Abstract
Acute necrotic encephalopathy in children is a very rare complication of severe acute respiratory syndrome coronavirus 2 infection and has rarely been reported worldwide. A 45-day-old girl was admitted to our hospital with fever and listlessness. A nose swab tested positive for the novel coronavirus nucleic acid, and her cerebrospinal fluid was positive for severe acute respiratory syndrome coronavirus 2. An early head magnetic resonance imaging scan indicated multiple abnormal signals in her bilateral cerebral hemispheres, and encephalitis was diagnosed. Twenty-three days after hospitalization, bilateral cerebral atrophy-like changes were observed on magnetic resonance imaging, with multiple softening lesions in the bilateral cerebral hemispheres, accompanied by convulsions. She was admitted to the hospital for mechanically assisted ventilation, and her condition improved after treatment of her symptoms with antiepileptic medication, anti-infection drugs, glucocorticoids, and immunoglobulins. Acute necrotic encephalopathy associated with severe acute respiratory syndrome coronavirus 2 infection in children should be detected and treated as early as possible. Satisfactory short-term efficacy can be obtained, but long-term neurological sequelae often linger.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can cause certain immune-mediated neurological diseases, including acute encephalitis, acute disseminated encephalomyelitis (ADEM), Guillain–Barré syndrome, myelin oligodendrocyte glycoprotein antibody disease, and anti-n-methyl-D-aspartic receptor encephalitis. 1 Encephalitis is a rare complication of SARS-CoV-2 infection. In the adult population, the neurological manifestations caused by coronavirus disease 2019 (COVID-19) include headache, dizziness, disturbance of consciousness, ataxia, hyposmia, anosmia, and seizures. In contrast, febrile convulsions and meningoencephalitis are more common in the pediatric population. 2 In a cohort study of 1695 patients aged <21 years who were hospitalized with COVID-19 between March and December 2020, only 15 (nearly half of whom were diagnosed with pediatric multisystem inflammatory syndrome) were found to have severe encephalopathy, and only 8 had acute central nervous system inflammation involving encephalitis, aseptic meningitis, or acute disseminated encephalomyelitis. 3
Acute necrotizing encephalopathy (ANE) is a very rare complication of secondary infection with SARS-CoV-2. We present a rare case of ANE in a young child with COVID-19, diagnosed by clinical features and neuroimaging.
Case presentation
A 45-day-old female infant was admitted due to fever for 2 days, lethargy for 1 day, and aggravation for a few hours. Two days prior to admission, the patient had a fever with a maximum body temperature of 38.5 °C, no convulsions, and occasional coughing. After treatment with ibuprofen, the temperature was reduced to normal; however, there was still recurrent low fever and discomfort such as loose stools (no vomiting), which were not considered serious. One day prior to admission, the child developed mental exhaustion, poor feeding, difficulty sleeping, and lack of activity, and was immediately admitted to the hospital. This was the mother’s second child, born naturally at a gestational age of 39 weeks, and with a birth weight of 2600 g. At birth, due to “aspiration pneumonia,” the infant was hospitalized in the intensive care unit of a local hospital and discharged after improvement (specific details were not available). The patient’s mother tested positive for COVID-19 and was asymptomatic. The patient was admitted to hospital with an altered respiratory rhythm at a slower rate, apnea, cyanosis of the oral mucosa, and SpO2 88%–92%. A diagnosis of respiratory failure was considered and assisted ventilation with tracheal intubation and a ventilator was started. Physical examination revealed the following: temperature 36.7 °C, heart rate 150 bpm, respiratory rate 40 times/minute, blood pressure 71/42 mmHg, weight 4 kg, SpO2 99%; fontanel tension degree, soft; pupils, enlarged, equally round, left 3.0 mm, right 3.0 mm, sensitive to light reflection; muscle strength under analgesia and sedation, not cooperative; and muscle tone, normal. Otherwise, the physical examination was normal.
