Abstract
Objective. To describe heated humidified high-flow nasal cannulas (HHHFNC) utilization in level III neonatal intensive care units (NICUs) in Saudi Arabia. Methods. A prospective cross-sectional study using an electronic web-based questionnaire. The survey targeted level III NICUs hospitals using HHHFNCs, covering HHHFNC availability, protocols, patient characteristics, and indications. It also collected opinions on the benefits of HHHFNCs compared to nasal continuous positive airway pressure (nCPAP). Results. Out of 47 government-level III neonatal intensive care units, 35 (74%) responded to the survey. Among the included units, 46% had guidelines for HHHFNC use. Additionally, 51% reported using HHHFNC in infants of all gestational ages. The primary indication for HHHFNC use was weaning off nCPAP (34%), with 60% of the respondents noting its advantages for kangaroo care and breastfeeding. Conclusion. HHHFNC are increasingly prevalent in NICUs in Saudi Arabia. However, there remain no clear policies or guidelines regarding their use in preterm infants.
Keywords
Introduction
Preterm infants, born with immature lungs, often experience difficulty breathing independently 1 and frequently require respiratory support. 2 Various modalities for respiratory support with minimal damage have been developed over the decades, 3 varying based on the infant’s gestational age, overall health, and respiratory status. 4 One example is the heated humidified high-flow nasal cannula (HHHFNC), which administers humidified and heated oxygen at a high flow rate. 5
In the last decade, HHHFNCs have become an alternative method for non-invasive ventilation in neonatal settings,3,6 enhancing spontaneous breathing by meeting inspiratory demands, decreasing oxygen dilution, washing out dead space, and providing distending pressure. This results in decreased breathing effort, fixed oxygen delivery, and improved lung compliance. 7 However, the safety and efficacy of HHHFNCs in premature infants remain unclear.
Globally, there has been a recent increase in HHHFNCs utilization in neonatal intensive care units (NICUs), 8 showing efficacy similar to other non-invasive ventilation methods in preventing death and chronic lung disease. 9 Additionally, the use of HHHFNCs is associated with significantly decreased nasal trauma. 10 However, the inability to monitor the distending pressure created by increased flow poses a risk of overdistension and lung injury. 10
In 2012, a survey in the United Kingdom described the indications, patient characteristics, and perceptions of HHHFNC use in neonatal units, 11 revealing widespread use without clear policies, guidelines, or evidence supporting safety and efficacy. This indicates the need to evaluate and describe the use of HHHFNCs in neonatal units. Therefore, this study was aimed to describe HHHFNC use in level III neonatal units in Saudi Arabia and establish the baseline for HHHFNC use, as no national-level study has been conducted.
Material and Methods
Study Design and Ethics Approval
This prospective cross-sectional study evaluated the usage of HHHFNCs in NICUs through an electronic web-based questionnaire. The study was conducted in Saudi Arabia between September 1, 2022, and March 30, 2023. Ethical approval was obtained from the King Abdullah International Medical Research Center, Riyadh, Saudi Arabia (SP22R/072/05). Prior to the study, participants received a brief description of the study, its aim, and the identity of the principal investigator was provided. Participation was voluntary, and written informed consent was obtained from all the participants prior to study initiation. The participants were assured of data confidentiality and anonymity.
Participants
All level III NICUs in Saudi Arabian governmental hospitals utilizing HHHFNCs were eligible for this survey. Of the 47 eligible NICUs, 35 (74%) successfully completed it. Participants were contacted twice for follow-up questionnaire completion following the initial distribution. If no response was received after the second follow-up, it was deemed a lack of response for analysis purposes. This study is an exploratory analysis aimed at engaging all governmental hospitals. Consequently, no sample size calculation was conducted. However, the collection of responses from 74% of all governmental units was achieved.
Survey
The questionnaire was designed and administered using Google Forms (www.google.com, Mountain View, CA, USA) and distributed through WhatsApp to garner responses from heads of different NICUs. Notably, it has been previously validated and used in the United Kingdom. 11 A permission of using the questionnaire was obtained. The questionnaire covers demographic characteristics, including the region within Saudi Arabia, hospital type, bed capacity, and NICU level. Furthermore, it also comprises items relating to the availability and type of HHHFNCs, existing protocols, patient characteristics, and indications. Finally, it gathers perspectives from NICU heads regarding the advantages of HHHFNCs over nasal continuous positive airway pressure (CPAP).
Statistical Analysis
Descriptive analyses were performed, presenting all categorical variables as numbers and percentages. Statistical analyses were carried out using STATA version 17 software (StataCorp LP, TX, USA).
