Abstract
Objective. This study evaluated the effects of serial clinical observation strategy complemented by point-of-care verification of blood culture volume in managing term and near-term newborns at risk for early-onset sepsis. Methods. We used a “before-and-after” approach. Infants born at ≥35 0/7 weeks’ gestation were eligible. Our strategy was based on serial clinical observation complemented with point-of-care verification of blood culture volume. Two separate 12-month periods were analyzed. The number of infants exposed to antibiotics started during the first 3 days of life was compared before and after introducing the strategy. Results. During the post-intervention period, 0.6% of infants received antibiotic therapy, compared to 4.1% during the pre-intervention period (P < .001; relative risk [RR]: 0.15; 95% CI: 0.08-0.28). Conclusion. Serial clinical observation complemented with verification of blood culture volume might reduce antibiotic utilization in newborns in the early postnatal period.
Keywords
Introduction
Evaluating newborns at risk of early-onset sepsis (EOS) is one of the most common responsibilities of neonatal care providers worldwide. Although decreasing incidence rates of EOS have been reported in recent years, 1 many newborns still receive antibiotics during the initial days of their lives.2,3 Concurrently, increasing evidence has indicated possible lifelong health hazards of early antimicrobial exposure.4-7 Most infants treated with antibiotics have negative blood culture and present no signs of illness. 8 However, clinical utility of the commonly used laboratory tests for diagnosing EOS is limited due to their low positive predictive values. 9 Practitioners are often reluctant to trust negative culture results because of concerns regarding false-negative results caused by an insufficient blood volume drawn for culture or by the effect of intrapartum exposure to antibiotics. Therefore, documenting the inoculated volume could be a solution; however, a visual assessment of the obtained volume could be unreliable.
A newborn’s clinical status is a subjective assessment that can deteriorate rapidly during the first hours of life. Conversely, perinatal risk factors and laboratory test results are objective assessments. Hesitant clinicians often prefer to treat at-risk infants with empirical antibiotics because of non-reassuring test results. In addition, the fear of missing a case of sepsis is escalated by the life-threatening nature of this curable illness in contrast to the unclear and postponed effects of antimicrobial misuse. Finally, the professional community has widely accepted the concept of culture-negative sepsis.11,12
Currently, infants at risk for EOS can be managed in the following 3 ways: (i) categorical risk assessment, which is the most traditional approach; (ii) multivariate risk assessment incorporated into a web-based neonatal EOS risk calculator; and (iii) serial clinical observation (SCO) or serial physical examination (SPE) strategies.1,10
The Polish perinatal care model enables widespread access to neonatologists and offers the provision of hospitalization for ≥48 hours postpartum and universal prenatal care. This model presents an ideal basis for introducing the SCO strategy, thus discontinuing the concept of culture-negative sepsis as proposed by Cantey et al.11-13
In 2019, the Division of Neonatology and Neonatal Intensive Care, First Department of Obstetrics and Gynecology, Medical University of Warsaw, introduced a new approach for managing term and near-term newborns at risk for EOS. Prior to this time, there was no written policy for managing such infants. Rather, the diagnostic assessment was at the attending physician’s discretion and mainly based on laboratory test results (C-reactive protein [CRP] concentration and complete blood count with differential) of infants with risk factors. In comparison, the novel approach was based on an SCO strategy complemented by bedside verification of the blood culture volume. Consequently, the hospital pharmacy reported decreased utilization of ampicillin and gentamicin by our department. The purpose of this study was to assess the effect of this novel combined approach on the antibiotic exposure in infants born at ≥35 weeks of gestation.
Materials and Methods
This single-center study used a “before-and-after” approach. The study setting was a tertiary-level maternity hospital in Warsaw, Poland, with nearly 2000 deliveries annually. Two separate 12-month periods (pre-intervention: June 2017 to May 2018; post-intervention: June 2019 to May 2020) were analyzed, with a 12-month transitional period between them.
Medical records were searched to find infants born at ≥35 weeks gestational age (GA) who commenced antibiotic treatment within the first 3 days of life. Collected data included basic patient characteristics, clinical status at treatment onset, blood culture results, and duration of antibiotic therapy.
