Abstract
Objectives. We aimed to compare the prevalence of co-infections, pathogens, and factors associated with SARS-CoV-2 acute respiratory infection (ARI) and non-SARS-CoV-2 ARI, among hospitalized children. Methods. We conducted an observational cross-sectional study of hospitalized children <15 years with ARI, and lasting respiratory symptoms <14 days, using polymerase chain reaction on nasopharyngeal specimens. Results. Of the 184 children with ARI analyzed, 122 were infected with SARS-CoV-2 and 62 were not. SARS-CoV-2 ARI had a significantly lower rate of co-infection than non-SARS-CoV-2 ARI (2.5% vs14.5%, P = .003). SARS-CoV-2 ARI children were significantly associated with a less empirical antibiotics (aOR = 0.09, CI = 0.03-0.21; P = .000), more pneumonia (aOR = 5.15, CI = 1.77-14.95; P = .003), and more abnormal chest X-ray (aOR = 2.81, CI = 1.38-5.71; P = .004). Conclusions. Although SARS-CoV-2 ARI in hospitalized children was associated with pneumonia and abnormal chest x-rays, empirical antibiotics may not be necessary for treating mild to moderate cases.
Introduction
Acute respiratory infections (ARIs), particularly in young children, account for 20% to 40% of hospital admissions globally and are one of the leading causes of morbidity and mortality.1,2 ARI can cause non-severe but widespread epidemics, contributing to the continuous circulation of pathogens within communities. According to the World Health Organization (WHO), ARI is defined as a sudden onset of respiratory symptoms like cough, coryza, fever and shortness of breath. 3 Multiple viral infections are more common in pediatric patients than in adult individuals. 4 A rise in hospital admissions, admissions to intensive care units, pediatric intensive care units (PICU), prolonged hospital stays, need of oxygen support and prolonged use of mechanical ventilation are associated with several viral infections.5-8 However, some studies indicated that disease severity, management, and outcome were not associated with multiple viral infections.9,10 In hospitalized children, especially those who are younger, and attend daycare, the incidence of simultaneous detection of multiple viruses alongside ARI typically varies between 10% and 30%.9,11-13 Moreover, the prevalence of respiratory viruses during episodes of ARI can range widely from 15% to 90%, depending on the detection methods employed and the breadth of viral investigation.14,15 The SARS-CoV-2 pandemic has necessitated significant changes to our clinical practice with regard to the treatment of children with ARI including circulate with other respiratory viruses, increasing or decreasing the probability of co-infections.16,17 The co-infection of other viral and bacterial agents in SARS-CoV-2 infected patients can lead to adverse outcomes in the form of increased mortality or prolonged hospitalization in these patients. Several studies have documented unfavorable outcomes in cases where viral co-detection occurs among children. 18 Empirical antibiotics have been administered to nearly 90% of the patients throughout the pandemic. 19
However, the diagnostic gap for SARS-CoV-2, other viruses, and co-infections has narrowed significantly in recent years. According to a previous study, rapid nasopharyngeal swab (NPS) tests have enabled doctors to diagnose viral infections with a positive predictive value of 88.6% and a negative predictive value of 75.0%. Swift NPS testing for respiratory viruses has also emerged as a valuable tool for confirming viral infections in children with ARI, minimize unnecessary antibiotic use in patients with SARS-CoV-2 and other viruses, especially in cases of mild illness. 20
The most commonly reported symptoms of SARS-CoV-2 infection include fever and cough, often accompanied by rhinorrhea, nasal congestion, undifferentiated upper airways inflammatory syndrome, and dyspnea. Additionally, various other symptoms such as gastrointestinal issues (nausea, vomiting, abdominal pain, and diarrhea) and anosmia have been documented. In general, children affected by SARS-CoV-2 typically display mild symptoms, but a notable proportion (15%-35%) may remain asymptomatic.21,22 Among children aged ≤9 years, prevailing symptoms comprise fever (46%), cough (37%), headache (15%), diarrhea (14%), and sore throat (13%) while in children aged 10 to 19 years, the most frequent symptoms are headache (42%), cough (41%), fever (35%), myalgia (30%), sore throat (29%), shortness of breath (16%), and diarrhea (14%). 23
Distinguishing between viral and bacterial illnesses clinically can be challenging. Therefore, laboratory testing to confirm the type of infectious agent is required to enable the prompt initiation of antibiotic treatment for bacterial pneumonia while restricting the use of antibiotics in viral pneumonia. 24 The gold standard for determining the cause of ARI and the detection of co-infections is multiplex RT-PCR,25,26 as the separate detection of these pathogens is time-consuming and labor-intensive. 27 Antimicrobial resistance (AMR), intestinal dysbiosis, and antibiotic-associated diarrhea are believed to result from the widespread and inappropriate use of antibiotics and have significant negative impacts on public health and the worldwide economy.28,29 As an institution dedicated to infection prevention and control in Thailand, Bamrasnaradura Infectious Diseases Institute(BIDI) monitors the rate of co-infections and risk factors in SARS-CoV-2 and other ARI during pandemics and analyzes the effects of these factors on practice. Therefore, we aimed to compare the prevalence of co-infection, types of respiratory pathogens involved in co-infection, and risk factors in children hospitalized with SARS-CoV-2 ARI and non-SARS-CoV-2 ARI.
