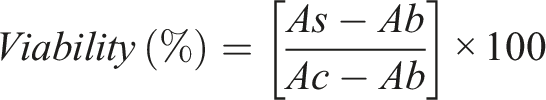Abstract
Propofol is well-known for its inhibitory effects in the central nervous system (CNS) as an intravenous anesthetic. Less is known about propofol’s impact on microglial activity. Studies have suggested a link to the endocannabinoid system (ECS). Propofol has been shown to indirectly modulate cannabinoid type 1 (CB1R) and type 2 (CB2R) receptors on microglia, with CB2R playing a key role in regulating anti-inflammatory immune responses. The objective of this study was to evaluate potential anti-inflammatory effects of propofol on microglia and to determine whether any observed effects are associated with the ECS. To investigate this, we treated LPS-stimulated SIM-A9 mouse-derived microglial cells with propofol, both in the presence and absence of antagonists for CB2R and CB1R, and assessed the cell viability and the production of the cytokines TNF and IL-6. The results demonstrated that cell viability was stable at propofol concentrations of 20, 40, and 80 µM. Production of TNF and IL-6 was reduced significantly upon propofol treatment. This effect of cytokine production did not change following administration of CB1R and/or CB2R antagonists. In conclusion, the results demonstrated that propofol exhibits anti-inflammatory effects, which do not appear to be mediated through ECS activation.
Introduction
Microglia originate from erythromyeloid progenitors in the yolk sac during early embryogenesis and migrate to the central nervous system (CNS) in fetal development. 1 Under homeostatic conditions, the microglial population is long-lived and maintained through slow, localized proliferation without external input. Following pathological insults, however, microglia undergo profound phenotypic and functional changes that critically influence disease outcomes.2–4 Traditionally, their activation was described as a binary polarization into proinflammatory “M1” or anti-inflammatory “M2” states. More recent transcriptomic and functional analyses, however, have shown that this classification oversimplifies microglial biology. 5 Instead, microglia display a broad spectrum of reactivity phenotypes that are dynamically shaped by the local microenvironment and disease context. This heterogeneity allows microglia to simultaneously express both pro- and anti-inflammatory features rather than fitting into rigid categories. Excessive proinflammatory activation, in particular, can drive an overly inflammatory milieu in affected brain regions, leading to neuronal injury and neurodegeneration.6–9 Thus, regulating microglial activation is critical for mitigating inflammation in CNS disorders.7,8
The endocannabinoid system (ECS) is integral to the development of the nervous system, ensuring proper neural formation and function. In the mature nervous system, the ECS regulates neuronal activity and network function. 10 The ECS comprises endogenous cannabinoids, cannabinoid receptors, and the enzymes responsible for the synthesis and degradation of endocannabinoids. Endogenous cannabinoids (eCBs) are signaling lipids that activate cannabinoid specific receptors. Among the agonists, 2-arachidonoyl glycerol (2-AG) and anandamide (AEA [N-arachidonoyl ethanolamine]) are the most well-characterized eCBs. Endocannabinoid signaling is often terminated by hydrolyzing the arachidonic group from either glycerol (2-AG) or ethanolamine (AEA). In the CNS, 2-AG is predominantly hydrolyzed by enzymes such as monoacylglycerol lipase (MAGL) or alpha/beta-hydrolase domain containing 6 (ABHD6). 11 Conversely, fatty acid amide hydrolase (FAAH) primarily degrades AEA, effectively terminating its activity. 12 In terms of receptors, cannabinoid type 1 receptor (CB1R) and cannabinoid type 2 receptor (CB2R) are the cannabinoid receptors. 10 Both receptors are G protein–coupled receptors (GPCRs) that primarily couple with inhibitory G proteins. The expression of CB1R on oligodendrocytes, oligodendrocyte precursors, and microglia is significantly lower, and their physiological roles are still being elucidated. CB2R are mainly found in cells of immune origin, including microglia, though they may also be present in neurons, especially under pathological conditions. Activation of the CB2R in microglia typically leads to anti-inflammatory effects. 13 It has been documented that CB2R activation in microglial cells results in the upregulation of M2 markers, effectively suppressing the expression of pro-inflammatory cytokines such as IL-1β, IL-6, and iNOS, while concurrently upregulating anti-inflammatory cytokines, such as IL-10. 13 Furthermore, inhibiting eCB-degrading enzymes can enhance eCB signaling by elevating the levels of endogenous ligands, further contributing to reducing inflammation. 13 This anti-inflammatory effect is achieved by downregulating pro-inflammatory cytokines, such as TNF, and upregulating anti-inflammatory cytokines, like IL-10. 13 Nonetheless, the impact of this inhibition on microglial polarization remains uncertain. 13
Propofol (2,6-diisopropylphenol) is an intravenous anesthetic widely used in clinical practice, known to have anti-inflammatory properties in CNS pathological conditions via modulating the activation of microglia.9,14 Propofol exerts protective and anti-inflammatory effects in CNS pathological conditions through a variety of mechanisms of action, such as maintaining mitochondrial function, reducing oxidative stress, decreasing excitotoxicity, preventing calcium overload, modulating programmed cell death, and promoting neurological recovery. 15 In a study intended to describe the activity of propofol, they determined that propofol acts as a competitive inhibitor of the anandamide-degrading enzyme, FAAH, which correlates with the observed increase in brain anandamide levels. 16 Additionally, they showed that propofol administration caused a variable increase in brain 2-AG content. 16 This is consistent with data indicating that 2-AG is a substrate for FAAH in vitro, although other brain processes, such as the enzyme MAGL, also inactivate 2-AG. 16 The unchanged metabolism of 2-AG in FAAH knockout mice suggests that propofol may inhibit MAGL or another metabolic pathway, leading to the observed increase in 2-AG levels. 16
While propofol has been well-documented for protective and anti-inflammatory effects in various CNS pathological conditions, there remains a significant gap in research regarding its impact on microglial activity through the ECS. Specifically, no studies have yet explored how propofol might modulate the endocannabinoid signaling pathways to exert its anti-inflammatory effects in microglial cells. Our hypothesis was that propofol primarily modulates microglia by activating the CB2R. To validate this hypothesis, we treated LPS-stimulated SIM-A9 (microglial) cells with antagonists for both CB2R (AM630) and CB1R (AM281) to observe the changes in microglial viability and production of the cytokines TNF and IL-6 when the cells were treated with and without propofol.