Our novel approach for managing term and near-term newborns at risk for EOS had 3 mainstays. First, the indications for the sepsis workup were limited and specified with the introduction of the new approach. Possible risk factors, such as prematurity, twin birth, or maternal diabetes, which were not considered independent risk factors, were not included (Table 1). Additionally, isolated symptoms, such as transient hypoglycemia or jaundice, were not indications for ruling out sepsis. Second, the SCO strategy was the first and only step in most cases. Asymptomatic infants at risk of EOS were managed with clinical observation alone. The nursing staff conducted clinical observations to evaluate the respiratory and heart rates, body temperature, and peripheral oxygen saturation. The frequency of the observations was at the attending physician’s discretion. Overall, the initial hours after birth required frequent observations (every 1-3 hours). Frequency was decreased to every 6 to 8 hours later. Observations were performed for 12 to 48 hours, depending on the infant’s risk category. Blood cultures were obtained from symptomatic infants or infants with a significant risk of EOS. However, other blood tests (eg, complete blood count with differential and CRP concentration) were not routinely ordered; they were limited to selected cases with more complicated clinical courses. The third mainstay of this approach was to draw adequate blood volumes for the culture. Volumes ≥1 mL were targeted to reliably exclude even low-level bacteremia. The volume of each sample was verified at the point of care by weighing the inoculated culture bottles using the same precision scale that had been used to prepare pre-weighed culture bottles. 14 Results were recorded and communicated to the patient’s physician and staff who collected the sample. The practical framework of the guidelines is presented in Table 1.
Guidelines Used During the Post-Intervention Period.

Abbreviations: PROM, prolonged rupture of membranes; GBS, group B Streptococcus; SCO, serial clinical observation; IAP, intrapartum antibiotic prophylaxis, GA, gestational age; IAI, intra-amniotic infection; CRP, C-reactive protein; CBC w/mdiff, complete blood count with manual differential; UTI, urinary tract infection.
Ethical Approval and Informed Consent
The Bioethics Committee of the Medical University of Warsaw granted a formal waiver for ethical approval of the study. Given that the study assessed the effect of a policy change, and no personal data were retrieved, the need for informed consent was waived.
Statistical Analysis
Collected data were analyzed using the R statistical package (version 4.1.1; R Foundation for Statistical Computing, Vienna, Austria). The significance level (α) was set to .05. The patients were divided into pre-intervention and post-intervention groups, and both groups were characterized and compared. Nominal variables are presented as n (% frequency), whereas continuous variables are presented as mean ± SD or median (Q1; Q3), depending on the distribution. Distribution normality was assessed using the Shapiro-Wilk test and involved the assessment of histograms, skewness, and kurtosis values. The correlation between nominal variables was analyzed using Fisher’s exact test. Differences in the levels of quantitative variables were analyzed using the Mann-Whitney U test or Student’s t-test. Odds/risk ratios and mean/median differences were estimated using 95% confidence intervals (CIs).
Results
This study recorded 2279 and 1952 live births during the pre-intervention and post-intervention periods, respectively. During each period, 92% of the infants were born at ≥35 weeks GA (2094 and 1791, respectively) and included in this study. Sex, mean birth weight, GA, Apgar scores after the first and fifth minutes of life, and mode of delivery were not significantly different between infants exposed to antibiotics during the pre-intervention and post-intervention groups (P > .05 in all cases). The characteristics of the patients exposed to antibiotics are presented in Table 2.
Characteristics of Patients Treated With Antibiotics.