Results
Thirty-five NICUs, which accounted for 76% of government-level III NICUs in Saudi Arabia, participated in the study. Over half of the surveyed units (n = 19; 54%) were located in Riyadh with a median bed capacity was 40. Only 46% of the units had established policies or clinical guidelines for HHHFNC use (Table 1).
Characteristics of the Participating Units.
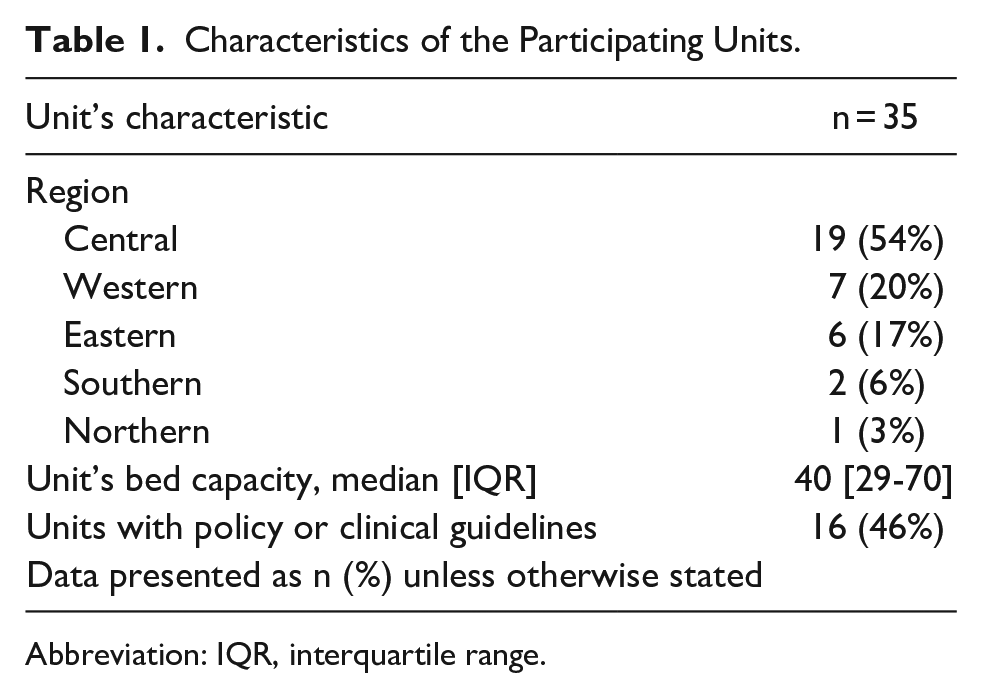
Abbreviation: IQR, interquartile range.
Characteristics of Eligible Infants and HHHFNC Setup
The gestational age and weight of infants connected to the HHHFNC are shown in Table 2 and Figure 1, respectively. Approximately 51% of the NICUs used HHHFNCs on infants of all gestational ages, with 32% using them on preterm infants born at ≥ 32 weeks. Regarding infant weight, 54% of the NICUs used HHHFNCs on children of any weight, while 31% on children weighing 1000 to 2000 g.
Description of HHHFNC Use in Units Based on Gestational Age.

Abbreviation: HHHFNC, heated humidified high-flow nasal cannula.