Methods
Study Setting and Design
This observational cross-sectional study compared the clinical characteristics, and management of SARS-CoV-2 and non-SARS-CoV-2 ARIs among in-patients at the BIDI, Ministry of Public Health, Nonthaburi, from January 2020 to December 2022. We also compared the prevalence of co-infections and types of pathogens in the residual nasopharyngeal samples.
Study Population
Approximately 1000 children are hospitalized annually at our institution, and 40% of all cases involve ARI. The study population included hospitalized children<15 years of age diagnosed with ARI with a duration of respiratory symptoms<14 days and an available residual nasopharyngeal specimen that was routinely collected for the detection of respiratory disease. Residual nasopharyngeal specimens were used to detect respiratory pathogens by multiplex polymerase chain reaction (PCR; QIAstat-Dx Respiratory SARS-CoV-2 Panel). Children previously enrolled in the same ARI episode, AOM, acute sinusitis, or with evidence of bacterial infection were excluded from the study. Data collected from the electronic medical records included demographic data, birth history, past medical history, family history, clinical manifestations, physical examination on admission, clinical diagnosis (upper respiratory tract infection or lower respiratory tract infection), clinical diagnosis (pneumonia or non-pneumonia), laboratory data, duration of illness before hospitalization, and empirical antibiotic use. Patients were categorized into three age groups: 0 to 5 years, 5 to 9 years, and 10 to 15 years. Coinfection was defined as the concurrent infection of a cell or organism with two or more pathogenic agents.30,31
Specimen Collection and Transportation Along With Detection of Co-infection by QIAstat-Dx Respiratory SARS-CoV-2 Panel
The QIAstat-Dx Respiratory SARS-CoV-2 Panel is a multiplexed, nucleic acid real-time PCR test intended for the qualitative detection, and differentiation of nucleic acids from multiple respiratory viral and bacterial pathogens, including SARS-CoV-2, in nasopharyngeal swabs eluted in universal transport media collected from patients suspected of SARS-2 by their healthcare providers. The QIAstat-Dx Respiratory SARS-CoV-2 Panel is intended for the detection and differentiation of nucleic acids from SARS-CoV-2 and subsequent pathogen types and subtypes. Amongst viruses, these are Adenovirus, Bocavirus, Coronavirus 229E, Coronavirus HKU1, Coronavirus NL63, Coronavirus OC43, SARS-CoV-2, Human Metapneumovirus A + B, Influenza A, Influenza A H1, Influenza A H3, Influenza A H1N1/pdm09, Influenza B, Parainfluenza virus 1, Parainfluenza virus 2, Parainfluenza virus 3, Parainfluenza virus 4, Rhinovirus/Enterovirus, Respiratory Syncytial Virus A + B, and amongst bacterium, these are Bordetella pertussis, Chlamydophila pneumoniae, and Mycoplasma pneumoniae. 32
Statistical Analysis
Data were analyzed descriptively. Continuous data are presented as mean ± SD as appropriate. Categorical data were presented as frequency (%), and continuous data were compared using the independent t-test or Mann-Whitney U test, as appropriate for distribution. Categorical data were compared using the independent chi-squared test or Fisher’s exact test, as appropriate.
The sample size was calculated using a proportional formula assuming a prevalence of co-infection of 19.8% among SARS-CoV-2 infected children, 24 the calculated minimum sample size for both groups was 61. A simple random sampling method was used to select the study participants.
