Abstract
Objective. Despite the remarkable progress made in the understanding and treatment of childhood nephrotic syndrome (NS), a lot is still unknown about its epidemiology in many African countries. This study sought to determine the clinicopathological features and treatment outcomes of children with NS at the largest tertiary hospital in Botswana. Methods. A retrospective study of 26 children with NS treated from 2009 to 2014 was conducted. Results. Mean age at presentation was 5.96 ± 3.06. Hematuria was found in 92.3%, low C3 in 26.7%, high creatinine in 19.2% and hypertension in 46.2% of the patients. 92.3% had primary NS out of which 23.1% had familial NS. HIV, tuberculosis and hepatitis B infections were diagnosed in 3.85%, 9.09% and 4.16% respectively. 69.2% had steroid-resistant nephrotic syndrome, focal segmental glomerulosclerosis (SRNS-FSGS). Conclusion. The frequency of primary SRNS-FSGS and familial SRNS appears to be much higher in Botswana highlighting the possibility of genetic causes.
Keywords
Introduction
Nephrotic syndrome (NS) remains an important cause of chronic kidney disease (CKD) in children. There is evidence that NS in children differs with age, sex, race or ethnicity and region. Our current understanding and therapeutic approaches of childhood NS were pioneered by a multinational investigative group established in 1965 called “International Study for Kidney Disease in Children” (ISKDC). The ISKDC found that the vast majority of children with NS in their study showed minimal change nephrotic syndrome (MCNS) on kidney biopsy and they respond to steroids. 1 For years, the management of children with NS has been based on the ISKDC recommendations where children with NS are empirically treated with corticosteroids, before a kidney biopsy is performed. Notably, the ISKDC studies were made up of predominantly Caucasian and Asian children from North America, Europe, and Asia. 1
Several studies have shown that the epidemiological pattern of NS in black children of African descent differ from ISKDC findings. A high prevalence of focal segmental glomerulosclerosis (FSGS) and poor response to steroids has been observed in these children. 2 There has been an increasing attention to genetics as one of the reasons to explain these geographic differences in the etiology of NS. Impressive progress has been made over the years toward our understanding of genetic causes of childhood NS. This has led to the development of targeted therapies against resultant mutant proteins. Despite this remarkable progress, a lot is still unknown about the epidemiology of childhood NS in many African countries and treatments are still based on studies undertaken outside the continent. Pediatric Nephrologists in Africa have since established a need to conduct studies in the continent in order to formulate guidelines specific to their patients. One such study is the multi-center multi-country collaborative research project of the Human Hereditay and Health in Africa Kidney Disease Research Network (H3AKDRN) which aims to address this knowledge gap. 3 In other African studies, infections such as malaria, tuberculosis (TB), Human immunodeficiency virus (HIV), hepatitis B and schistosomiasis have been suggested to be some of the major secondary causes of NS in the pediatric population.4-6
The difference in patterns of NS suggest that NS is affected by circumstances such as genetics, incidences of systemic diseases or infections, climatic and socio-economic conditions unique to a country. The epidemiology of childhood NS has not been described in Botswana. While there is a need to carry out an international study to validate the continued relevance of the ISKDC, it is equally critical to determine if these findings are relevant for the local population in Botswana. Given the high TB and HIV prevalence in Botswana, it was also important to find out if these infections also contribute to cases of NS in Botswana.7,8 The primary objective of this study was to describe the clinical characteristics, pathologic features, and treatment outcomes of children with NS at Princess Marina Hospital, Botswana.
