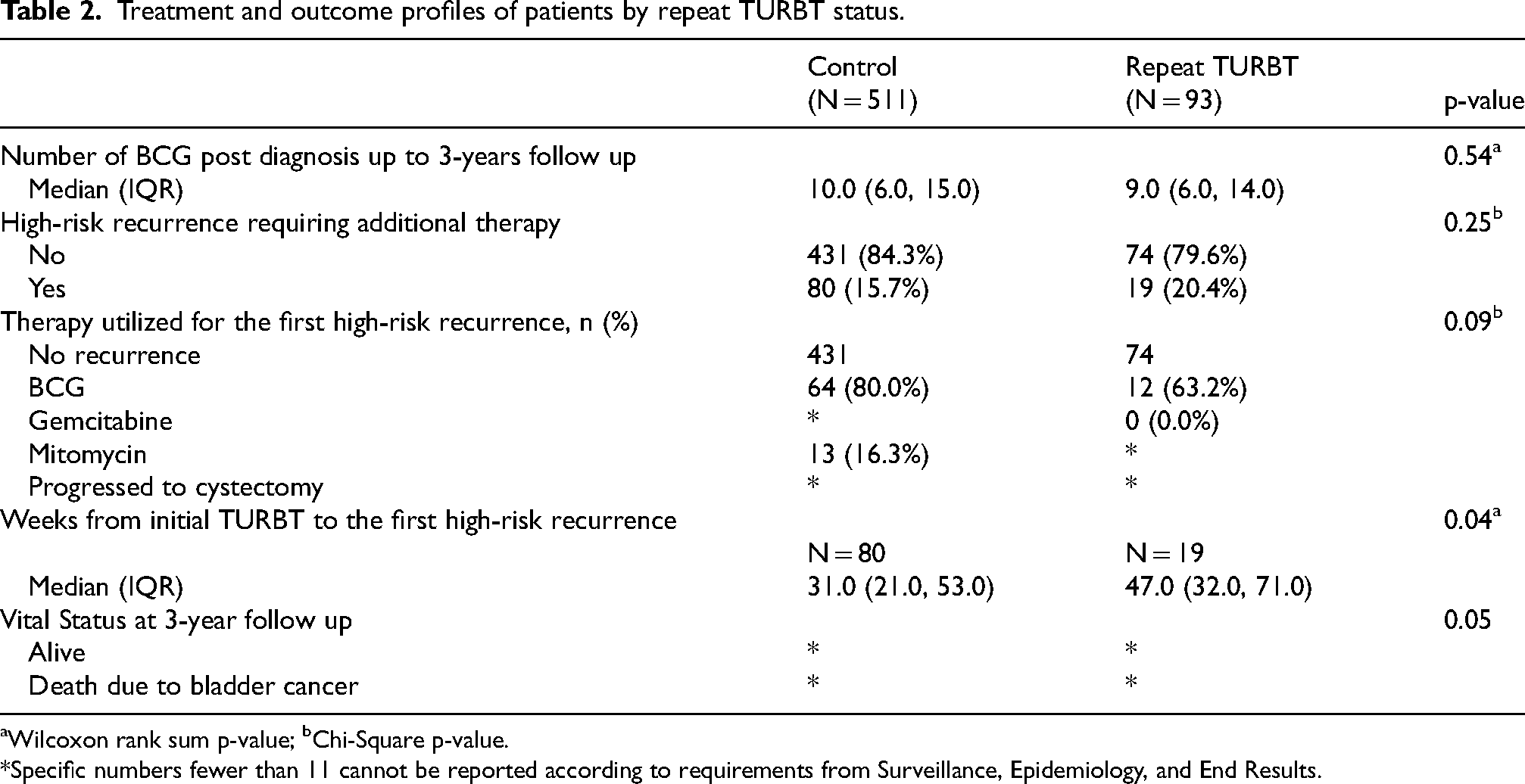
Wilcoxon rank sum p-value; bChi-Square p-value.
*Specific numbers fewer than 11 cannot be reported according to requirements from Surveillance, Epidemiology, and End Results.