Results
Study Population
As shown in Figure 1, we screened 235 cases of ARI in hospitalized children under the age of 15 years, covering the period from January 2020 to December 2022. Out of these cases, 28 exhibited respiratory symptoms for a duration ≥14 days, and 9 cases had unavailable residual nasopharyngeal specimens. Following the initial screening, we identified 198 patients who progressed to the subsequent analysis phase. From this pool, we excluded 14 cases for the following reasons: 10 cases were previously enrolled for the same ARI episode, one case had acute otitis media, one case presented with acute sinusitis, and two cases showed evidence of bacterial infection. In total, 184 individuals were included in the final analysis. Among them, 122 (63.3%) were diagnosed with SARS-CoV-2 infection, and 62 (33.7%) were not have SARS-CoV-2 infection. All 184 residual nasopharyngeal specimens were used to detect respiratory pathogens using multiplex PCR. None of the hospitalized children died.

Flowchart of SARS-CoV-2 infection with ARI and non-SARS-CoV-2 infection with ARI among hospitalized children.
As shown in Table 1. Multiplex polymerase chain reaction (PCR) was used to analyze 184 nasopharyngeal specimens for the presence of various pathogens. Among the 167 pathogens detected, the distribution was as follows: SARS-CoV-2 (n = 122, 73.05%), Rhinovirus/Enterovirus (n = 19, 11.38%), Bocavirus (n = 8, 4.79%), RSV (n = 6, 3.59%), Parainfluenza-3 (n = 4, 2.39%), Adenovirus (n = 2, 1.20%), Influenza A-H3 (n = 2, 1.20%), Coronavirus 229E (n = 1, 0.60%), Coronavirus OC43 (n = 1, 0.60%), HMPV A/B (n = 1, 0.60%), and Mycoplasma pneumoniae (n = 1, 0.60%). Twelve hospitalized children with ARI experienced co-infections, comprising 3 cases involving SARS-CoV-2 and 9 cases without SARS-CoV-2 involvement. In the single pathogen category, there were 119 cases with SARS-CoV-2 and 22 cases without SARS-CoV-2. Notably, 31 patients in the non-SARS-CoV-2 group had no identifiable pathogens.
Prevalence of Respiratory Pathogens in 184 Hospitalized Children with ARI.

Prevalence of Co-infection and a Comparative Analysis Between SARS-CoV-2 ARI and Non-sARS-CoV-2 ARI Among Hospitalized Children
As shown in Figure 2, SARS-CoV-2 ARI had a significantly lower rate of co-infection than non-SARS-CoV-2 ARI (3/122 (2.5%) vs 9/62 (14.5%), P-value = .003). The proportions of SARS-CoV-2+Rhinovirus/Enterovirus, and SARS-CoV-2+ Coronavirus 229 were 1.64% (2/122), and 0.82% (1/122), respectively. Among the 62 non-SARS-CoV-2 ARI children with multiple pathogen infections, the most common combination of pathogens was Bocavirus+ Rhinovirus/Enterovirus (n = 3), followed by Bocavirus + Parainfluenza virus 3 (n = 2), Rhinovirus/Enterovirus + Human Metapneumovirus A + B (n = 1), Rhinovirus/Enterovirus + Parainfluenza virus 3 (n = 1), and Influenza A H3+ Mycoplasma pneumoniae (n = 1). The most common combination for four infectious was Bocavirus+ Parainfluenza virus 3 + Rhinovirus/Enterovirus+ Respiratory Syncytial Virus A + B (n = 1).