Material and Methods
This was a retrospective descriptive study of all children treated at Princess Marina Hospital (PMH) from January 2009 to December 2014 with a diagnosis of NS. This is the largest and busiest government referral hospital in Botswana, which provides both secondary and tertiary level services. Included in this study are all children aged between 1 year and 13 years with the diagnosis of NS treated during the study period. Exclusion criteria included patients with a diagnosis of congenital and infantile NS, non-citizens and grossly incomplete files
The following patient information was extracted from medical records: (1) Age at presentation, gender, initial blood pressure, results of the urine analysis. (2) Laboratory data at presentation included, serum albumin, serum creatinine, cholesterol, hepatitis B and C, HIV, antinuclear antibodies (ANA), anti-streptolysin O titer (ASOT), complement 3 and 4 and TB screening (3) Response to initial steroid therapy (documented as either steroid-sensitive NS or steroid-resistant NS), and (4) Histopathology results. Kidney biopsy was done in all patients who failed to respond to steroid therapy or those who presented with atypical symptoms.
Definitions
The definitions were based on Princess Marina Hospital guidelines. At the time of the study, these guidelines were similar to the commonly used definitions of NS in many parts of the world.9-13 The definitions have since been revised and published by the International Pediatric Nephrology Association (IPNA) in 2020. 14
Nephrotic syndrome
Proteinuria of 3+ or 4+ on urine dipstick in a first morning void or urine protein creatinine ratio of ≥2 mg/mg on the first morning sample or >40 mg/m2/hour of protein in 24-hour urine sample, hypoalbuminemia ≤ 25 g/l and edema. It should be noted that the results for urine protein creatinine ratio (UPCR) ratio were not available for majority of the patients and urine dipstick results were used instead.
On confirmation of the diagnosis of NS, patients were placed on a course of steroids 2 mg/kg/day (maximum 60 mg/day) for a period of 4 to 6 weeks, following which responsiveness to treatment could be determined.
Hypertension
The definition of hypertension is according to the fourth report of the National High Blood Pressure Education Program (NHBPEP) Working Group on Children and Adolescent. NHBPEP defines pediatric hypertension as systolic and diastolic blood pressure in the 95th percentile or greater for age, sex and height. In this study if the diagnosis of hypertension was made by a pediatrician, it was assumed that it was in accordance with the NHBPEP guidelines. 15
Sample Size
Our sample was calculated using the one-sample population proportion formula taking the prevalence of steroid-resistant NS at 20% from a study done in Nigeria. 16 A margin of error of 10% was adopted and after the correction for finite population, the final calculated sample size was 27, but finally 26 patients were recruited. Due to logistical challenges, 10% instead of the traditional 5% was used.
Statistical Analysis
Data analysis was conducted using statistical software STATA version 14.0. The population demographics and laboratory results were summarized using simple descriptive statistics such as mean ± SD. Percentages were used for categorical data. Dichotomous variables were compared using the fisher exact test. Continuous variables were compared with the Wilcoxon rank sum. P-value <.05 was considered significant.
Ethical Approval and Informed Consent
This study was approved by the University of Botswana Institutional Review Board (IRB) UBR/RES/IRB/269, Princess Marina Hospital IRB PMH 5/79(259-1-2016), and Ministry of Health- Botswana HPDME/Research Division: 13/18/1 Vol. X (610). The IRBs waived the need to obtain consent for the collection, analysis and publication of the retrospectively obtained and anonymized data for this non-interventional study.
Results
Patient Demographic Characteristics
A total of thirty-two (32) patients were admitted with a diagnosis of NS over the study period. All patients included in the study were Botswana citizens and were of the black race. Six (6) patients met the exclusion criteria, as shown in Figure 1. The age at presentation was categorized into 3 groups, as shown in Table 1. Their ages ranged from 1.42 to 12 years, with a mean age of 5.96 ± 3.06 years. There was an overall female predominance with a male: female ratio of 1:1.6. Patients in the age group between 5 and <10 years accounted for the majority of the patients.

Flow diagram of participants in the study.
Comparison of Demographic and Clinical Features at Presentation in Different Age Groups.

Serum creatinine values standardized for age 1 to <5 years (17.7-70.7), 5 to <10 years (17.7-79.6) and 10 to 13 years (26.5-106.1).
Results for C3 and C4 were available for 15 patients, n = 15. For age 1 to < 5 years (n = 6), 5 to <10 years (n = 8); 10 to 13 years (n = 1).
