Abstract
Introduction
Cerebral palsy (CP) is a non-progressive neurological disease during brain development. CP is caused by damage to the central nervous system (CNS). Although CP lesions are non-progressive, the child with CP has a secondary set of physical and mental disorders.1-3 Because brain development occurs during the first 2 years of life, the prevalence of CP is higher during infancy.2,3 CP often occurs congenitally and without any specific cause. 3 In addition to motor deficits, speech problems, seizures, and digestive problems are seen in children with CP.4-6 One of the most common gastrointestinal symptoms in children with neurological disorders is constipation, so the prevalence of constipation in children with CP has been reported more than healthy children.7-9 Constipation occurs with stool retention, hard stools, decreased stool frequency, and difficult bowel movements. 10 If the number of defecation is less than 3 times a week and a hard and large stool is felt during the abdominal examination, the child has constipation. 11 Constipation affects a person’s quality of life, 10 so it is managed through lifestyle changes, education to the family or child, and treatment with fecal disimpactions. 12 Lactulose, mineral oils, magnesium milk, sorbitol and polyethylene glycol (PEG) are among the fecal disimpactions.13,14 PEG is non-toxic and water soluble agent and is not absorbed in the gastrointestinal tract and increases stool water.15,16 Studies have reported that low-dose PEG is effective in treating constipation in children.15,17 domperidone is also a drug that increases motility and regulates gastrointestinal movements. 18 It also has some benefitson gastrointestinal symptoms including constipation. 19
Due to the prevalence of constipation in patients with CP, the refracting of constipation to treatment, and the lack of sufficient studies in this field, this study was performed to compare the effect of 2 treatment regimens of PEG with domperidone and PEG with placebo on CP patients with constipation.
Methodology
This study is a double-blind randomized clinical trial. Participants include CP patients with constipation who were referred to the pediatric gastroenterology service of Mohammad Kermanshahi hospital of Kermanshah city during 2019.
The sample size was calculated to detect a difference in the response rates to the treatment between the groups, assuming a response rate to the treatment of 0.79% for the A group and 0.4% for the B group based on the results of previous study. 20 therefore, with confidence level of 95% and a power of 80%, the minimum sample size required was 42 subjects, which was increased to 50 to allow for dropouts.
Inclusion criteria were CP children with untreated chronic constipation, reduced gastrointestinal motility, and without cardiovascular defects (based on ECG and approval by a pediatric cardiologist). Participants were randomly divided in a ratio of 1:1. In order to create an allocation sequence, simple random allocation was applied using an Excel file. Forty-two eligible patients were included in the study (21 in each group). The research assistant assigned participants to groups using the generated allocation sequence. To conceal the sequence of allocation, the study arm for each patient was placed in a sealed envelope numbered 1 to 42.
Exclusion criteria include patients who were not able to take the drug properly for any reason, patient (parent) refusal to participate in the study, and the existence of an organic cause for constipation (based on their history and/or physical examination) such as no retention of feces in the rectum, hard anal sphincter, history of delayed excretion of neonatal meconium, explosive excretion of feces after rectal examination, and presence of urinary/bladder problems and extraintestinal defects (Figure 1).