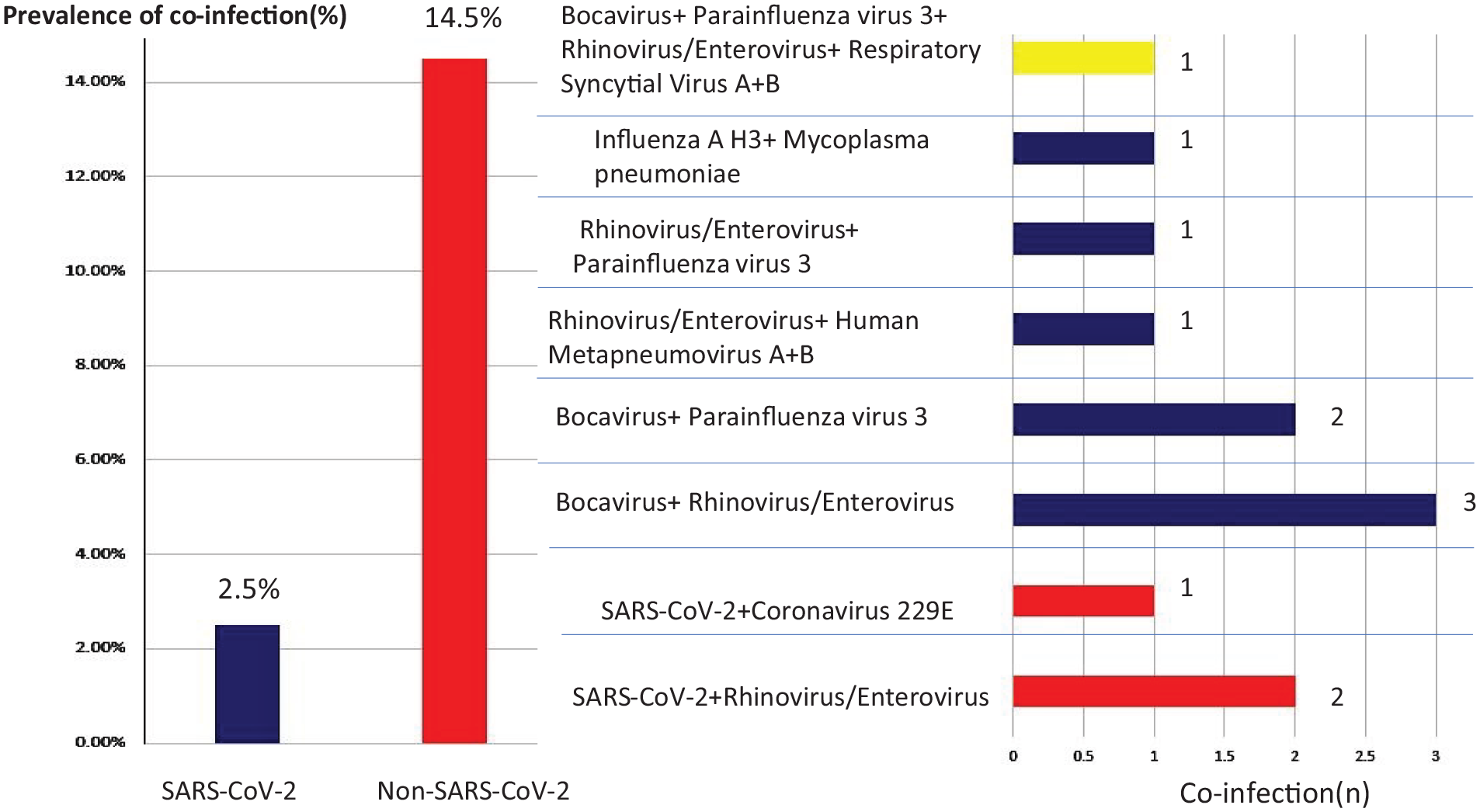
Prevalence of co-infection and comparative analysis between SARS-CoV-2 ARI and non-SARS-CoV-2 ARI among hospitalized children.
Comparison of Characteristics, Data, Symptoms, Signs, and Laboratory Tests Between SARS-CoV-2 ARI and Non-sARS-CoV-2 ARI Among Hospitalized Children
As shown in Table 2, the mean age ± SD in SARS-CoV-2 ARI children was 3.99 ± 3.91 years while it was 4.00 ± 3.82 years in non-SARS-CoV-2 ARI children. SARS-CoV-2 ARI children had significantly more pneumonia diagnose, more abnormal chest X-ray findings, and less empirical antibiotic use than non-SARS-CoV-2 ARI children (all P < .05). Age, age category, weight, sex, underlying medical condition, proportion of diagnosis (upper respiratory tract infection and lower respiratory tract infection), fever, cough, rhinorrhea, sore throat, sputum, diarrhea, vomiting, loss of appetite, loss of taste, smell, seizures, peak body temperature on the first day, WBC count, hemoglobin level, platelet count, duration of illness before hospitalization, oxygen support, and PICU admission were not significantly different between the groups(P > .05).
Comparison of Characteristics, Data, Symptoms, Signs, and Laboratory Tests Between SARS-CoV-2 ARI and Non-SARS-CoV-2 ARI Among Hospitalized Children.

P value <.05 significant.
Table 3 shows the exploratory multivariate logistic regression model of SARS-CoV-2 ARI children. In the exploratory multivariate analysis, independent predictors of SARS-CoV-2 infected children with ARI were less empirical antibiotics use (aOR = 0.09, 95% CI = 0.03-0.21; P-value = .000), more pneumonia diagnosis (aOR = 5.15, 95% CI = 1.77-14.95; P-value = .003), and more abnormal chest X-ray findings (aOR = 2.81, 95% CI = 1.38-5.71; P-value = .004).