Clinical Characteristics and Laboratory Results at Presentation
The clinical and laboratory results of the patients at presentation are shown in Table 1. The most frequent presenting complaint was generalized edema, with 15 out of 26 (57.7%) presenting with severe hypoalbuminemia. Microscopic hematuria of varying degrees on urine test strip were observed in 24 (92.3%) patients. Patients whose ages were ≥10 years had highest frequency (100%) of hematuria. A breakdown of the prevalence of hematuria in the sample population is shown in Table 2. Hypertension was present in 12 (46.2%) patients at presentation and all of them required treatment with antihypertensive drugs. Results for C3 and C4 were available for only 15 patients. In this study the frequency of patient presenting with low C3 levels was 4 (26.7%). At presentation serum creatinine and urea were elevated in 5 (19.2%) and 10 (38.5%) of patients respectively. There was no significant difference (P < .05) found between the laboratory characteristics at presentation for the 3 age groups.
Breakdown of Children With Hematuria at Presentation.

Secondary causes of nephrotic syndrome
Regarding secondary causes of NS, this study looked at HIV, Hepatitis B and TB infections. Given the high TB prevalence and HIV in Botswana, it was important to find out if there are any association of TB and HIV to childhood NS in Botswana.7,8 All patients were tested for HIV on admission and one (1) was HIV positive from vertical infection and had been on antiretroviral drugs for approximately 9 years. Clinical evaluation of TB was available for 22 patients and 2 (9.09%) of these patients were diagnosed with TB disease. The results for hepatitis B were available for 24 patients, and only 1 (4.16%) patient tested positive for hepatitis B. It is important to note that 1 patient tested positive for all the 3 infections. If a patient had no evidence of HIV, hepatitis B and TB infections or there was no systemic disorder identified, primary NS was considered. No secondary etiology was elicited for 24 (92.3%) patients and were classified as primary NS.
Treatment Outcome
All patients received the initial steroid and once stable they were discharged from the hospital before completion of their 4-week treatment. Some were discharged to continue follow-up care in their referring hospitals. At the time of the study, our assumption was that they were all steroid sensitive. Those who did not attain remission were usually referred to Princess Marina Hospital. It is possible that the group with unknown treatment outcomes could have attained remission and therefore, would have not required referral back to PMH. It is recognized that this assumption may not be true for all the 8 patients as some might have been SRNS and truly lost to follow-up.
Since all the 18 patients with known treatment outcome did not respond to steroid therapy (SRNS), the group was further compared with the unknown treatment outcome (presumed SSNS) as shown in Table 3. This was to determine if one can use clinical and laboratory characteristics at the time of diagnosis to predict the treatment outcome or clinical course for the population under study. The characteristics used are those commonly described in the literature as predictors of poor response to steroids. For each of these characteristics, with the exception of mean age at presentation, there was no significant statistical difference (P < .05) between SRNS and presumed SSNS. Kidney biopsies were performed on the 18 patients who did not respond to therapy. Kidney biopsies revealed FSGS in all the 18 patients.
Comparison Between Steroid-Sensitive Nephrotic Syndrome and Steroid-Resistant Nephrotic Syndrome Using Clinical and Laboratory Characteristics at Presentation.

Low C3 (n=15) where Presumed SSNS (n=4), SRNS (n=11).
