Abstract
Objective. The objective of this public health surveillance project was to estimate the proportion of persons under the age of 18 years with confirmed SARS-CoV-2 infection who developed chronic symptoms and to further characterize the symptoms and associated factors. Methods. Parents provided information during a telephone interview or through a web-based questionnaire. Results. Of 212 infants, children, and adolescents, 11 (5.2%) experienced a wide range of mild to severe chronic symptoms following SARS-CoV-2 infection, with 90% indicating inability to continue normal activities. Factors associated with chronic symptoms were pre-existing chronic medical conditions overall, certain categories of symptoms (eg, neurologic/psychiatric symptoms), and specific symptoms experienced with the acute SARS-CoV-2 infection (eg, fatigue, vomiting). Conclusion. Future research regarding chronic symptomatology among persons with confirmed SARS-CoV-2 infection would benefit from the use of standardized definitions of the timing and duration of chronic symptoms and from the enrollment of comparison groups.
Introduction
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) was first identified in late 2019 and has affected millions of persons worldwide. 1 Subsequently, the phenomenon of long-term clinical sequelae (“long COVID”) of SARS-CoV-2 infection was reported. 2 However, the definition of long COVID is not universally agreed upon, and the complete spectrum of clinical manifestations, duration, and risk factors for long COVID have not been completely characterized.
The Vermont Department of Health (VDH) developed a public health surveillance project in collaboration with the University of Vermont to assess chronic symptoms among patients of all ages with confirmed SARS-CoV-2 infections. We estimated the proportion of persons with chronic symptomatology and characterized the symptoms experienced, the severity and duration of such symptoms, and factors associated with the development of these symptoms. This paper addresses these objectives for infants, children, and adolescents under 18 years of age.
Methods
All SARS-CoV-2 diagnostic test results in Vermont are reported to the VDH. The source population for this public health surveillance project was persons who had a positive SARS-CoV-2 result by polymerase chain reaction (PCR) assay between March 28 and July 24, 2021. These persons were eligible for inclusion in the study population if they met the following criteria: resident of Vermont at the time of specimen collection, reached for an initial interview by the VDH contact tracing team shortly after (generally within 24-48 hours of) the positive PCR assay result, without a baseline neurological disability as ascertained during the initial contact tracing team interview, were still alive, and were not inmates in a correctional facility. Those who met inclusion criteria were stratified by age at the time of diagnosis of SARS-CoV-2 (positive SARS-CoV-2 PCR assay) (<18, 18-49, and 50 years or more) and by calendar week (17 weeks) of the enrollment period. A maximum of 300 persons per week were included in the population eligible for interview—using random selection (for those weeks with more than 100 persons in each age group) or else incorporating up to 100 persons per age group and per week if less than 100 persons/age group for a given week. For those under the age of 18 years, a parent or legal guardian provided responses to questions.
Persons in the population eligible for interview were contacted either by telephone for an interview or, if an email address was available, by email to complete a web-based questionnaire. The same questions were included in the telephone interview and the web-based questionnaire. The questionnaire was not validated or pilot tested. Interviews were conducted by research staff in the Office of Clinical Trials Research at the Larner College of Medicine (LCOM)/University of Vermont Medical Center according to all relevant guidelines and regulations. Interviews/questionnaires were scheduled to occur at up to 4 time points (3, 6, 9, and 12 months) after the positive SARS-CoV-2 PCR assay. Three-month interviews were scheduled to occur 13 weeks after the date of the positive PCR assay, with windows for the interviews (−2 weeks/+6 weeks). Subsequent interviews were scheduled to occur 26, 39, and 52 weeks (6, 9, and 12 months) after the date of the positive PCR assay, with windows for the interviews (−2 weeks/+2 weeks). For interview contacts attempted, no more than 3 attempts were made over a 3-week period. At each attempted telephone contact, if direct contact was not successful, a voice mail message was left indicating the person who called, and that the person was calling from LCOM on behalf of the VDH and providing a telephone number and email address where the participant/parent of the participant could reach project staff. If no response was received from the parent/legal guardian after the third attempted contact, then no further contact attempts were made for that time point. For individuals who provided an email address to the VDH at the time of their initial contact tracing interview, the parent/legal guardian was sent 3 email messages over a 3-week period. If no response, no further email messages were sent for that time point. The University of Vermont Medical Center’s translation services were utilized for persons whose primary language was not English. Study data were collected and managed using REDCap electronic data capture tools hosted at LCOM.3,4
A participant would be categorized as “Off Study” for any of the following reasons: (1) the team was not able to contact the participant (eg, invalid contact information); (2) the team was not able to reach the participant after three attempts within the allowable timeframes at 13 weeks (−2/ + 6 weeks) and at 26, 39, and 52 weeks (±2 weeks) after the date of the positive PCR assay; (3) the participant declined to participate; (4) the participant completed a survey but then declined to continue in future surveys; or (5) the participant had 2 time points where they reported no symptoms (or, if symptomatic at the 3-month time point, but asymptomatic at either 6 months or 9 months).