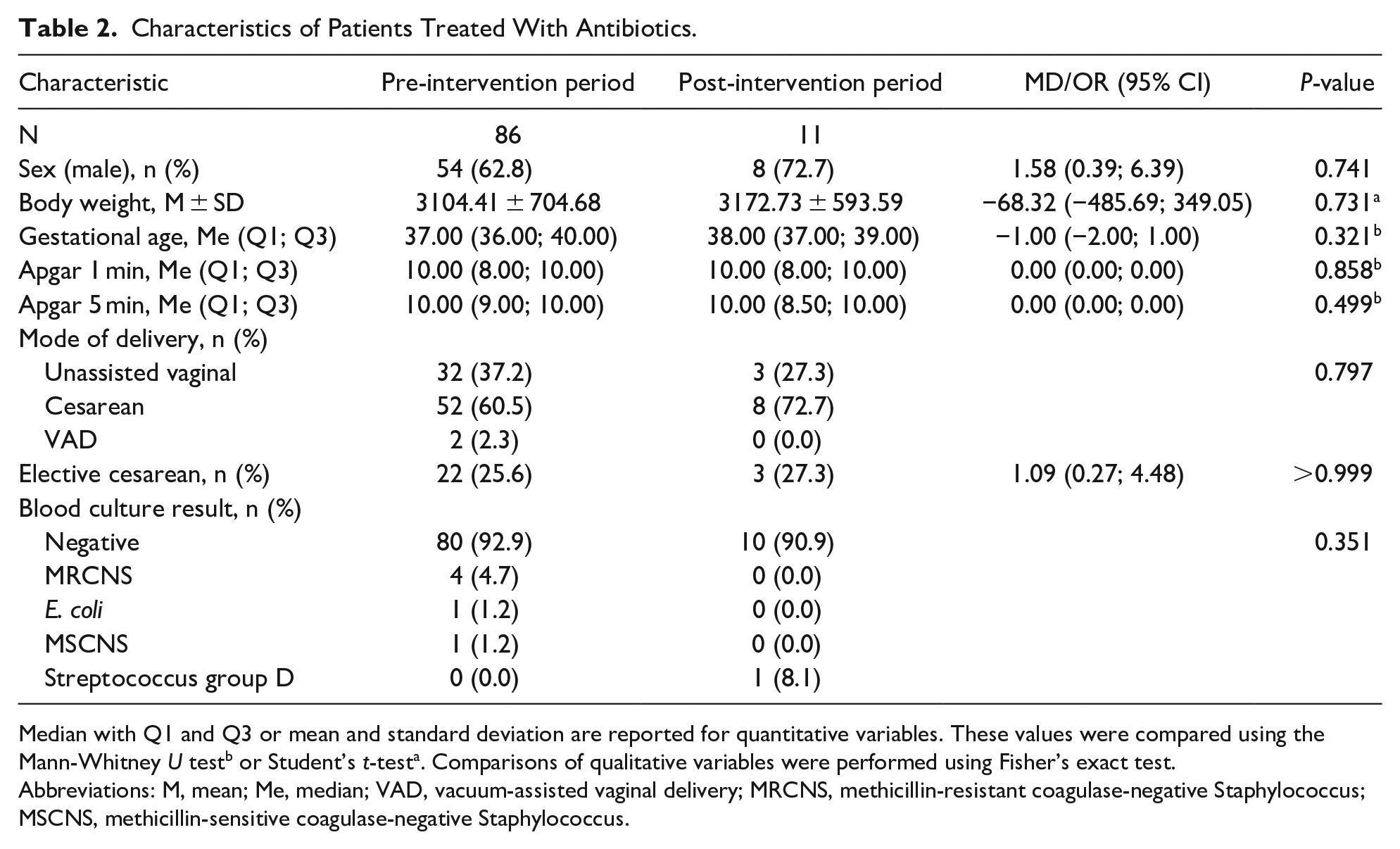
Median with Q1 and Q3 or mean and standard deviation are reported for quantitative variables. These values were compared using the Mann-Whitney U testb or Student’s t-testa. Comparisons of qualitative variables were performed using Fisher’s exact test.
Abbreviations: M, mean; Me, median; VAD, vacuum-assisted vaginal delivery; MRCNS, methicillin-resistant coagulase-negative Staphylococcus; MSCNS, methicillin-sensitive coagulase-negative Staphylococcus.
During the post-intervention period, 0.6% of infants commenced antibiotic therapy, whereas 4.1% of newborns received antibiotics during the pre-intervention period (P < .001; relative risk [RR]: 0.15; 95% CI: 0.08-0.28) (Figure 1).