Positive Family History of Nephrotic Syndrome (Familial NS)
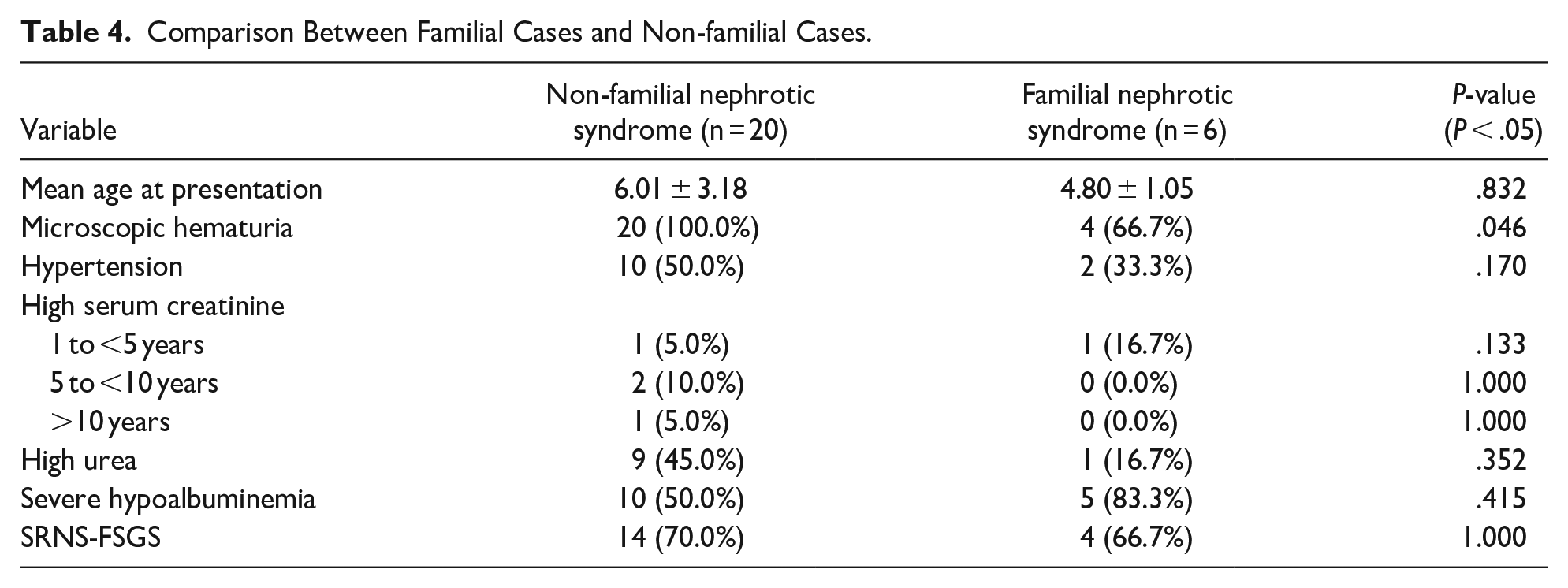
Six (23.1%) patients were sibling pairs. Two sibling pairs had familial SRNS-FSGS, and the remaining 2 siblings had no follow-up information on their response to initial steroid therapy. They were included in the presumed SSNS group. Table 3 shows comparison between those with positive family history of NS and those without using their clinical and laboratory features. As shown in Table 4, except for the presence of hematuria at presentation, there was no statistically significant difference between those with a positive family history of NS and those without a family history of NS.
Comparison Between Familial Cases and Non-familial Cases.
Discussion
Despite being a common cause of CKD and chronic kidney failure in children, childhood nephrotic syndrome has not received significant attention in many African countries. This could largely be because of a lack of data describing its epidemiology in these countries. This study is a description of 26 children with NS treated between January 2009 and December 2014 at the largest tertiary hospital in Botswana. All children included in the study were Botswana citizens and were of the black race.