The impact of re-resection at different time points on high-risk recurrence was additionally evaluated at 6 and 10 weeks to assess the potential impact of lead-time bias. Multivariable Cox regression showed no significant association between repeat TURBT and 3-year high-risk recurrence at 6 or 10 weeks (HR (95% CI): at 6 weeks 0.89 (0.46, 1.72); p = 0.72 and at 10 weeks 1.26 (0.75, 2.10) p = 0.38) (Supplementary Figure 1). The Cumulative Incidence Function curves by re-resection group using a landmark analysis revealed no significant difference in recurrence-free survival (Repeat TURBT within 6 weeks: p = 0.66 and 10 weeks: p = 0.33) (Supplementary Figure 2). Additionally, a sensitivity analysis of all patients, regardless of whether they had received BCG induction therapy, confirmed the null association between repeat TURBT and high-risk recurrence (repeat TURBT 22.7% vs. control group 22.7%, p = 0. 99; Supplementary Table 1).
Discussion
The primary finding of our study is that repeat TURBT does not significantly improve 3-year high-risk recurrence-free survival in patients who complete induction BCG therapy for high-volume (>5 cm) HG Ta NMIBC. This result challenges current clinical guidelines and practices that advocate for repeat TURBT in this patient population. However, we do not recommend the cessation of repeat TURBT based on these data alone. Instead, we suggest that there is sufficient uncertainty to justify experimental studies to investigate this question further.
Our findings are consistent with, but also distinct from, prior studies on the subject. Previous research has demonstrated the potential benefits of repeat TURBT for high-grade Ta disease.19–21 For example, Grimm et al. found improved 5-year recurrence-free survival in patients who underwent repeat TURBT, but this study included both Ta and T1 patients and did not isolate the effects on high-grade Ta disease or incorporate standardized BCG treatment protocols. 19 Similarly, Lee et al. showed a significant improvement in recurrence-free survival with repeat TURBT in a retrospective analysis, but their study included high-grade Ta tumors of all sizes and did not focus on high-risk tumors specifically. 20 A sub-analysis of patients who received BCG instillation also revealed improved recurrence-free survival rates (81.7% vs. 61.8%; p = 0.004). Another single-institution retrospective cohort of 209 high-grade Ta bladder cancer patients by Hensley et al. evaluated the role of repeat TURBT in the context of adequate BCG. 21 Of these patients, 104 (50%) underwent repeat TURBT. Analysis of AUA high-risk criteria patients within this population revealed that restaging TURBT decreased 5-year recurrence (67% vs. 15%, p = 0.002) and progression (25% vs. 6%, p = 0.109). However, progression-free survival outcomes did not vary based on the presence of residual disease on repeat TURBT (p = 0.574). It is unclear how generalizable these results are, given the tertiary-referral nature of the institution.
Unlike previous studies that often suffer from small sample sizes, lack of standardized treatment protocols, and inherent selection biases, our study utilizes the SEER-Medicare database to offer a more generalizable and robust analysis. The findings indicate that repeat TURBT does not significantly improve high-risk recurrence-free survival in this patient population, challenging current clinical guidelines and practices. This null association was also observed in a sensitivity analysis at different re-resection timepoints and in a sensitivity analysis including all patients regardless of whether they received induction BCG. While the time to recurrence after initial TURBT was longer in the repeat TURBT group (median difference of 16 weeks), it is unclear from the SEER-Medicare data to what degree the delayed recurrence is attributed to delays from timing of re-resection and recovery prior to proceeding with surveillance. This calls attention to the need for prospective data ideally from a randomized clinical trial setting. This study is important as it underscores the need for a critical reevaluation of the utility of repeat TURBT, considering the associated morbidity, financial costs, and potential delays in adjuvant therapy. Our results highlight the necessity for further research to optimize treatment protocols and improve patient outcomes in high-grade Ta NMIBC.