Materials and methods
Cell culture and maintenance
SIM-A9 female mouse microglia cells (ATCC, Cat# CRL-3265, Manassas, VA, USA) were removed from −80°C storage. A T25 tissue culture flask was prepared with 10 mL of pre-warmed complete growth medium (e.g., DMEM/F-12 supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin) and placed in a humidified incubator at 37°C with 5% CO2 to equilibrate. The frozen cell vial was rapidly thawed in a 37°C water bath, then transferred into the T25 flask containing pre-warmed medium. Cells were incubated for 24 h after which the medium was replaced with fresh complete medium. Once cells reached approximately 80–90% confluence, the supernatant was transferred to a 50 mL conical tube, and 0.25% trypsin-EDTA was added to the flask. The flask was returned to the 37°C, 5% CO2 incubator for 15 min to facilitate cell detachment. Any remaining attached cells were dislodged by gentle tapping and rinsing with fresh medium; the resulting cell suspension was combined with the previously collected supernatant and centrifuged at 1000 rpm, 4°C for 5 min. Cells were subsequently passaged in a 1:4 ratio until Passage 7 (P7).
Drug preparations
Propofol (Thermo Scientific, Cat# L06841.14, Waltham, MA, USA) was prepared at final concentrations of 0, 20, 40, 80, 160, and 320 µM in serum-free medium (SFM). Lipopolysaccharide (LPS; Sigma-Aldrich, Cat# L2630, St Louis, MO, USA) was used at 100 ng/mL, while the CB1R antagonist AM281 (Tocris Bioscience, Cat# 1115, Bristol, UK) and CB2R antagonist AM630 (Tocris Bioscience, Cat# 1120, Bristol, UK) were each used at 1 µM. The concentration of 1 µM for CB1R (AM281) and CB2R (AM630) antagonists was selected based on their reported binding affinities (Ki values) and receptor selectivity. This concentration allows effective inhibition of the target receptor while minimizing off-target interactions with the alternate cannabinoid receptor, thereby ensuring selective receptor blockade. All drug stocks were made in 100% DMSO; when diluted for final use, none of the treatment wells exceeded 0.5% DMSO. Control wells received only SFM, and vehicle-control wells received 0.5% DMSO (in SFM) without any active drug. Drug-treatment wells were prepared by mixing the appropriate volumes of each working solution in SFM just prior to application to cells, ensuring that the total DMSO concentration did not exceed 0.5%.
Plating, treatment, and sample collection
Cells at Passage 7 were harvested for plating into 96-well plates. The supernatant was transferred to a 50 mL tube and the cell layer was incubated with 0.25% trypsin-EDTA at 37°C, 5% CO2 for 15 min. After cell detachment, the suspension was again collected in the 50 mL tube and centrifuged at 1000 rpm, 4°C for 5 min. The pellet was resuspended in 1 mL of complete medium. A 1:50 dilution was prepared by adding 10 µL of the resuspended cells to 490 µL of complete medium; 10 µL of this diluted suspension was mixed with 10 µL of trypan blue or ink solution, and 10 µL of the mixture was transferred to a counting chamber. Cells were seeded in three 96-well plates (n = 3) at a density of ∼10,000 cells/well. Cells were then plated (150 µL/well) and incubated for 48 h at 37°C, 5% CO2 to allow attachment and recovery. After 48 h, media from each well was aspirated and replaced with 150 µL of the respective treatment solutions. LPS and propofol were added simultaneously. All treatments were performed in duplicate and the plates were incubated for another 24 h. The plates were then centrifuged at 1000 rpm, 4°C for 5 min, and 100 µL of supernatant from each well was collected and stored at −80°C for subsequent TNF and IL-6 ELISA assays.