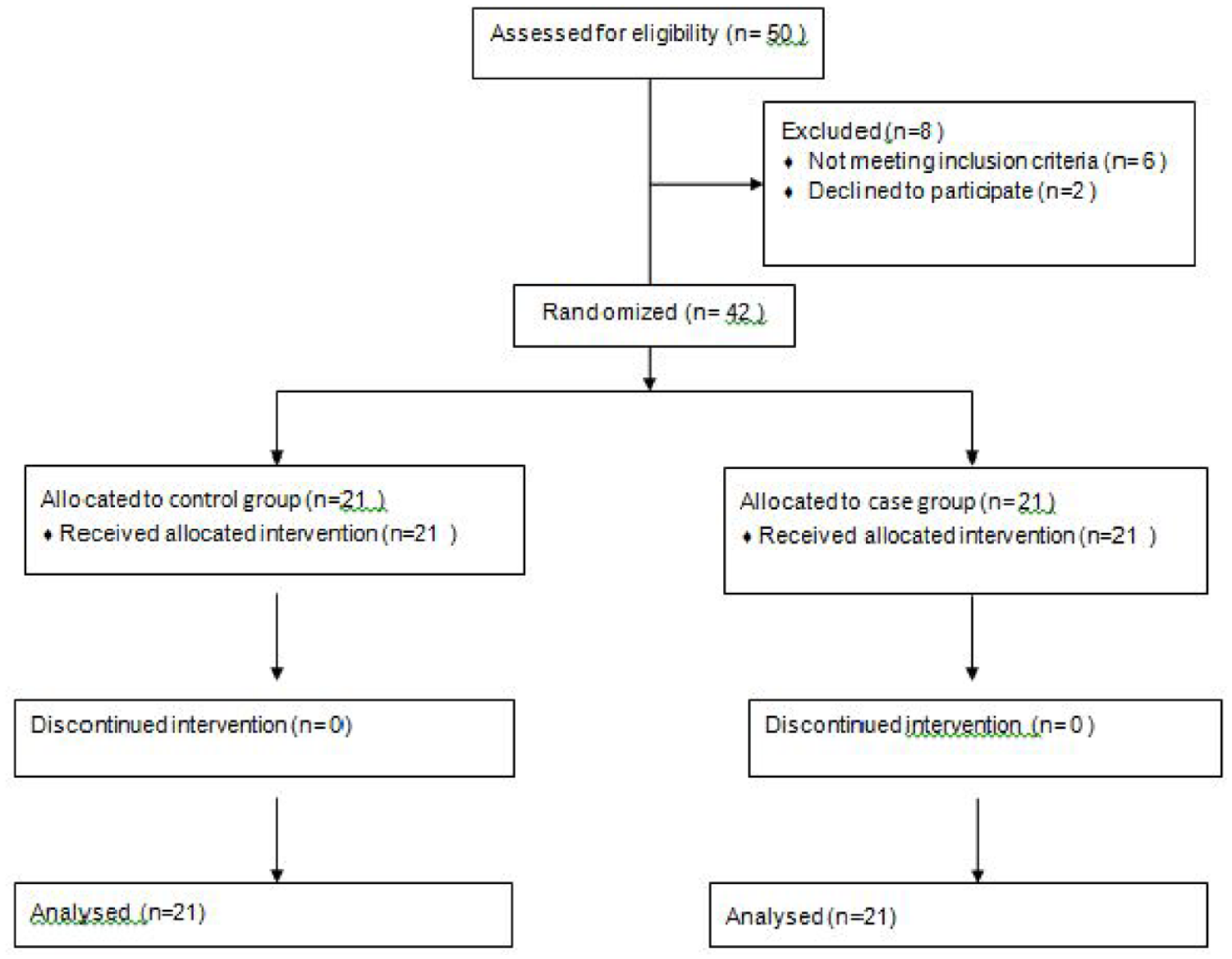
Consort diagram of comparison of the effect of two therapeutic interventions for the treatment of chronic constipation in children with cerebral palsy.
Patients with chronic constipation were selected based on the Rome IV criteria for a diagnosis of Functional Constipation mentioned in Table 1. 21 The patients were divided into 2 groups case and control.
Rome IV Criteria for Constipation.

The patients were placed in 2 different treatment groups in such a way that closed treatment packages were prepared in the form of treatment group number A or B, one of which was given to the patient by chance.
Each of the treatments including PEG with domperidone and PEG with placebo (Distilled water), sufficient and equal amount of drug was considered according to the patient’s weight. Patients were treated with Polyethylene glycol with domperidone (0.4 gr/kg/dose 3 times a day + domperidone syrup in the amount of 0.2 mg/kg/dose 3 times a day) and Polyethylene glycol with placebo (0.4 gr/kg/dose 3 times a day + placebo (containing distilled water) in the amount of 0.2 mg/kg/dose 3 times a day).