In both the telephone interview and the web-based questionnaire, participants answered questions regarding medical history (health status before their SARS-CoV-2 infection and any chronic medical conditions), symptoms experienced with their acute SARS-CoV-2 infection, and subsequent symptoms. For the purposes of this analysis, participants who reported at any time point (3, 6, 9, or 12 months after the positive SARS-CoV-2 PCR assay) that they were doing worse than before the date of the positive assay were categorized as having chronic symptomatology after acute SARS-CoV-2 infection.
Statistical Analysis
Descriptive statistics were computed using medians, ranges, and interquartile ranges for continuous variables and percentages for categorical variables. Patients with and without chronic syptomatology were compared using Wilcoxon rank sum tests for continuous variables and Fisher’s exact tests for categorical variables. Analyses were conducted using SAS 9.4 (Cary, NC: SAS Institute Inc.).
Ethical Approval and Informed Consent
The study protocol was reviewed by offices of both the Vermont Agency of Human Services and the University of Vermont Institutional Review Boards (IRBs). Both deemed the study did not meet the federal definition of research; therefore, neither IRB approval nor written informed consent from participants (or, for minors, written informed consent from parent(s)) was required.
Results
Derivation of the Study Population
Of 6169 persons with positive SARS-CoV-2 PCR assay results during the study period, 2602 persons did not meet inclusion criteria: 761 were out-of-state residents, 1777 were not reached for an initial interview by the contact tracing team, 209 had a baseline neurological disability, 65 were deceased, and 53 were inmates at a correctional facility. Of the remaining 3567 persons, 2636 were eligible for interview (following stratification by age and week, and selection as described above). Of these 2636 persons, 895 (34%) were less than 18 years of age at the time of the positive SARS-CoV-2 PCR assay.
Size and Characteristics of the Study Population
Of the 895 persons under the age of 18 years, 212 (24%) agreed to participate in telephone interview(s) (n = 114) or complete web-based questionnaire(s) (n = 98) at 3 and/or 6 months (Figure 1). Ninety-nine participated at 3 months only, 52 at 6 months only, and 61 at both time points. There were no differences between those who did (212) and those who did not (683) participate in terms of age, sex, and primary language (English vs other) (Table 1).

Derivation of the study population.
Characteristics of the Pediatric Participants and Non-participants (N = 895).