Viability assay
Following supernatant collection, 10 µL of CCK-8 reagent (APExBIO, Cat# CK04, Boston, MA, USA) was added to each well and the plates incubated for 2 h at 37°C with 5% CO2. The absorbance of the formazan product in the centrifuged wells was measured at 450 nm using a microplate reader. Finally, the viability test results were calculated using the following formula: As: Absorbance of treated wells. Ab: Absorbance of blank wells. Ac: Absorbance of control wells (SFM).
TNF and IL-6 ELISAs
TNF and IL-6 levels were measured using commercial ELISA kits (Thermo Fisher Scientific, Cat# 88-7324 for TNF and Cat# 88-7064 for IL-6, Waltham, MA, USA) according to the manufacturer’s protocols. Each supernatant sample was thawed, added to the ELISA plate, and processed through the blocking, washing, and detection steps as specified. Absorbance readings of each well were used to determine cytokine concentrations by comparison to standard curves generated using known concentrations of TNF and IL-6.
Statistical methods
All statistical analyses were performed using GraphPad Prism (GraphPad Software, San Diego, CA, USA; version 10.4.2). Data were first assessed for distribution using tests for normality and lognormality, with the Shapiro-Wilk test applied for assessing normality. For comparisons between groups, an ordinary one-way analysis of variance (ANOVA) was conducted, followed by Dunnett’s multiple comparisons test. A p-value of less than 0.05 was considered statistically significant.
Results
Viability
A cell viability assay was first conducted on microglial cells exposed to increasing concentrations of propofol, revealing no significant effect on viability up to 40 μM, whereas a pronounced decline was observed at higher concentrations (80–320 μM); the control group was defined as the reference baseline (100%), with all treatment values normalized accordingly, and therefore error bars were not displayed for the control condition (Figure 1(a)). Microglial viability was also assessed following treatment with propofol (0–320 μM) along with LPS challenge. A substantial and significant decrease in cellular viability was observed at higher concentrations of 160 μM and 320 μM propofol combined with LPS (Figure 1(b)). When CB1R was blocked using AM281, the results demonstrated a slight, statistically insignificant increase in microglial viability at propofol concentrations of 20 μM and 40 μM (Figure 1(c)). However, a pronounced and significant decline in