Multivariate Logistic Regression Model of SARS-CoV-2 Infected Children with ARI.

P value <.05 significant.
Discussion
In the SARS-CoV-2 pandemic era, we found a significantly lower rate of co-infection in SARS-CoV-2 infected children with ARI than in those with non-SARS-CoV 2 (2.5% vs 14.5%, respectively). Similarly, Wang et al in a study from Wuhan, China, found the amongst 613 patients tested for the presence of 13 respiratory pathogens, only 5.8% of SARS-CoV 2 infected patients, and 18.4% of non-SARS-CoV 2 had co-infection. 33 In Singapore, Wee et al demonstrated a co-infection rate between SARS-CoV-2, and other respiratory viruses was low at 1.4%. 34 Conversely, in the United States, Kim et al showed that amongst 1217 patients, 19.8% of SARS-CoV-2 infected patients and 26.5% of non- SARS-CoV-2 patients had co-infection, and there was no significant difference in co-infection among these patients. 35 In the present study, we have demonstrated that co-infection with other respiratory pathogens was less common among the SARS-CoV-2 children at one center in Thailand. The possible reason could have been competitive advantage of SARS-CoV-2 in modulating the host’s immunity. It has been hypothesized that competitive advantage may play a role in the SARS-CoV-2 interaction with other respiratory viruses during co-infection, and that this may be one reason why the co-infection rate in SARS-CoV-2 patients is much lower. 36 However, the prevalence of each respiratory pathogen may have varied between studies due to population, seasonality, geographic areas, and detection techniques. 14 Furthermore, the co-infection rate during the SARS-CoV-2 pandemic is affected by clinical, demographic, outcome patterns and country-specific policy. For example, a previous study conducted in Italy has revealed that 57.25% of co-infections occurred in children hospitalized with lower respiratory tract infections who tested positive for viral infections during the SARS-CoV-2 pandemic, and the severity of these cases was increased. 8
The present study demonstrated that the majority of respiratory pathogens were Rhinovirus/Enterovirus among non-SARS-CoV-2 infected children. A similar result was found by Danielle et al in the United State that demonstrated Rhinovirus/Enterovirus persisted, and was the most common respiratory pathogen. Rhinovirus/enterovirus remains a leading factor in the ARI health care burden, and active ARI surveillance in children, and adolescents remains critical for defining the health care burden of respiratory viruses. 37 The simultaneous detection of multiple pathogens, especially viruses, does not necessarily implicate a pathogenic effect at the time of detection, especially when molecular methods are used. It is also difficult to distinguish between active viral infection, viral shedding, and potentially non-pathogenic viral infections. For instance, human bocaviruses and human adenoviruses may exhibit prolonged viral shedding. 11
In the current study, antibiotic use for children with SARS-CoV-2 ARI was lower. Numerous previous studies have suggested that empirical antibacterial therapy is unnecessary in children with ARIs for several reasons. Firstly, children with fever who have tested positive for viral infection should be initially diagnosed with viral infections until proven otherwise. Antibiotics should only be administered if bacterial infection is strongly suspected and is supported by further diagnostic evaluations. 20 Secondly, bacterial co-infection in patients with SARS-CoV-2 ARI is relatively uncommon. For instance, a study conducted in Spain during the early months of the pandemic has found that less than 10% of SARS-CoV-2 patients had bacterial co-infection. 38 Additionally, a prospective observational study conducted across multiple European centers, representative of the target population for NPS testing, has reported that 10% to 15% of children presenting with fever are diagnosed with a bacterial infection. 39 Lastly, according to our institution’s treatment guidelines for SARS-CoV-2 and other viral infections during the pandemic, empirical antibiotic therapy is not recommended for mild to moderate cases. Furthermore, upon admission for SARS-CoV-2, isolation is strongly advised, particularly for children.