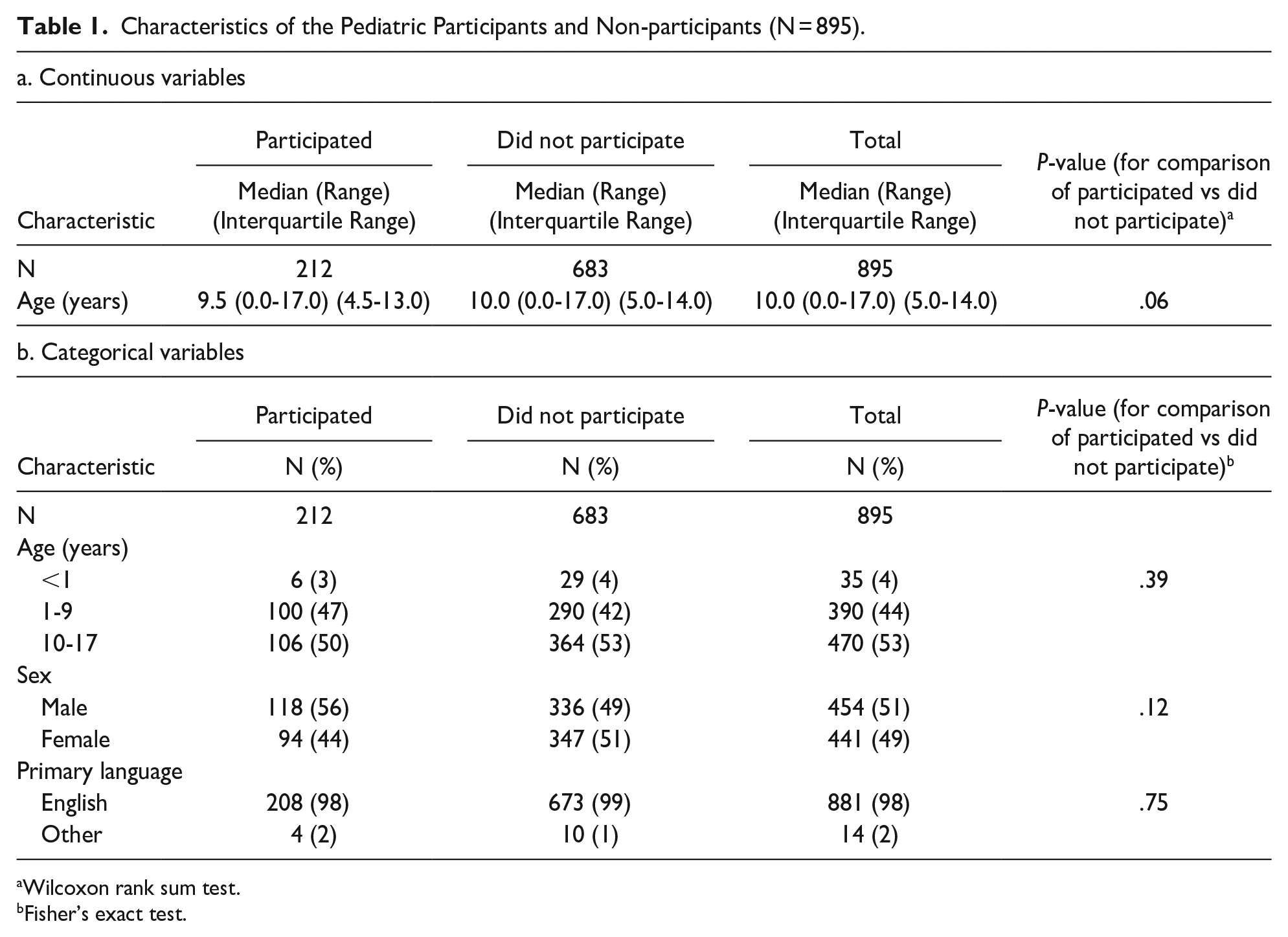
Wilcoxon rank sum test.
Fisher’s exact test.
Characteristics of the 212 are shown in Table 2, overall and according to whether categorized as having or not having chronic symptoms after the acute SARS-CoV-2 infection. Only 3% of the children in the study population were infants, with the rest almost evenly divided between 1 and 9 and 10 to 17 years old. Almost all parents had English as their primary language, with the remainder speaking Nepali, Romanian, and Vietnamese. All children were described by their parent(s) as being healthy or very healthy before their SARS-CoV-2 infection. Seventeen percent of the children had 1 or more chronic medical conditions. Most (64%) were symptomatic with their SARS-CoV-2 infection but, of these, only 34% had a moderate or severe illness. No children were hospitalized. Three percent reported missing school or child-care after their SARS-CoV-2 infectious period ended.
Characteristics of the Study Population (N = 212).

Wilcoxon rank sum test.
Fisher’s exact test.
Of the 212 who participated in the 3- and/or 6-month interviews/questionnaires, 33 were asymptomatic at both time points, 33 did not participate in the 3-month interview and were asymptomatic at the 6-month time point, 24 were symptomatic at 3 months but asymptomatic at 6 months, and 10 withdrew from the study following the 6-month time point (Figure 1). Therefore, 112 were remaining and were eligible to participate in subsequent interviews/questionnaires. Of these 112, 10 were invited to interview or complete the questionnaire at 9 months but declined, and 85 did not respond to outreach for the 9-month interview/questionnaire. The remaining 17 persons completed the 9-month interview/questionnaire. Of the 17, 15 were asymptomatic at the 9-month time point and 2 did not respond to outreach for the 12-month interview.