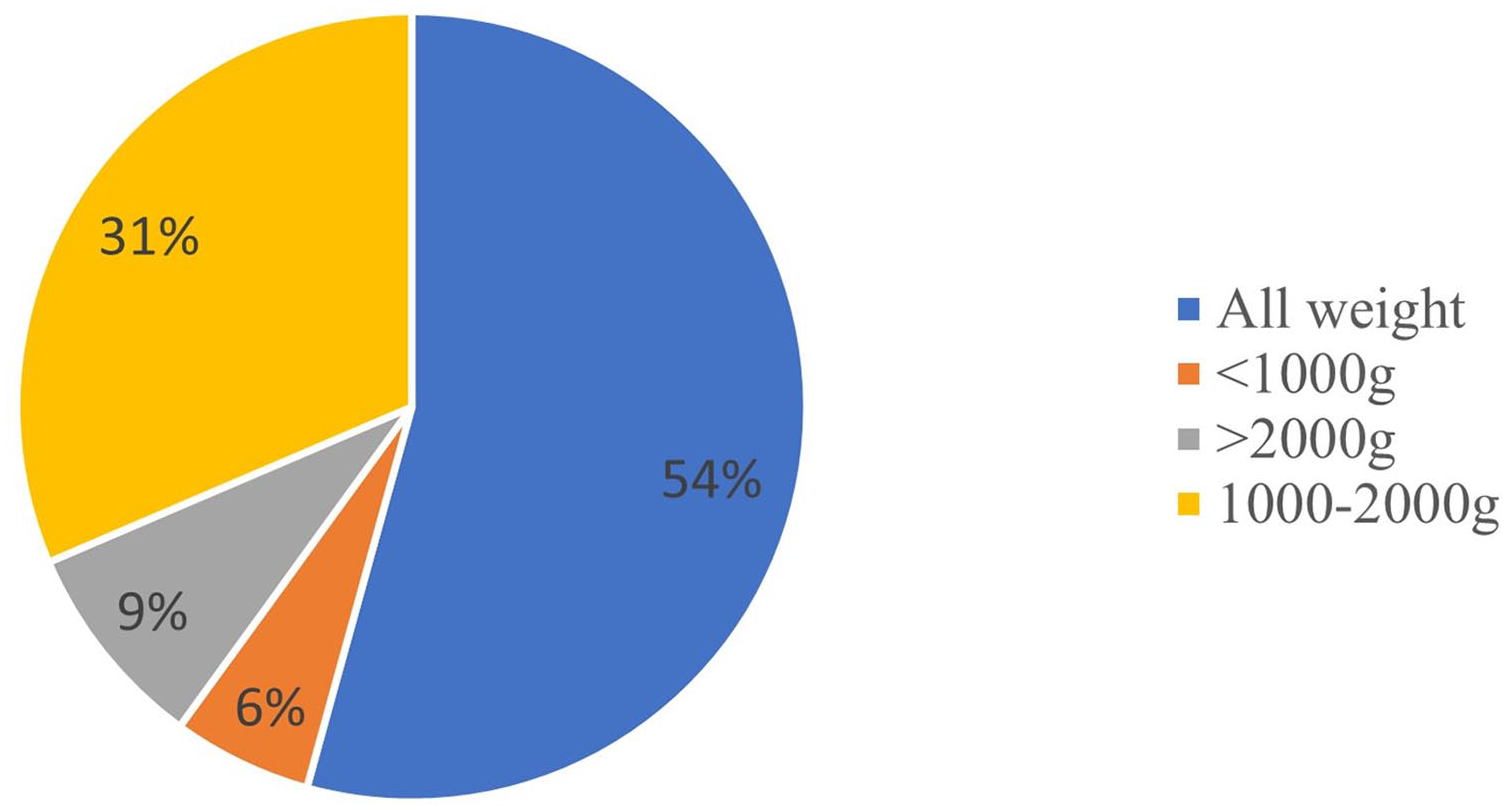
Data regarding the weight characteristics of infants connected to the heated humidified high-flow nasal cannulas (HHHFNC).
Table 3 provides information on HHHTNC setups. A majority of units (54.3%) used flow rates of 2 to 5 L/minute. Additionally, 49% of the units chose the cannula interface that offered the best fit, while 31% selected the fitting interface that allowed for leakage. Approximately 69% of the units did not employ active measures to keep the infants’ mouths closed. HHHFNC weaning was uniformly described as a reduction in the flow rate across all units (100%).
Data Regarding Setting Up and Using HHHFNCs.

Abbreviation: HHHFNCs, heated humidified high-flow nasal cannulas.
Indications and Advantages of Using HHHFNC
The most common indication for HHHFNC use was weaning off CPAP (34%), followed by managing apnea of prematurity (23%). Moreover, approximately 17% of units used HHHFNCs as an alternative to CPAP. A majority of units (60%) agreed that HHHFNCs held a unique advantage over CPAP by enabling kangaroo care and breastfeeding. Figure 2 illustrates the additional reported advantages of using HHHFNCs.

Advantages of using heated humidified high-flow nasal cannulas (HHHFNCs) over continuous positive airway pressure (CPAP).
Discussion
In our survey, over half of the units lacked policies or guidelines for HHHFNC use. Additionally, more than half of the NICUs used HHHFNCs at any gestational stage and/or weight. Approximately 60% of NICUs emphasized HHHFNC’s superiority over CPAP in facilitating kangaroo care and breastfeeding.
Despite the common use of HHHFNCs, only 46% of the surveyed units had policies or clinical guidelines, which are important for providing efficient patient care. 12 In the absence of evidence-based policies, units may rely on professionals’ knowledge, hindering patient safety and quality of care, especially among vulnerable preterm infants. 13
In our study, most units used HHHFNCs in preterm and term infants of any gestational age or weight. The national popularity adoption of HHHFNC therapy stems from its established safety and efficacy in neonatal care, corroborated by findings elucidated in prior Cochrane review. 6 This review have supporting the favorable outcomes associated with HHHFNC therapy in neonatal populations. 6 However, 20% of the units used HHHFNCs in extremely preterm infants (infants with a gestational age <32 weeks). This finding challenges the justification for not utilizing HHHFNCs in late preterm and full-term infants, despite evidence supporting their efficacy. 6 This could be attributed to the low percentage of units with established policies or clinical guidelines, as observed in our study.