The present study demonstrated that SARS-CoV-2 ARI children were associated with pneumonia and a high proportion of abnormal chest X-ray findings. Similarly, a previous study demonstrated a high prevalence of pneumonia diagnosis in SARS-CoV-2 infection at 62.5%, which was higher than other pathogens, such as the prevalence of pneumonia diagnosis as H1N1 influenza, which was low at 11%. 40 In addition, Oterino et al and Palabiyik et al in children demonstrated that pulmonary abnormalities on chest X-ray were found in 46% to 90% of cases.41,42 However, the radiologic abnormalities of SARS-CoV-2 infection among children are non-specific, and chest X-rays alone are insufficient for confirmed diagnosis. 43 It is interesting to note that whereas SARS-CoV-2 ARI children were linked to greater pneumonia diagnoses than non-SARS-CoV-2 group, the severity of pneumonia in both groups was the same. However, the cases of adult SARS-CoV-2 pneumonia, it is different. Preschoolers’ immune cell repertoire is 5 to 10 times bigger than that of a 50-year-old and 20 times larger than that of an 80-year-old, owing to the immune response in children being different from that of adults, which gradually deteriorates with age. The extent to which this may help prevent the virus from spreading and the cytokine signaling cascade initiated by SARS-CoV-2 in relation to serious adult outcomes remains unknown. 44
Our study has limitation of address. Firstly, the study had a small sample size with, variation in the number of included children because of the laboratory-based cross-sectional retrospective survey nature of the study. Secondly, our questionnaire was pilot-tested, which may be misleading in small sample size. However, pilot testing enables us to assess the entire questionnaire under survey conditions, identify problems, and examine the validity of each question. Lastly, our PCR panel did not include all frequently encountered respiratory viruses, such as other endemic Coronaviridae.
Conclusions
Empirical antimicrobial therapy is crucial for severe infections, especially nosocomial cases caused by Gram-negative bacteria. which are often responsible for critical conditions. Timely initiation of appropriate antibiotics improves survival rates. However, excessive use of empirical therapy, particularly in cases of viral infections and non-severe conditions, undoubtedly contributes to the development of bacterial resistance to antibiotics in pediatric patients worldwide.
We suggest that empirical antimicrobial therapy is not required for the routine management of children hospitalized with mild SARS-CoV-2 ARI, even though pneumonia and abnormal chest radiography findings were identified in cases of SARS-CoV-2 ARI.
Supplemental Material
sj-docx-1-gph-10.1177_2333794X241275267 – Supplemental material for Prevalence of Co-Infections and Pathogens in Hospitalized Children with Acute Respiratory Infections: A Comparative Analysis Between SARS-CoV-2 and Non-SARS-CoV-2 Cases
Supplemental material, sj-docx-1-gph-10.1177_2333794X241275267 for Prevalence of Co-Infections and Pathogens in Hospitalized Children with Acute Respiratory Infections: A Comparative Analysis Between SARS-CoV-2 and Non-SARS-CoV-2 Cases by Visal Moolasart, Ravee Nitiyanontakij, Srisuda Samadchai, Somkid Srisopha, Priyanut Atiburanakul, Suthat Chottanapund and Sumonmal Uttayamakul in Global Pediatric Health
Footnotes
Acknowledgements
We thank all the study participants and staff of Bamrasnaradura Infectious Diseases Institute, Department of Disease Control, Ministry of Public Health, Nonthaburi, Thailand.
Author Contributions
VM: Contributed to conception, design, acquisition, analysis, interpretation, Drafted the manuscript, Critically revised the manuscript, Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy. RN: Contributed to conception, design, acquisition, analysis, interpretation, Drafted the manuscript, Critically revised the manuscript, Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy. SS (Srisuda Samadchai): Contributed to conception, acquisition, analysis, Drafted the manuscript, Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy. SS (Somkid Srisopha): Contributed to conception, design, Critically revised the manuscript, Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy. PA: Contributed to conception, design, Critically revised the manuscript, Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy. SC: Contributed to conception, design, Critically revised the manuscript, Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy. SU: Contributed to conception, design, acquisition, analysis, interpretation, Drafted the manuscript, Critically revised the manuscript, Gave final approval, Agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was conducted by the Bamrasnaradura Infectious Diseases Institute. The PCR respiratory pathogen panel was provided by the Japan International Cooperation Agency for the Capacity Building of Infectious Disease Institute in responding to COVID-19.
Ethics Approval and Informed Consent
Ethical clearance was obtained from the ethical review and research committee of the Bamrasnaradura Infectious Diseases Institute, situated at the Ministry of Public Health in Nonthaburi, Thailand (Postal Code: 11000), After a thorough assessment of the study for its scientific contribution and ethical issues, approval was granted with the reference code SO12h/65. The requirement for informed consent was waived due to the retrospective nature of the study, which relied on secondary data collected from patients’ medical charts and residual nasopharyngeal samples. Furthermore, all methods were performed in accordance with the ethical standards outlined in the Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent for Publication
Not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