microglial viability was observed at higher concentrations of 160 μM and 320 μM (Figure 1(c)). Similar results were observed when CB2R was blocked with its antagonist AM630 (Figure 1(d)). No difference was observed when both receptors were blocked concurrently (Figure 1(e)). Effect of propofol on microglial cell viability under various experimental conditions. (a) Microglial cells treated with increasing concentrations of propofol (20–320 μM) in the absence of LPS exhibited a significant reduction in viability at 80 μM, 160 μM, and 320 μM. (b) Co-treatment with LPS (100 ng/mL) and propofol revealed a marked decline in cell viability at 160 μM and 320 μM. (c) In the presence of the CB1 receptor antagonist AM281, LPS (100 ng/mL), and propofol, a significant reduction in viability was observed at 160 μM and 320 μM. (d) In the presence of the CB2 receptor antagonist AM630, LPS (100 ng/mL), and propofol, the greatest reduction in viability occurred at 160 μM and 320 μM. (e) Combined treatment with LPS (100 ng/mL), AM281, AM630, and propofol also resulted in significantly decreased viability at 160 μM and 320 μM. Data are represented as mean ± SD (n = 3). *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001, ****p ≤ 0.0001; Statistical analysis was performed using one-way ANOVA followed by Dunnett’s multiple comparisons test.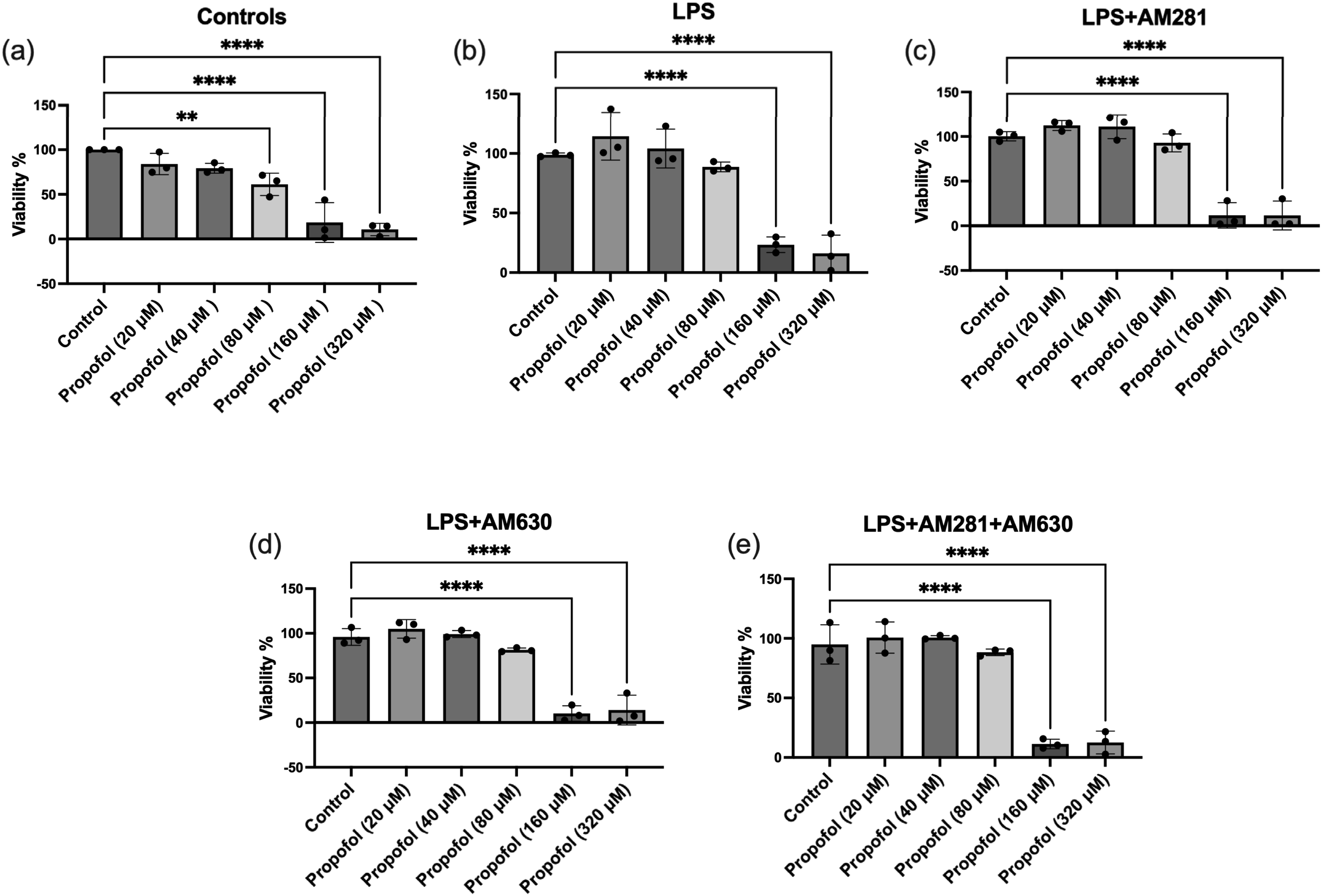
TNF release
ELISA assays were conducted to evaluate the effect of propofol on TNF release in SIM-A9 microglial cells under various treatment conditions. Initially, cells were treated with four graded concentrations of propofol to assess its isolated impact on TNF production. The results demonstrated that propofol alone did not induce TNF release at any tested concentration, confirming that it does not intrinsically trigger pro-inflammatory cytokine production (data not shown). To determine whether propofol modulates TNF production under inflammatory conditions, microglia were co-treated with LPS and increasing concentrations of propofol. LPS alone induced robust TNF release, whereas the addition of propofol significantly reduced TNF levels across all concentrations (Figure 2(a)). Next, the role of cannabinoid receptors in this anti-inflammatory effect was investigated. When CB1R was blocked using AM281, the suppressive effect of propofol on LPS-induced TNF production persisted, with significantly reduced TNF levels observed at all propofol concentrations (Figure 2(b)). Similarly, blocking CB2R