The patients were explained how to take the medicine and after prescribing the medicine, the patients were asked to see a doctor again after a 2-week treatment period to check the results of the treatment. The treatment response criterion was explained to the parents and they were asked to record the cases. The treating physician reviewed the response to treatment without knowing the treatment group.
This study was registered in the Iranian Registry of Clinical Trials (IRCT registration code: IRCT20130812014333N126). Moreover, written informed consent forms were obtained from the neonates’ parents.
The protocol approval by the Ethics Committee of Kermanshah University of Medical Sciences (IR.KUMS.REC.1398.067).
Statistical Analysis
SPSS software version 22 was used to enter the data and analyze them. First, Kolmogorov–Smirnov test was used to evaluate the normality of quantitative variables and then independent
Results
Forty-two children aged 2 to 16 years with CP and constipation who met the inclusion criteria were included in the study. Twenty-one patients were treated with PEG with domperidone (case group) and 21 patients were treated with PEG + placebo (control group).
The mean age of the case and control groups was 53.52 ± 30.71 and 72.52 ± 38.2 months, respectively. The mean weight of patients in case and control groups was 12.52 ± 3.32 and 14.19 ± 6.72 kg, respectively. There was no significant difference between the patients’ age, sex, and weight in the two groups (
Characteristics of the Patients in Case (Polyethylene Glycol + Domperidone) and Control (Polyethylene Glycol + Placebo) Groups.

There was no significant difference between clinical symptoms and history of drug use in the two groups (
Comparison of Status of Clinical Symptoms and History of Drug Use in Case (Polyethylene Glycol + Domperidone) and Control (Polyethylene Glycol + Placebo) Groups Before and After Treatment.

There was no significant difference (
Comparison of Rome IV Criteria in Case (Polyethylene Glycol + Domperidone) and Control (Polyethylene Glycol + Placebo) Groups Before and After Treatment.

Based on the results of comparisons of Rome IV criteria, in both groups, there was a significant difference (
However, in both groups, there was no significant difference between fecal incontinence greater than or equal to once a week before and after treatment (
The success of the treatment response to PEG with domperidone was significantly higher than the PEG + placebo in children with CP and chronic constipation (

Frequency of response to treatment in case(polyethylene glycol with domperidone) and control (polyethylene glycol + placebo) groups.
Discussion
CP is a group of non-progressive chronic movement and muscle tone disorders that result from injury to the central nervous system (CNS) during its development. 22
Chronic constipation is one of common complaint in children with CP that its prevalence varies from 26% to 74%. 22 These need to long-term treatment with laxative agents but gold standard treatment yet has not been found for them.
Children with chronic constipation need long-term laxative treatment, so the laxative used should be safe and without side effects. 23 polyethylene glycol (PEG) is a non-poisonous polymer, soluble in water, with high molecular weight, and which does not absorb after ingestion. PEG acts as an osmotic agent an increases stool water content end is safe for chronic use.15,16
This study included 42 children with Cerebral Palsy (CP) and chronic constipation, who were randomly divided into 2 treatment groups.
In the present study all of 21 patients in the PEG group had painful defecation of large and hard stools, and defecation frequency fewer than 2 times per week. after 2 weeks of treatment with PEG, 13 patients (61.9%) had defecation frequency of more than 2 times a week, 18 patients (85.7%) had decreased size of stool diameter, and 16 patients (76.1%) had painless and soft defecation.