In our study, more than half of the units used a flow rate of 2 to 5 L/minute based on infant weight and respiratory status, which is consistent with previous rates for preterm infants. 14 Furthermore, approximately half of the units chose the cannula that provided the best fit, potentially increasing distending pressure. 15 To address this concern, especially in preterm infants, it is recommended to use a cannula that allows leakage.16,17
Approximately one-third of the units used HHHFNCs to wean infants off CPAP, and only 17% used it as an alternative. This aligns with the general opinion that HHHFNC is inferior to CPAP due to the lack of conclusive evidence regarding the safety and efficacy of HHHFNCs. 6 However, two-thirds of NICUs considered HHHFNCs better than CPAP for kangaroo care and breastfeeding, given the challenges associated with securing the CPAP interface. 18
Our study has several strengths. First, the responses covered three-quarters of the population of interest, which increases the representativeness of our findings. In addition, NICUs’ heads completed the responses; therefore, the responses may have reflected each unit’s common practices rather than the individual practices of healthcare professionals.
However, our study has some limitations. First, this study was conducted at the national level, which limits the generalizability of our findings to other countries. Second, since the heads of NICUs completed the survey questionnaire, conformity bias was possible. Third, no power calculation for sample size was conducted.
Nevertheless, our findings demonstrate the importance of establishing and implementing evidence-based policies and clinical guidelines for using HHHFNC in NICUs. This study informs quality improvement projects for HHHFNC setup and practices.
Conclusion
HHHFNCs are widely used in NICUs in Saudi Arabia, specifically for preterm neonates of any gestational age or birth weight. However, a notable need exists for additional policies and guidelines to inform the proper use of HHHFNCs in these units. Therefore, establishing evidence-based policies and guidelines for using HHHFNCs in NICUs is urgent.
Supplemental Material
sj-pdf-1-gph-10.1177_2333794X241258142 – Supplemental material for Use of High-Flow Nasal Cannulas in Saudi’s Neonatal Level III Intensive Care Units: A Nationwide Questionnaire Study
Supplemental material, sj-pdf-1-gph-10.1177_2333794X241258142 for Use of High-Flow Nasal Cannulas in Saudi’s Neonatal Level III Intensive Care Units: A Nationwide Questionnaire Study by Saleh S. Algarni, Khalid Alshammari, Meshal Alkhalifah, Waleed Almutairi, Abdulrahman Aljaidi, Arwa Alruwaili, Abdulkarim S Alqarni, Tareq F Alotaibi, Mohammed M Alqahtani, Hassan Aljohani, Taha T Ismaeil, Abdullah M M Alanazi, Abdulrhman S Alghamdi, Khaled Alanazi, Khalid S Alwadeai, Rayan Siraj, Turki M Alanazi, Ali Almudeer, Kamal Ali and Saif Alsaif in Global Pediatric Health
Footnotes
Acknowledgements
A sincere gratitude to all the head of neonatal units participated in the study.
Author Contributions
Saleh S. Algarni: design, supervise, and approve the manuscript; Khalid Alshammari, Meshal Alkhalifah, Waleed Almutairi, Abdulrahman Aljaidi, Arwa Alruwaili, and Abdulkarim S. Alqarni: collecting the data; Tareq F. Alotaibi, Mohammed M. Alqahtani, Hassan Aljohani, Taha T. Ismaeil, Abdullah M. M. Alanazi, and Abdulrhman S. Alghamdi: writing the manuscript; Khaled Alanazi, Khalid S. Alwadeai, and Rayan Siraj: perform and review statistical analysis; Turki M. Alanazi: data management; Ali Almudeer, Kamal Ali, and Saif Alsaif: proof reading and revising the manuscript.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
The study was conducted in Saudi Arabia between September 1, 2022, and March 30, 2023. Ethical approval was obtained from the King Abdullah International Medical Research Center, Riyadh, Saudi Arabia (SP22R/072/05). The participants were assured of data confidentiality and anonymity.
Consent to Participate
Prior to the study, participants received a brief description of the study, its aim, and the identity of the principal investigator was provided. Participation was voluntary, and the responders provided consent prior to completing the questionnaire.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