with AM630 did not abolish propofol’s effect; TNF production remained markedly lower in propofol-treated cells compared to those treated with LPS alone (Figure 2(c)). To examine whether simultaneous blockade of both CB1R and CB2R influences propofol’s efficacy, microglia were treated with LPS, AM281, AM630, and propofol. Even under these conditions, propofol led to a significant reduction in TNF levels across all tested concentrations (Figure 2(d)). Effect of propofol on TNF production in microglial cells under various treatment conditions measured by ELISA. (a) Co-treatment with LPS (100 ng/mL) and various concentrations of propofol resulted in a significant reduction in TNF release at all tested doses compared to LPS alone. (b) In the presence of the CB1 receptor antagonist AM281, LPS (100 ng/mL), and propofol, TNF levels were significantly decreased at all propofol concentrations. (c) Treatment with LPS (100 ng/mL), the CB2 receptor antagonist AM630, and propofol also resulted in a marked reduction in TNF production across all concentrations. (d) Combined exposure to LPS (100 ng/mL), AM281, AM630, and propofol led to a significant reduction in TNF levels at all propofol concentrations. Data are represented as mean ± SD (n = 3). *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001, ****p ≤ 0.0001; statistical analysis was performed using one-way ANOVA followed by Dunnett’s multiple comparisons test.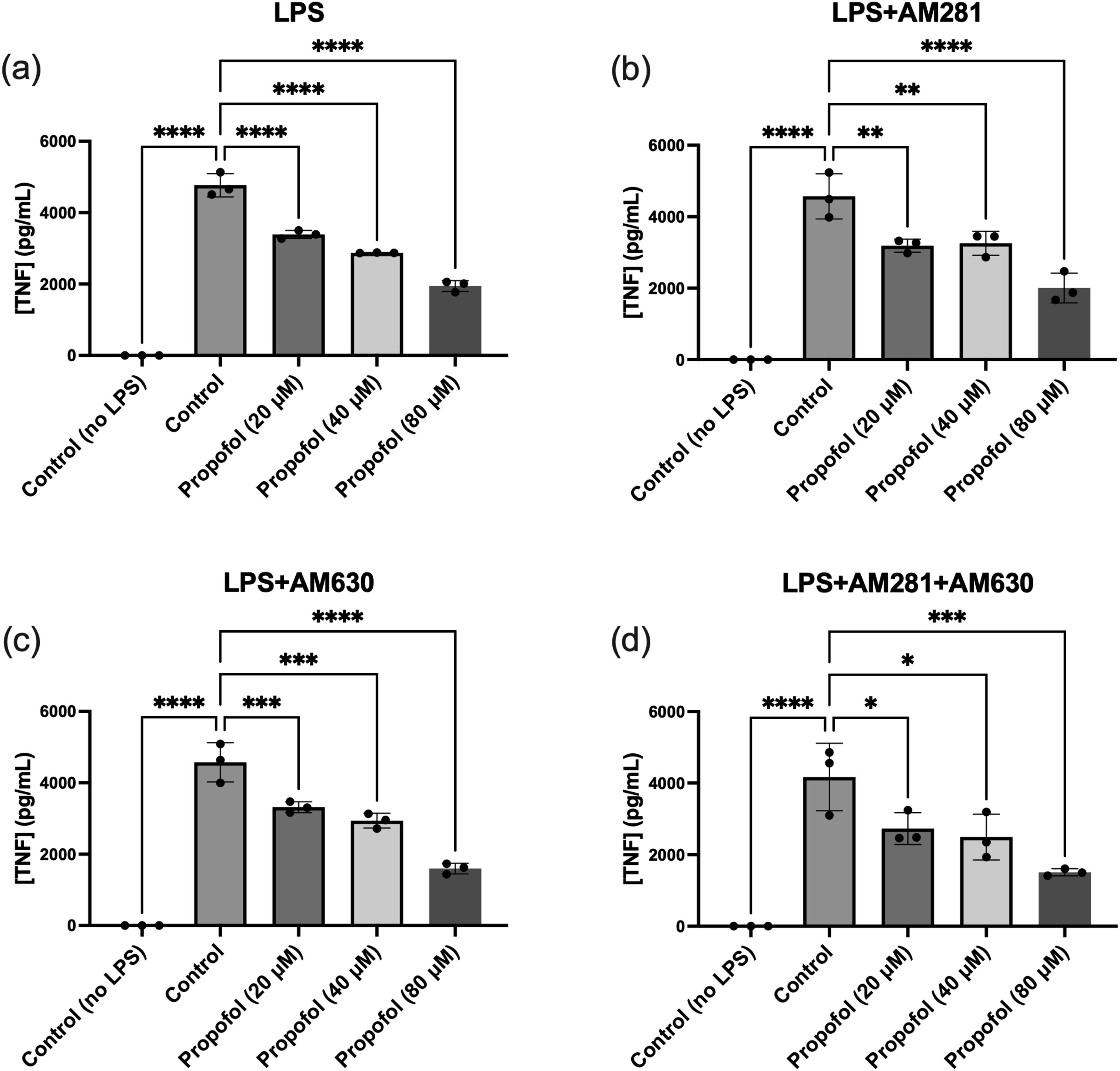
IL-6 release
ELISA assays were conducted to evaluate the effect of propofol on IL-6 production in SIM-A9 microglial cells under various treatment conditions. To assess the independent impact of propofol, microglia were treated with four increasing concentrations of propofol without any additional stimuli. The results demonstrated that propofol alone did not induce IL-6 release at any tested concentration, confirming that it does not intrinsically trigger pro-inflammatory cytokine production (data not shown). To examine the anti-inflammatory potential of propofol under inflammatory conditions, microglia were co-treated with LPS and varying concentrations of propofol. LPS stimulation significantly increased IL-6 release, whereas co-treatment with propofol led to a significant reduction in IL-6 levels across all concentrations tested (Figure 3(a)). The role of cannabinoid receptors in this response was then investigated. In the presence of the CB1R antagonist AM281, propofol continued to significantly reduce IL-6 