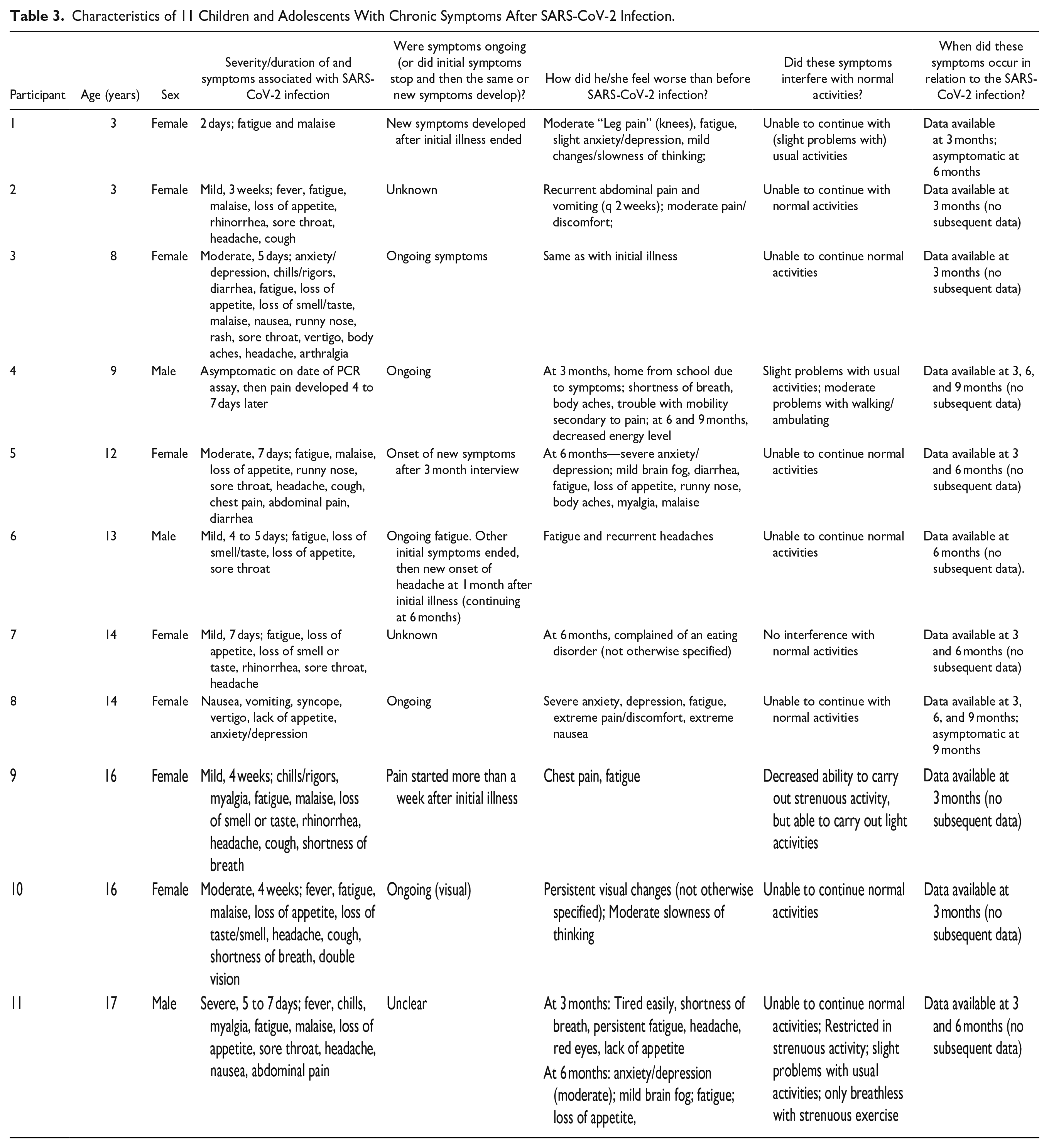
Overall, 11 (5%) were categorized as having chronic symptoms following the acute SARS-CoV-2 infection. Characteristics of these 11 children are shown in Table 3. The age range was 3 to 17 years, with most (n = 7, 64%) aged 12 years or more. Most (n = 8, 73%) were female. Only 1 child was asymptomatic with the SARS-CoV-2 infection, and the remaining 10 children’s symptoms ranged from mild to severe. Of 8 children with responses, 5 reported that symptoms of their SARS-CoV-2 infection continued and 3 reported their SARS-CoV-2 infection symptoms resolved and they remained asymptomatic until new symptoms (or recurrence of the same symptoms) began. When asked why they felt worse than before their SARS-CoV-2 infection, a wide range of symptoms were reported, including, most commonly, fatigue, pain (including myalgia and headache), “brain fog” or slowness of thinking, and anxiety and/or depression. Shortness of breath and gastrointestinal symptoms were reported less commonly. All except 1 child reported difficulty in continuing with normal activities. The duration of symptoms experienced after the initial SARS-CoV-2 infection could be estimated for only 2 children (one symptomatic at 3 and 6 months, but then asymptomatic at 9 months; the other symptomatic at 3 months but asymptomatic at 6 months); the remaining participants did not respond to invitations to re-interview at later time points (n = 8) or withdrew from the study (n = 1).
Characteristics of 11 Children and Adolescents With Chronic Symptoms After SARS-CoV-2 Infection.
Factors Associated With Chronic Symptoms After Acute SARS-CoV-2 Infection