Ten patients had blood in the stool and 7 patients (70%) had appropriate response to treatment. Stool incontinence was seen in 38.1% of patients before treatment and after the treatment period, about 33.3% still had this compliant. In the Imanieh et al study, as in our study, after treatment with PEG, the bowel the frequency was more than twice week in 58.8% of patients, and in 80% of them did not observe any blood on stool. However, stool incontinence was seen in 17.6% of patients, and after treatment about 5.8% still had this complaint. 24 The results of the study by Thomson et al showed that the number of weekly stools in children who used PEG was significantly increased compared to placebo. 25
Studies have shown that domperidone is a peripheral dopamine antagonist which increases smooth muscle ton motility of stomach and intestine. It has been also shown that has no effect in the treatment of constipation.26-29 Imanieh study was reported that domperidone had the least effect on constipation end only 6.6% of patients with hard and painful stool improved with domperidone. It had not any effect on the stool incontinence and blood on stool. 24 Dehghani et al also showed that domperidone had no effect in treating chronic constipation. 30 but, the results of our study and other studies showed that domperidone had a synergistic effect on PEG.24,30
In our study, of 21 patients treated with PEG with domperidone, 90.4% had bowel movement more than twice a week after treatment, the stool diameter decreased in 90% patients, and the stool became soft and painless in 90% of patients. Nine patients had blood in the stool and 8 patients (88.8%) responded to treatment. Three patients (14.3%) had fecal incontinence that one (4.8%) of them still had this complaint. Therefore, treatment response to PEG plus domperidone was significantly higher than PEG plus placebo in children with CP and chronic constipation. Imanieh et al showed that PEG with domperidone has the best therapeutic effect in the treatment of chronic constipation in children with CP. However, domperidone alone had no effect and PEG alone had a moderate effect. 24 Dehghani et al reported that in the treatment of pediatric chronic constipation, PEG regimen with domperidone and PEG with placebo are effective, but the combination containing PEG and domperidone was not superior to treatment with PEG alone. 30 In our study and the Imanieh study, children with CP and constipation were studied for 2 weeks, while in the Dehghani study, children with only chronic constipation (no CP) followed up for a longer period. In addition, due to the limited number of studies evaluating the effect of PEG with domperidone on chronic constipation in children, no further comparison was possible.
In the present study, the response rate to treatment in the PEG with domperidone group was 90.5% and, in the group, treated with PEG + placebo was 61.9%. In the study of Imanieh et al, the response rate to treatment with PEG alone was 58.8%, in the group treated with PEG + domperidone was 94.4% and in the group treated with domperidone alone was 6.6%. 23 In a systematic review study by Candy and Belsey, the response rate PEG treatment was 56% to 84%. 31
Domperidone improves the function of the upper gastrointestinal tract and PEG has osmotic properties. Therefore, combining the two together can cause painless defecation.
None of the patients complained of side effects during the treatment period. In a study by Llerena et al, PEG had higher safety than other fecal dis impactions such as lactulose or lactitol. 32 Chen et al showed that osmotic laxatives are effective and safe in treating patients with chronic constipation. 33
Our study has some limitations including the low sample size, and the presence of few similar studies in the literature to be compared to our results.
Conclusion
PEG with domperidone had a positive treatment effect on children with CP and chronic constipation. Although further studies are needed, this combination therapy due to its effect on one of the most significant problems in children with cerebral palsy and due to non-expensively of it can be used in all of the world.
Footnotes
Acknowledgements
GHY, MN, KGH involved in study design, writing the manuscript; RCL, SKH involved in the collection of samples and preparation of data, drafting and revising of the work; JH involved in the data analysis; MR, MS drafting and revising of the work; All authors read and approved the fnal manuscript.
Authors’ Note
Sepideh Khodamoradi is now affiliated to Department of Microbiology, shahr-e-Qods Branch, Islamic Azad University, Tehran, Iran.
Author Contributions
KGH, GYF designed the study. SKH, and RCL recorded the data of the patients. MR analyzed and interpreted the data. KGH, MS, HD, MN, JH have drafted the work. All authors read and approved the final manuscript
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The protocol approval by the Ethics Committee of Kermanshah University of Medical Sciences (IR.KUMS.REC.1398.067).
Trial Registration
This study was registered in the Iranian Registry of Clinical Trials (IRCT registration code: IRCT20130812014333N126).