production, indicating that CB1R does not mediate this effect (Figure 3(b)). When CB2R was inhibited using AM630, a reduction in IL-6 release was still observed with propofol treatment; however, this decrease did not reach statistical significance (Figure 3(c)). Furthermore, when both CB1R and CB2R were concurrently blocked using AM281 and AM630, propofol still elicited a significant decrease in IL-6 levels across all tested concentrations (Figure 3(d)). Effect of propofol on IL-6 production in microglial cells under various experimental conditions. (a) Co-treatment with LPS (100 ng/mL) and increasing concentrations of propofol resulted in a dose-dependent reduction in IL-6 release. (b) In the presence of the CB1 receptor antagonist AM281 and LPS (100 ng/mL), propofol significantly reduced IL-6 levels at all tested concentrations compared to treatment without propofol. (c) Treatment with LPS (100 ng/mL) and the CB2 receptor antagonist AM630 resulted in only a modest, statistically non-significant reduction in IL-6 release with increasing concentrations of propofol. (d) Combined exposure to LPS (100 ng/mL), AM281, AM630, and propofol significantly reduced IL-6 levels across all concentrations compared to the non-propofol-treated group. Data are represented as mean ± SD (n = 3). *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001, ****p ≤ 0.0001; statistical analysis was performed using one-way ANOVA followed by Dunnett’s multiple comparisons test.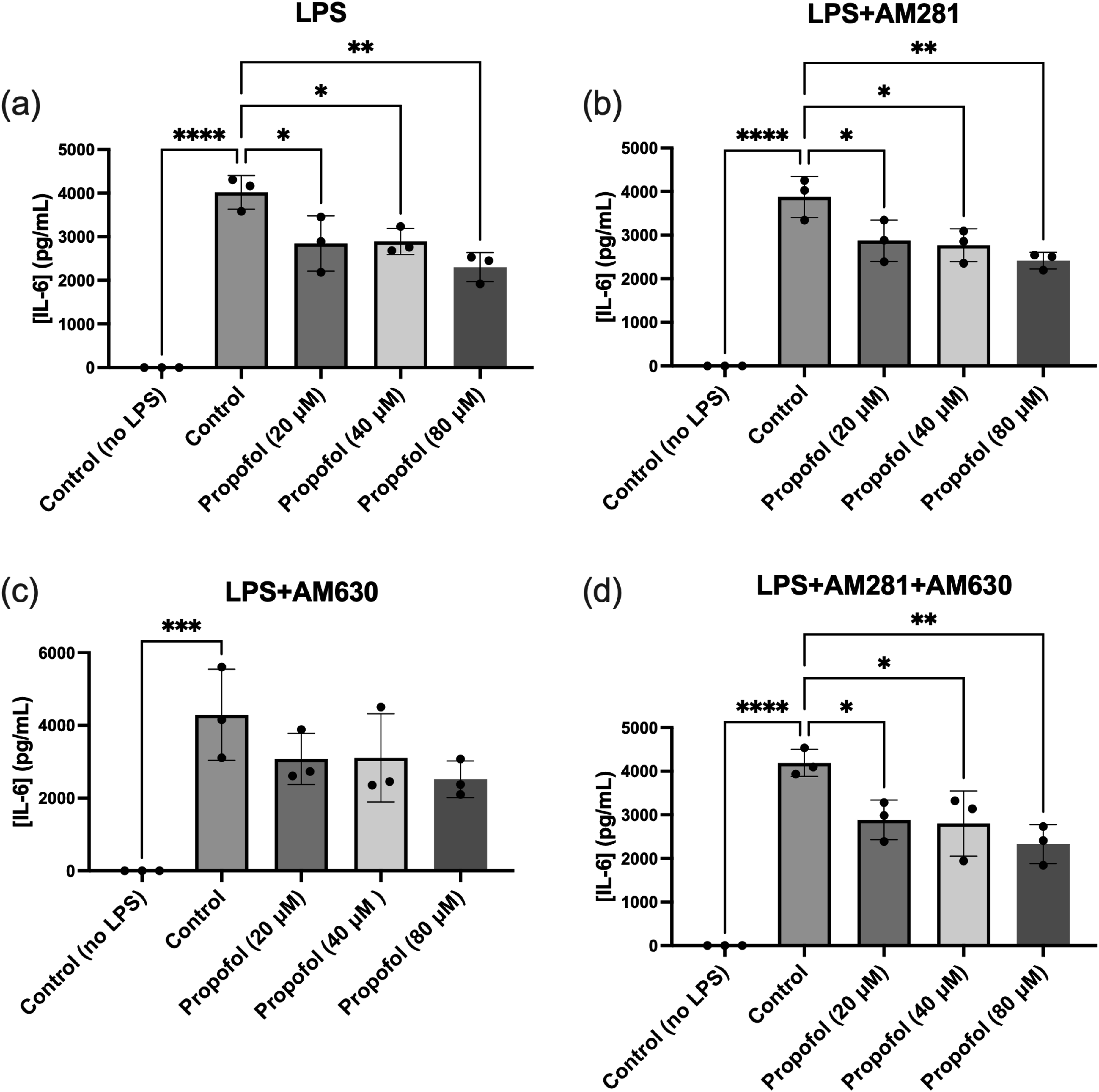
Discussion
The cell viability results demonstrate that propofol is safe at concentrations up to 80 μM, which is consistent with previous studies in BV-2 microglia where no significant reduction in viability was observed between 1 and 100 μM, although cytotoxicity was evident at 300 μM with an ∼81% decrease in viability. 17 In our study, while 80 μM propofol alone reduced cell viability (Figure 1(a)), this effect was not observed when cells were co-treated with LPS, likely due to the protective signaling effects induced by LPS. Indeed, several studies have reported that LPS stimulation promotes cellular tolerance and survival by redirecting TLR4 signaling toward the TRIF–IRF3 axis and suppressing NF-κB activity, 18 inhibiting NF-κB/caspase-3–mediated apoptosis, 19 and enhancing PI3K/Akt-driven cytoprotection. 20 Collectively, these protective mechanisms counteract propofol-induced cytotoxicity and help preserve microglial viability under co-treatment conditions.