There was a female predominance with a male: female ratio of 1:1.6 in contrast to studies from South Africa, Egypt, Iran and Saudi Arabia where a male preponderance has been reported.11,17-19 The small sample size might explain why we found a female preponderance. However, some of the studies with smaller sample sizes similar to this study also demonstrated a high male-to-female ratio, which is consistent with studies with larger sample sizes. 17 Regarding the mean age at presentation, most studies have shown inconsistent results. This study observed mean age at presentation of 5.96 ± 3.06, which is comparable to a study done in Libya (mean age of 5.7), but lower than in Benin, Nigeria (8.2 ± 0.5), and Pakistan (9.97 ± 4.59) and higher than Iran (4.87 ± 3.24) and Egypt (4.43 ± 2.7).18,20-23
More than half (57.7%) of patients presented with severe hypoalbuminemia (defined as serum albumin of <15g/L) and there was no difference noted across age groups. A potential explanation for the high percentage of patients presenting with severe hypoalbuminemia is the delay in making the diagnosis by the healthcare workers. Almost all patients were incorrectly diagnosed as allergies. Impaired kidney function, hematuria, low C3 level and hypertension are atypical features of NS, and when present, the outcomes are poor compared to the classical presented NS.24,25 In this study, only 19.2% of patients had elevated serum creatinine similar to 18.6% found in the Namibian study and lower than 32% found in Benin-City, Nigeria.4,20 This is another possible reflection of late diagnosis. Hematuria was observed in 92.3% of patients compared to 70.3% found in Namibian children. Interestingly, all children with SRNS had hematuria at presentation. Hypocomplementemia (low C3) was observed in 26.7% of patients, which is higher than 6% and 7% reported from Cairo, Egypt and Benghazi, Libya, respectively.22,23 Some studies have noted that patients who had hematuria and hypocomplementemia at presentation showed poor response to steroid therapy. This study corroborates these findings. The prevalence rate of hypertension was 46.2% which is similar to the 41.4% found in Benin, Nigeria. 20 A lower prevalence rate (21.0%) of hypertension was reported in Egypt. 23 From these findings it does seem a significant number of our patients had atypical and more severe presentations. An atypical NS is associated with a more aggressive course, with faster deterioration in kidney function than typical NS.
No etiology was found in 24 (92.3%) patients. This finding makes primary NS the most common type of NS in this population, which is consistent with the literature.26,27 In African studies, infections such as malaria, TB, HIV, hepatitis B and schistosomiasis have been suggested as the major secondary causes of NS in children. Studies by Ramsuran et al 5 and Ademola et al 28 found that NS was a common presentation of kidney disease in HIV- infected children. During the study period, Botswana had the third highest HIV prevalence in the world. 8 Despite this high prevalence rate only 1 (3.85%) was found to have HIV-associated NS. In Botswana, there is the provision of universal antiretroviral drugs with more than 95% antiretroviral coverage among children living with HIV. 8 This could explain the low incidence of HIV-associated NS. Similarly, there was no significant association of NS to Hepatitis B (4.16%). This was in contrast to findings in Namibia where an association of NS and Hepatitis B was found to be one of the major causes of secondary NS. 4 The main contributing factor cited was the exclusion of the Hepatitis B vaccine in their national immunization program. Hepatitis B vaccine has been part of the Botswana Child Immunization Schedule since 2005 and this could be the reason for the low incidence rate of Hepatitis B infection in this study.
Eighteen (at least 69.2%) children did not respond to steroids (primary SRNS). What is surprising is that all the patients with SRNS who had a biopsy done showed only FSGS (69.2%) and not any other glomerular lesions, such as membranous glomerulopathy, membranoproliferative glomerulonephritis (MPGN), MCNS, etc. This is similar to studies done in both African Americans and in black Africans where FSGS was common, and patients had poor response to steroid therapy.2,11,27 This may be explained by environmental, ethnic or genetic factors unique to the population under study. However, it is also possible that we might not be seeing other glomerular lesions if children with SSNS are successfully treated at lower levels and never get to reach tertiary facilities. The indications for kidney biopsy at Princess Marina Hospital are (1) Age at presentation <12 months and >10 years, (2) SRNS, (3) SDNS, (4) frequent relapse, and (5) Atypical presentation. These strict criteria might have also influenced histopathology found in this study. Due to unavailability of electron microscopy (EM), the kidney biopsies were not evaluated by EM. It is possible that pathologies that could only be picked up by EM such as Alport syndrome or thin basement nephropathy were missed.