A nasal swab polymerase chain reaction test was positive for SARS-CoV-2 nucleic acid. A routine blood test revealed the following: white blood cells, 13.2 × 109/L (reference value, 4.3–14.2 × 109/L); hemoglobin, 101 g/L (reference value, 97–183 g/L); platelets, 598 × 109/L (reference value, 183–614 × 109/L); lymphocytes, 21.1% (reference value, 20%–40.0%); neutrophils, 63.6% (reference value, 26.0%–83.0%); hypersensitive C-reactive protein, <0.50 mg/L (reference value, 0.00–10.00 mg/L); procalcitonin, 0.30 ng/mL (reference value, 0–0.05 ng/mL); renal function/myocardial enzymes/electrolytes, normal; and blood culture and sputum culture, negative. A 1d lumbar puncture suggested a brain pressure of 150 mmH2O (reference value 0–100 mmH2O). A routine cerebrospinal fluid and biochemical assay revealed the following: the fluid was colored light yellow, clear and transparent, and had 114.1 mmol/L chlorine (reference value, 120.0–130.0 mmol/L), 1.60 mmol/L glucose (reference value, 2.80–4.50 mmol/L), and 0.64 g/L↑ of cerebrospinal fluid protein (reference value, 0.15–0.45 g/L); the number of nucleated cells was 6.00 × 106/L (reference value, 0.00–15.00 × 106/L; laboratory parameters are detailed in Table 1).
Detailed laboratory parameters.

During hospitalization, three rounds of cerebrospinal fluid biochemical review were conducted, and only one instance of slightly higher protein levels was noted. RNA (ribonucleic acid) pathogen detection in the cerebrospinal fluid (assessed by Difei Medical) revealed the following: SARS-CoV-2 positivity (sequence number 69), with no obvious abnormality in DNA (deoxyribonucleic acid) pathogen detection. A 1d head magnetic resonance imaging (MRI) showed multiple abnormal signals in bilateral cerebral hemispheres, raising the possibility of encephalitis (Figure 1). The patient was admitted to the neonatal intensive care unit for tracheal intubation and mechanically assisted ventilation. Ceftriaxone was administered as an anti-infection agent, mannitol dehydration as a cranial pressure reduction agent, and low-dose hormone (methylprednisolone sodium succinate 2 mg/kg/d)- and high-dose immunoglobulin (2 g/kg)-based therapies.
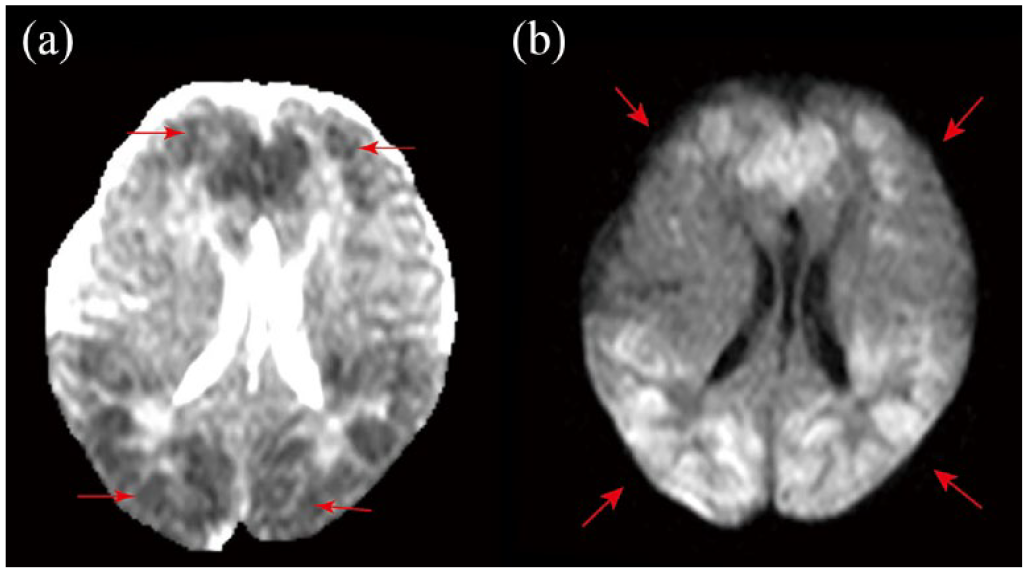
Multiple patchy and small patchy abnormal signals observed in both cerebral hemispheres, showing low signal on T1-weighted imaging (T1WI), high signal on T2-weighted imaging (T2WI), and high signal on diffusion-weighted imaging (DWI), suggesting the possibility of encephalitis.