As shown in Table 2, those with 1 or more chronic medical conditions were more likely to have chronic symptoms after acute SARS-CoV-2 infection (P = .003). Certain symptoms of the SARS-CoV-2 infection [eg, fatigue (P = .04), vomiting (P = .005), anxiety and/or depression (P < .001)] were associated with chronic symptoms, as were categories of symptoms [eg, symptoms related to the abdomen/gastrointestinal tract (P = .003) and neurologic/psychiatric symptoms (P < .001)].
Discussion
In this study of 212 infants, children, and adolescents, 11 (5.2%) experienced chronic symptomatology following acute SARS-CoV-2 infection. In some of these 11 participants, the chronic symptoms experienced represented a continuation of the original symptoms (experienced with the acute SARS-CoV-2 infection), but in other cases there was resolution of the original symptoms with subsequent recrudescence or the onset of different symptoms. Of those reporting chronic symptoms, most indicated the onset at or before 3 months following the acute SARS-CoV-2 infection. A wide range of chronic symptoms were reported by these participants, including neuropsychiatric (eg, anxiety, depression), gastrointestinal (eg, nausea, recurrent abdominal pain with vomiting), pulmonary (eg, dyspnea), and generalized symptoms (eg, fatigue). Participants with chronic symptomatology characterized their symptoms as mild to severe, and interference with normal activities because of chronic symptoms ranged from none to inability to continue these activities. Factors associated with chronic symptoms following acute SARS-CoV-2 infection were pre-existing chronic medical conditions overall, certain categories of symptoms (eg, symptoms related to the abdomen/gastrointestinal tract and neurologic/psychiatric symptoms), and specific symptoms experienced with the acute SARS-CoV-2 infection (eg, fatigue, vomiting, anxiety, and/or depression).