Six (23.1%) had familial NS. This is higher than in Egypt where 5% of patients had a positive family history. 23 There has been a growing interest in primary SRNS and familial NS because of their association with a genetic etiology and the treatment challenge they pose to nephrologists all over the world.2,29 The International Pediatric Nephrology Association recommends genetic testing for children with familial SRNS and primary SRNS before biopsy. 14
For many years, studies have tried to determine what clinical and laboratory characteristics at presentation can be used to predict either the treatment outcome with steroids or the glomerular histopathology. As previously mentioned, the presence of hematuria, hypertension, low complement levels (C3 and C4) and high creatinine at presentation have been interpreted as atypical presentation and these indicated a poor prognosis (poor response to steroids or histopathology other than MCNS).24,25 A significant proportion of patients had atypical presentation that is, age at presentation >5 years (61.5%), female predominance (61.5%), hematuria (92.3%), hypertension 12 (46.2%), low C3 (26.7%), and high creatinine 5 (19.2%). All characteristics except for the mean age at presentation (P = .028) were found to have no statistically significant differences between the children who had a biopsy (SRNS) and those (presumed SSNS) whose outcome was unknown. These results need to be critically considered as the sample size may have been too small to allow accurate analysis and to find significant relationships from the data. However, even in a multicenter study with a large cohort like the ISKDC study, prediction of histopathology from clinical and laboratory characteristics was far less accurate than generally believed. This study found a significant difference only when comparing MPGN to MCNS or FSGS.24,25 Similarly, some of the studies with smaller cohort compared to ISKDC also found little or no statistically significant difference to predict histopathology or treatment outcome using clinical and laboratory characteristics. For example, the Chinese study looked at fourteen clinical and laboratory characteristics and 13 out of 14 showed no statistically significant difference. 25 It is therefore reasonable to believe that our findings might not change even if we increase our sample size.
This study has important limitations inherent to its retrospective design and the small sample size. Since this study relied heavily on existing data from patients records, some of the limitations encountered in this study included poor documentation and record keeping. Another key limitation of the study is that it was unable to ascertain the treatment outcome of 8 patients who were discharged before completion of their 4-week initial steroid therapy and had no follow up data. Owing mainly to resource constraints in our hospitals, there is no electron microscopy (EM) and genetic testing in the country and these tests were not done for the patients. Since our patients present with atypical symptoms, EM would have been beneficial.
Conclusion
This study has shed some light on the etiological profile and pattern of childhood NS in Botswana. The results from this study support that the clinicopathological features and treatment outcomes of children with NS in Botswana are similar to other countries in the Sub-Saharan region. However, the frequency of primary SRNS-FSGS and familial NS appears to be much higher in Botswana compared to other African countries, highlighting the possibility of genetic causes, and therefore, the need for genetic testing in our setting. Genetic testing will provide an accurate diagnosis and prognosis and guide in choosing appropriate or targeted therapy. The study also highlights the critical need for EM in identifying pathologies not visible on light microscopies.
Footnotes
Acknowledgements
We would like to thank Mr Joseph Joseph, librarian at the University of Botswana for his assistance in the literature search. We would also like to acknowledge the doctors and nurses who cared for these patients.
Author Contributions
All authors have made significant contribution to the concept and design of this study; data collection, analysis and interpretation; drafting and revision of the manuscript. TDM: Conceptualization and study design, proposal writing, data collection and analysis, discussion, drafting and editing the manuscript for publication. TAM: Conceptualization and study design, data analysis, discussion and editing the manuscript for publication. ER, LM and DJ: Conceptualization, study design, discussion and editing the manuscript for publication. TVM: Data analysis, discussion and editing the manuscript for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was approved by the University of Botswana Institutional Review Board (IRB) UBR/RES/IRB/269, Princess Marina Hospital IRB PMH 5/79(259-1-2016), and Ministry of Health-Botswana HPDME/Research Division: 13/18/1 Vol. X (610). The IRBs waived the need to obtain consent for the collection, analysis and publication of the retrospectively obtained and anonymized data for this non-interventional study.