On the 3rd day of admission, the patient developed twitching at the corners of her mouth and upper limbs, with her eyes staring upward during the twitching, lasting for about 10 seconds; this would then cease spontaneously. No convulsions recurred following anticonvulsive treatment with levetiracetam. Electroencephalography (EEG) showed abnormal results, including multifocal sharp wave, sharp slow wave, spinous wave, multispinous wave, fast wave, slow wave emission, sleep period, and frequent left or right central area focal onset or electrical attack. EEG on day 11 revealed an abnormal topographic, diffuse, and multifocal sharp wave, sharp slow wave, spinous wave, slow wave, and fast wave release. A head MRI on day 8 showed multiple abnormal signals in the bilateral cerebral hemispheres, basal ganglia region, thalamus, and corpus callosum, indicative of encephalitis (Figure 2). Another head MRI at 19 days showed bilateral brain atrophy-like changes and multiple softening foci in bilateral cerebral hemispheres; these were considered residual changes after brain injury (Figure 3). A test for RANBP2 mutation was negative. A nucleic acid test of the patient’s cerebrospinal fluid was negative 19 days after admission, and her condition improved 23 days after admission, at which point she was discharged. After discharge, there were no more convulsive attacks, but the muscle tension of the patient’s limbs remained on the high side. As of the writing of this report, the patient was receiving levetiracetam orally to control convulsions, and the rehabilitation department was performing regular limb rehabilitation treatment.

(a and b) Multiple patchy and small patchy abnormal signals observed in bilateral cerebral hemispheres, basal ganglia, and thalamus showing low signal on T1-weighted imaging (T1WI), high signal on T2-weighted imaging (T2WI), and high signal on diffusion-weighted imaging (DWI) (c and d). Abnormal signal of the corpus callosum.
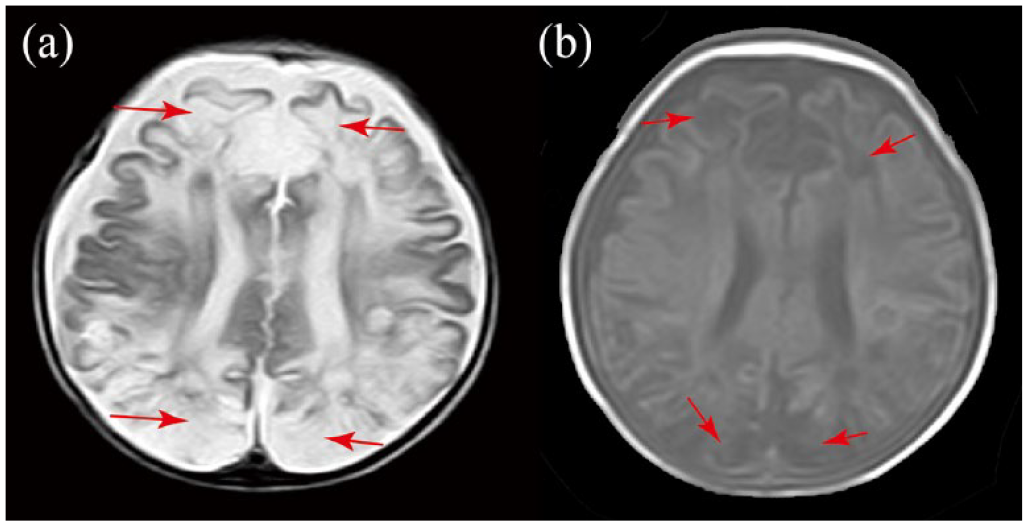
(a and b) The bilateral brain is symmetrical and small in size, and the lamellar T1-weighted imaging (T1WI) shows low signal, the T2-weighted imaging (T2WI) shows high signal, and the diffusion-weighted imaging (DWI) shows low signal, the bilateral basal ganglia is reduced in shape, the structural boundary is unclear, and the bilateral cerebral sulci, brain cleft, and extracerebral space are generally widened, suggestive of bilateral atrophic-like brain changes. There are multiple softening foci in bilateral cerebral hemispheres.