Notably, although clinically measured plasma concentrations of propofol are typically lower, its pharmacokinetic properties support the use of higher concentrations in in vitro systems. Propofol is highly lipophilic, extensively protein-bound in plasma, and undergoes extensive metabolic clearance, with only a small fraction present in the unbound form. 21 In human studies, cerebrospinal fluid (CSF) concentrations of propofol are substantially lower than plasma levels, representing only a small percentage of circulating concentrations. 22 Furthermore, propofol exists in both free and bound forms within the CSF, indicating that binding dynamics contribute to its distribution within the central nervous system. 23 In addition, experimental in vitro models have demonstrated that propofol exposure can alter blood–brain barrier integrity and increase permeability. 24 Therefore, higher concentrations may be required in in vitro systems to compensate for the absence of protein binding, metabolic clearance, and physiological barriers present in vivo.
The TNF ELISA results revealed that propofol significantly reduces TNF production in SIM-A9 microglial cells under inflammatory conditions induced by LPS. This anti-inflammatory effect persisted even when CB1R or CB2R were blocked individually or together, indicating that propofol’s suppression of TNF is not mediated by ECS activation. This suggests that propofol may exert its modulatory effects through alternative signaling pathways independent of cannabinoid receptor activation. Furthermore, the IL-6 ELISA results demonstrated that propofol significantly attenuates IL-6 release. This anti-inflammatory effect remained evident even with CB1R blockade. A decrease in IL-6 release was also observed with CB2R blockade, although it was not statistically significant, suggesting that propofol’s ability to reduce IL-6 release may be partially mediated through CB2R. However, when both CB1R and CB2R were blocked simultaneously, propofol still significantly reduced IL-6 production, reinforcing that its anti-inflammatory action may involve alternative signaling pathways. These findings support the view that propofol modulates inflammatory cytokine release through complex, possibly multifaceted regulatory systems beyond cannabinoid receptor activation. The anti-inflammatory effects of propofol through modulation of other cytokine pathways have been confirmed by multiple studies. In a study, propofol demonstrated significant anti-inflammatory effects in both in vitro and in vivo models of ischemia/reperfusion (I/R) injury. 25 Specifically, propofol treatment led to a marked reduction in the production of the pro-inflammatory cytokine IL-1β and a simultaneous increase in the anti-inflammatory cytokine IL-10. These changes were observed in hypoxia/reoxygenation-injured microglial cells and were associated with decreased microglial activation. The findings suggest that propofol modulates the inflammatory response by downregulating IL-1β and upregulating IL-10, thereby contributing to its neuroprotective effects. In another study, propofol was shown to reduce oxidative stress in CoCl2-induced hypoxic BV2 microglial cells by significantly decreasing reactive oxygen species (ROS) production. 26 CoCl2 exposure led to elevated ROS levels and reduced antioxidant activity, including diminished superoxide dismutase (SOD) activity and total antioxidant capacity (T-AOC). Propofol treatment reversed these effects by restoring SOD and T-AOC levels, thereby inhibiting ROS overproduction and subsequent inflammatory responses. This antioxidative action was associated with suppression of nuclear factor kappa B (NF-κB) activation and prevention of Hypoxia inducible factor-1α (HIF-1α) stabilization, ultimately reducing pro-inflammatory signaling. Additionally, propofol improved mitochondrial membrane potential, further supporting its protective role against oxidative damage in microglial cells. Furthermore, in one study, propofol significantly reduced nitric oxide (NO) production in LPS-activated BV2 microglial cells, highlighting its anti-inflammatory potential. 27 The study demonstrated that propofol significantly reduces nitric oxide (NO) production in LPS-activated BV2 microglial cells. LPS stimulation alone caused a 5-fold increase in NO levels compared to the control. However, treatment with propofol at concentrations ranging from 12.5 to 100 μM inhibited this response in a dose-dependent manner. At the highest concentration (100 μM), propofol reduced NO production by approximately 46% during LPS challenge. These effects were consistent at both the transcriptional (iNOS mRNA) and translational levels.