There are limitations to consider in this analysis. First, the completeness of the initial resection and presence of muscle in the specimen was unknown based on SEER-Medicare data, which could bias the results away from the null. However, since no association between repeat TURBT and recurrence-free survival was observed, the impact of this potential bias is thought to be low. Another major limitation to the study is the absence of subsequent pathologic data as SEER-Medicare only provides pathology from the initial diagnosis. This precludes evaluation of residual disease on repeat TURBT or subsequent recurrence or progression pathology. While no significant difference in recurrence-free survival was seen, we do acknowledge that repeat TURBT is performed to ensure no residual disease remains and accurate pathological staging is completed. Unfortunately, the present study was unable to assess this. Additionally, the database lacks comprehensive pathologic risk stratification data, including multifocality, prostatic urethral involvement, and lymphovascular invasion, which introduces potential confounders into our analysis. Of note, there is also no way to control for inherent surgeon selection bias when deciding on deferring versus proceeding with repeat TURBT. Furthermore, repeat TURBT was classified by a TURBT within 8 weeks of initial diagnosis and a sensitivity analysis was performed to demonstrate the null impact of timing on high-risk recurrence. However, it is possible that clinically patients underwent repeat TURBT beyond 6–10 weeks and were not identified correctly. Moreover, patients with high-risk recurrence could have declined further intravesical therapy or cystectomy and been mischaracterized due to the limitations of the definition of high-risk recurrence in this study. Finally, the repeat TURBT rate was low overall compared to some other reported studies and is potentially another source of selection bias. However, this could represent the actual adherence rate to this guideline, suggesting that most providers do not offer HR HG Ta patients a repeat TURBT. Despite these considerations, the present study provides evidence that the clinical impact of repeat TURBT in HR Ta patients may be limited.
In conclusion, in patients with large volume HG Ta NMIBC with completion of induction BCG therapy repeat TURBT was not associated with decreased high-risk recurrence-free survival. While repeat TURBT does likely identify residual disease in a subsection of this population, the implementation of a high-quality initial resection in conjunction with the treatment benefit of BCG therapy may nullify the therapeutic benefit of repeat TURBT. These data underscore the need for additional studies evaluating the utility of repeat TURBT in HR Ta NMIBC, ideally in a cooperative group setting.
Supplemental Material
sj-docx-1-blc-10.1177_23523735241303350 - Supplemental material for Repeat TURBT in large volume high-grade non-invasive bladder cancer
Supplemental material, sj-docx-1-blc-10.1177_23523735241303350 for Repeat TURBT in large volume high-grade non-invasive bladder cancer by Adri M. Durant, Mimi Nguyen, Mouneeb M. Choudry, Lanyu Mi, Jack R. Andrews and Mark D. Tyson in Bladder Cancer
Footnotes
Abbreviations
Acknowledgements
This study used the linked SEER-Medicare database. The interpretation and reporting of these data are the sole responsibility of the authors. The authors acknowledge the efforts of the National Cancer Institute; the Office of Research, Development and Information, CMS; Information Management Services (IMS), Inc.; and the Surveillance, Epidemiology, and End Results (SEER) Program tumor registries in the creation of the SEER-Medicare database.
The collection of cancer incidence data used in this study was supported by the California Department of Public Health pursuant to California Health and Safety Code Section 103885; Centers for Disease Control and Prevention's (CDC) National Program of Cancer Registries, under cooperative agreement 1NU58DP007156; the National Cancer Institute's Surveillance, Epidemiology and End Results Program under contract HHSN261201800032I awarded to the University of California, San Francisco, contract HHSN261201800015I awarded to the University of Southern California, and contract HHSN261201800009I awarded to the Public Health Institute. The ideas and opinions expressed herein are those of the author(s) and do not necessarily reflect the opinions of the State of California, Department of Public Health, the National Cancer Institute, and the Centers for Disease Control and Prevention or their Contractors and Subcontractors.
Author contributions
AD – Conception, data interpretation; drafting/revisions; MC - data interpret.; Revisions; LM – Stat analysis; JA - data interpret.; Revisions; MT – Conception; data interpret.; drafting/revisions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Grant Number P30 CA015083 from the National Cancer Institute (NCI) and from the Mayo Clinic Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery (M.D.T.), the Christian Haub Family Career Development Award for Cancer Research Honoring Dr Richard Emslander (M.D.T.), Eric and Gail Blodgett (M.D.T.) and the Endras Family (M.D.T.).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data available on request from the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