Discussion
ANE has a unique neuroimaging pattern, including changes in the thalamus, medial temporal lobe, pons, and medullary, as well as in the striatum and subcortical peripherals. 4 In 2021, Claudia Lazarte-Rantes et al. 5 first reported a case of ANE associated with SARS-CoV-2 infection in a pediatric patient in the United States. The 14-month-old boy presented with fever, irritability, left focal seizure, and febrile focal seizure status as the first symptoms that improved after symptomatic management such as mechanical ventilation; however, severe neuropsychiatric symptoms remained. Clinical studies have shown that the onset of ANE in children peaks between 1 and 3 years of age; furthermore, it shows a male predominance. The main manifestations include fever (100%), seizures (86%), and disturbance of consciousness (100%). 6 Due to the low incidence of COVID-19 associated ANE in children and few reported cases, we found through searching the database that the age of onset of the disease ranged from 5 weeks to 13 years old, and almost all patients had fever accompanied by consciousness disorder. Most patients were treated with high-dose immunoglobulin and methylprednisolone, while a few patients were treated with tolumab. Most of the patients had residual sequelae of neurological damage (see Table 2). MRI reveals increased T2 and T1 signal densities in multifocal and symmetric brain lesions (bilateral thalami, 100%) in all patients during the acute phase. Most patients have severe neurological sequelae.
Characteristic of included stduies.

IVIG: intravenous immunoglobulin.
The pathogenesis of COVID-19-related neurological diseases is still unclear, and recent studies suggest that it is related to the neurotropic properties of SARS-CoV-2. 14 According to this theory, the direct neuroinvasive effect of the virus could be explained by its retrograde movement into the central nervous system (CNS) through the olfactory or peripheral lung nerves or by hematogenic migration through CNS endothelial cells that express angiotensin-converting enzyme 2 receptors. 15 Common viruses responsible for this include herpes simplex virus, adenovirus, SARS-CoV-2, influenza virus, human rhinovirus, and others. 16
However, the underlying pathogenesis of ANE is often attributed to immune-mediated mechanisms rather than direct viral cytopathy effects. A strong systemic immune response triggered by a prior infection produces an inflammatory cytokine storm that leads to a breakdown of the blood–brain barrier, followed by edema, pestectomy, and necrosis. 17 Scholars have found that interleukin-8 and tumor necrosis factor-α levels increase in COVID-19-related encephalopathy, but decrease with clinical improvement. 18
It has been found that ANE is related to RANBP2 mutation, which shows familial inheritance. In 2009, Neilson et al. 19 published a landmark article which reported that many family members suffered from ANE. The RANBP2 gene is located on chromosome 2q2-11, and RANBP2 mutation is inherited in an autosomal dominant manner. However, incomplete penetrance is reported (40%). RANBP2 encodes a nuclear porin that plays a role throughout the cell cycle, and mutations in RANBP2 are closely associated with ANE through multiple processes, including mitochondrial function, viral entry, cytokine signaling, immune response, and blood–brain barrier maintenance. 17
ANE with RANBP2 mutations has a poor prognosis. Lee et al. 20 first reported about familial ANE with RANBP2 mutations in East Asia, reporting that both female children died. Pongpitakmetha 21 first reported the case of a previously healthy 29-year-old adult with COVID-19-associated ANE and RANBP2 mutation who died within 6 days after treatment. Due to genetic susceptibility, ANE with RANBP2 mutation can be recurrent. 22 Forest 23 first reported the case of a 10-year-old girl who was infected with the new coronavirus and experienced necrotizing encephalopathy. The child was ultimately diagnosed with a RANBP2 gene mutation and admitted to the pediatric intensive care unit at the age of 2 years due to fever, flu symptoms, and insanity. A brain MRI revealed hyperintensity of the external capsules, cerebellar cortex, hypothalamus, and pons on fluid-attenuated inversion recovery sequences with restricted differentiation on diffusion-weighted images (DWIs). The child failed to recover and was in a vegetative state. Children with ANE should be tested for RANBP2 mutations to determine if they are at risk for recurrent ANE attacks. Our patient in this case was negative for RANBP2 mutations; hence, she had a relatively good prognosis.