As the anti-inflammatory effects of propofol through modulation of the ECS had not yet been investigated, this study is the first to examine whether propofol’s anti-inflammatory effects may be mediated via the ECS. The ECS is essential for managing microglial activity during neuroinflammation. 28 Under inflammatory conditions, microglia generally adopt a proinflammatory M1 state, releasing cytokines such as IL-1β, TNFα, and IL-6. Activating the ECS, especially via CB2R, mitigates this effect by encouraging a transition to the anti-inflammatory M2 state. This transition results in higher levels of anti-inflammatory markers like IL-10 and Arg-1. However, the findings of this study indicate that the anti-inflammatory effects of propofol, particularly its ability to suppress TNF and IL-6 production in microglial cells, are not solely dependent on CB1R or CB2R activation. While CB2R may play a partial role in mediating IL-6 reduction, the continued effectiveness of propofol in reducing both cytokines even when both cannabinoid receptors are blocked strongly suggests the involvement of alternative receptor-independent pathways in its anti-inflammatory mechanism. In line with this interpretation, previous studies have similarly reported that cytokine suppression can occur independently of classical CB1R/CB2R signaling, although more recent findings highlight a partial regulatory role for CB2R in IL-6 modulation. Previous studies have shown that neither the CB1-selective antagonist SR141716A nor the CB2-selective antagonist SR144528 was able to reverse the inhibition of cytokine mRNA expression by levonantradol in microglial cells, indicating that cannabinoid-mediated cytokine suppression can occur independently of classical CB1R/CB2R blockade. 29 In contrast, more recent findings demonstrated that activation of CB2R with selective agonists such as JT11 significantly reduces LPS-induced IL-6 release in human PBMCs, and this effect is partially reversed by CB2R antagonism, confirming a regulatory role of CB2R in IL-6 production. 30 Consistent with these observations, our data showed that AM630 (a CB2R antagonist) partially inhibited the reduction of IL-6, suggesting that CB2R signaling may contribute to IL-6 regulation in our model. However, as propofol’s overall immunomodulatory effects persisted despite cannabinoid receptor blockade, these findings indicate that its anti-inflammatory properties are not primarily mediated through ECS activation, although CB2R signaling may partially influence IL-6 modulation under specific conditions.
This study has a few limitations that should be considered. First, the microglial cells used were obtained from female mice. Since research increasingly shows that immune responses, particularly within the central nervous system, can differ based on sex, it is possible that microglia from male mice might display varying activation patterns, cytokine release, or sensitivity to propofol and cannabinoid receptor antagonists. Future research should investigate whether these sex-related differences affect propofol’s anti-inflammatory effects via the ECS. Second, only a single concentration (1 μM) of the cannabinoid receptor antagonists AM281 and AM630 was tested in this study. While this dosage was informed by previous studies, relying on one concentration restricts the ability to evaluate dose-dependent effects or explore whether higher or lower doses might produce different results; however, 1 µM was sufficient to achieve receptor saturation based on reported Ki values while minimizing potential cross-activity between AM281 and AM630, and the rationale behind this choice is multifactorial.31,32 Future experiments using a broader range of antagonist concentrations could better elucidate receptor involvement and potential competitive binding. Finally, this study was conducted exclusively in vitro and has not yet been translated into human models. Although in vitro systems are valuable for exploring cellular mechanisms, they do not fully replicate the complex interactions of a living organism. Therefore, caution must be taken when generalizing these results to human physiology. Future studies involving human-derived microglia or clinical trials will be necessary to validate the relevance of these findings in the context of human neuroinflammation and anesthetic care. These limitations highlight important areas for future investigation that could improve the applicability and robustness of the findings.
Conclusions
In conclusion, this study demonstrates propofol’s notable anti-inflammatory effects on microglial cells, as evidenced by its ability to reduce TNF and IL-6 production. The observation that these effects occur independently of CB1R and CB2R activation suggests the involvement of alternative, non-cannabinoid mechanisms. Given its widespread clinical use as an intravenous anesthetic with a well-characterized safety and pharmacokinetic profile, propofol may represent a feasible therapeutic option in acute stroke care. Beyond its role in providing sedation during procedures such as mechanical thrombectomy, its anti-inflammatory and neuroprotective properties may offer additional benefit by attenuating early neuroinflammatory responses. This dual functionality is particularly relevant in the hyperacute phase of stroke, where rapid modulation of inflammation may influence secondary brain injury. Furthermore, in scenarios where revascularization procedures have already been performed, or where such interventions are not applicable, early administration of low or sub-anesthetic doses of propofol may contribute to regulating microglial activation, reducing oxidative stress, and limiting apoptotic signaling in the peri-infarct region. However, these potential applications remain speculative and require validation in in vivo models and clinical studies to determine their therapeutic relevance and optimal use. While these findings contribute to our understanding of propofol’s broader immunomodulatory properties, further research is required to identify the specific pathways involved and to evaluate its therapeutic potential in clinical conditions such as stroke. Additionally, factors such as sex differences, a range of antagonist concentrations, and the use of human models should be considered in future studies to enhance the validity and applicability of the results.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Data Availability Statement
Data are available on request.