Data regarding chronic symptomatology after acute SARS-CoV-2 infection are sparser for pediatric populations than those for adults. However, several pediatric studies have been published with sample sizes ranging from less than 100 to over 10,000.5-25 Of note, definitions of chronic symptomatology (ie, timing of when symptoms occurred in relation to the acute SARS-CoV-2 infection and duration of chronic symptoms) varied significantly across studies. Study designs have included cross-sectional studies,5-8,16,17,22-24 prospective cohort studies,12,18,19 and retrospective cohort studies.10,14,15,21 Comparison groups were included in some studies.6,8,11,12,14,20,21,24 A wide range of chronic symptoms were reported in our study and previous studies, with the extent of functional impairment described in our study and certain others.6,13,14 Our finding of approximately 5% of participants developing chronic symptomatology after acute SARS-CoV-2 infection is less than that reported in some studies5,7,9,11,14,16,18,21,23 but similar to or more than in other studies.10,12,17,19,24 In contrast to our results, factors previously associated with chronic symptomatology include greater age,7,8,12,21 higher body mass index, 7 and longer duration of the initial SARS-CoV-2 infection. 7 We found that having 1 or more chronic medical conditions overall (but no specific chronic medical condition) at baseline was associated with chronic symptomatology, but other studies reported a past medical history (of pulmonary disease, 15 neuropsychiatric disorders 18 including schizophrenia and attention deficit hyperactivity disorder (ADHD), 17 and allergic disorders17,18) was associated with chronic symptomatology. Hospitalization for the acute SARS-CoV-2 infection,12,17 symptomatic acute SARS-CoV-2 infection, 17 and the severity of the acute SARS-CoV-2 infection 15 have been associated with chronic symptomatology. Among those with symptomatic acute SARS-CoV-2 infection, certain symptoms such as headache and arthralgia were associated with chronic symptomatology in our study, while another study 12 reported an association between the number of symptoms experienced with the acute SARS-CoV-2 infection and chronic symptomatology.
Our study was based on systematic surveillance of Vermont residents with a positive SARS-CoV-2 PCR assay. However, this study does have limitations. First, only 212 (23.7%) of those eligible agreed to participate in interviews/questionnaires at 3 and/or 6 months after the acute SARS-CoV-2 infection. Subsequently, only a small proportion of those eligible to participate in interviews/questionnaires at 9 and/or 12 months after acute infection agreed to do so, thereby limiting the ability to characterize the duration of chronic symptomatology. Of note, the response rate in our study is similar to or greater than those reported in previous studies involving the administration of questionnaires administered through telephone interviews or web-based systems.6,8 As noted above, the questionnaire utilized was not validated or pilot tested. Second, the study elicited subjective responses to questions and did not involve objective evaluations of the participants. Third, there was no control group in this study and thus it is not possible to ascribe chronic symptoms experienced by participants as the result of SARS-CoV-2 infection (ie, long COVID). Finally, eligible participants for this study had confirmed SARS-CoV-2 infection (positive SARS-CoV-2 PCR assay results) from March to July 2021 when alpha (B.1.1.7) and delta (B.1.617.2) were the predominant variants circulating in Vermont. Whether the results of this study can be generalized to other pediatric patients with infection due to subsequent SARS-CoV-2 variants is unknown.
Conclusions
Future research regarding chronic symptomatology among persons with confirmed SARS-CoV-2 infection would benefit from the use of standardized definitions of the timing and duration of chronic symptoms and from the enrollment of comparison groups. Comparing the results of different studies to date is difficult because of the lack of consistent definitions of outcomes and from the inability to determine how the prevalence of chronic symptoms among those with a history of acute SARS-CoV-2 infection compares to the baseline prevalence in a given population. Further understanding the proportion of infants, children, and adolescents with acute SARS-CoV-2 infection who go on to develop chronic symptomatology as well as the range of symptoms experienced, the severity and duration of such symptoms, and factors associated with the development of these symptoms is critical to anticipate the health care needs of this population.
Footnotes
Author Contributions
JR conceptualized the study. JR, DS, KL, and KP designed the study. DS, KL, and JL made substantial contributions to data acquisition. PC performed statistical analyses. JR drafted the manuscript. JR, DS, KL, PK, PC, JL, and ML critically revised the manuscript for important intellectual content and approved the final manuscript.
Availability of Data and Materials
The minimal data set comprises confidential and privileged public health records transmitted to the Vermont Department of Health and maintained by the Commissioner of Health pursuant to 18 Vermont Statutes Annotated (V.S.A.) § 1001(a). The data set was provided to the researchers solely for public health surveillance and epidemiological follow-up pursuant to 18 V.S.A. § 1001(d)(3), and its redisclosure is prohibited by 18 V.S.A. § 1001(e)(1)-(4).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: U.S. Centers for Disease Control and Prevention, grant number NU50CK000520.
Ethical Approval and Consent to Participate
The study protocol was reviewed by the Vermont Agency of Human Services Institutional Review Board and the University of Vermont Institutional Review Board for Human Subject Research. Both of these institutional review boards deemed the study did not meet the federal definition of research and thus did not require institutional review board approval. Therefore, in effect, the need for participants’ formal consent to participate was waived.