The clinical course of ANE can be divided into the prodromal stage, acute encephalopathy stage, and convalescence stage. The prodromal symptoms of ANE are nonspecific and variable due to viral infection and include fever, upper respiratory tract infection, chest infection, diarrhea, vomiting, and headache. As ANE progresses, seizures, disturbances of consciousness, and focal neurological deficits may manifest as brain dysfunction. The common clinical manifestations of ANE are seizures, encephalopathy, and dysmotility, which progress rapidly within days, with a fatality rate of up to 30% despite aggressive immunomodulatory therapy. Patients who survive usually have poor neurological outcomes, with <10% of survivors recovering completely. 17
Treatment options include early intensive care, supportive therapy (such as seizure treatment and increased intracranial pressure control), antiviral therapy, intravenous corticosteroids, intravenous immunoglobulin (IVIG), and immunomodulators. Steroid administration early in the disease course may improve clinical performance by reducing cytokine storms and metabolic dysfunction, as well as relieving inflammation. Khan 24 also reported the case of a 5-week-old infant who received high-dose intravenous methylprednisolone, tozizumab, and IVIG treatment, showing dramatic short-term clinical improvement but long-term sequelae. However, in our study, the child was treated with low-dose methylprednisolone and high-dose immunoglobulin in the early stage of the disease and showed drastic clinical improvement in the short-term, but there were still residual paroxysmal convulsions and other neurological sequelae.
This case describes the youngest patient with COVID-19-related necrotizing encephalopathy reported in Asia to date, and the prognosis resulting from SARS-CoV-2 infection is indistinguishable from those resulting from other infections such as herpes simplex viruses, adenovirus, neocoronavirus, influenza virus, and human rhinovirus. With regard to therapeutic aspects, the early use of corticosteroids, IVIG, and biological agents according to cytokine examination are currently good choices.
Conclusions
Based on known case reports globally, ANE in pediatric COVID-19 is a rare disease with poor prognosis and high fatality rate, whereas the youngest case of ANE in pediatric COVID-19 reported to date in Asia had a relatively good prognosis. However, it is not possible to determine whether the young age of the patient resulted in the better prognosis. Further studies on larger samples are required to draw any conclusions. However, early detection, diagnosis, and treatment are key factors to improve prognosis.
Footnotes
Author contributions
J.G.W. and X.Z.L. developed the concept for the article. J.G.W. and Y.P.T. wrote the article. J.G.W. and X.Z.L. revised and approved the final draft.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Self-Funded Scientific Research Project of Guangxi Zhuang Autonomous Region Health Commission (nos. Z20210123) and The Guangxi Science and Technology Program Project (Guike AD22035121).
Availability of data and materials
The data used during the study are available from the corresponding author upon request.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the minor(s)’ legal guardian/next of kin for the publication of any potentially identifiable images or data included in this article.