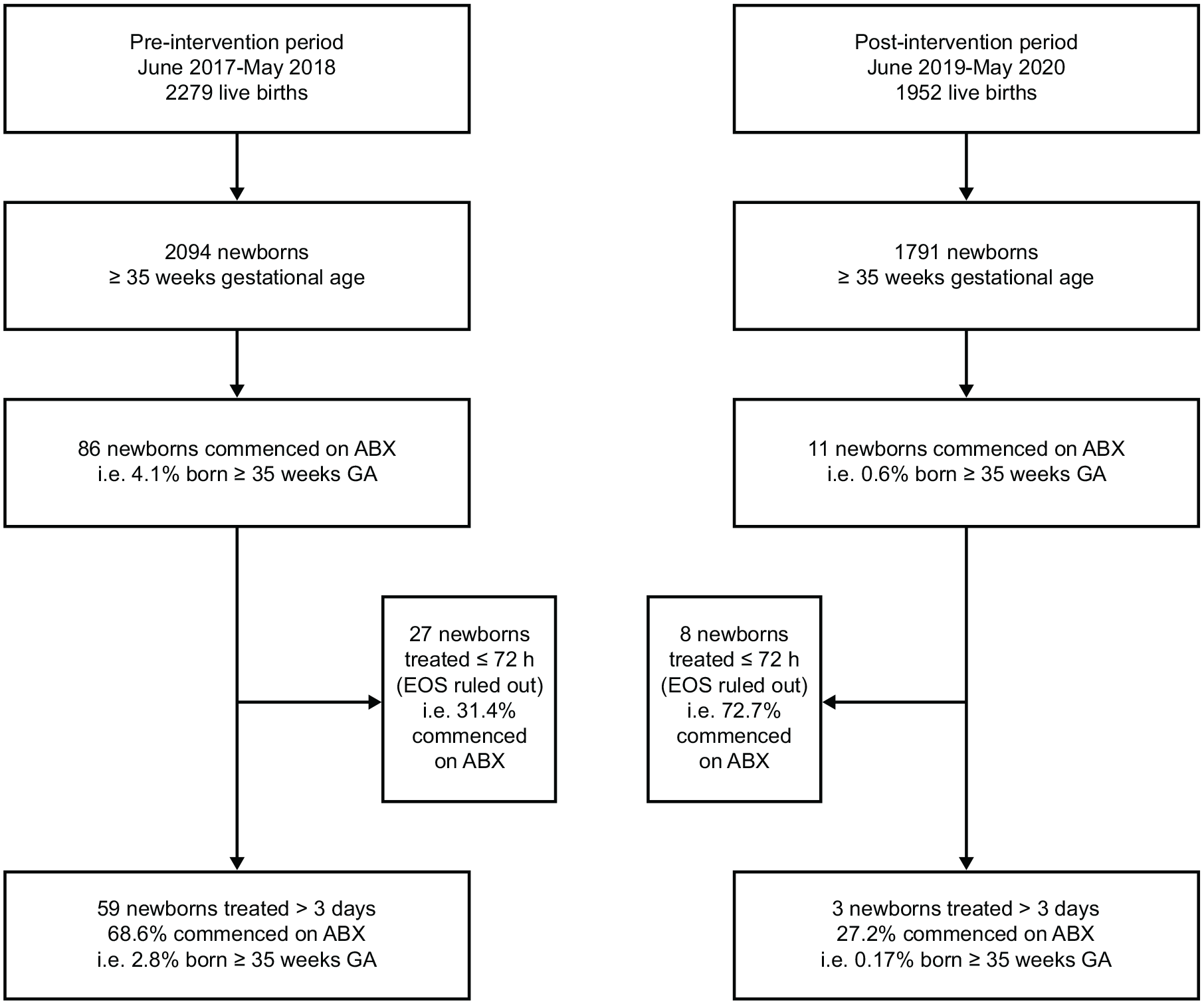
Flow diagram of the study.
Newborns started on antibiotics were less likely to be treated for >72 hours in the post-intervention group than in the pre-intervention period group (27% vs 69%; P = .015; RR: 0.43; 95% CI: 0.27-0.70).
Blood culture results were negative for 93% and 91% of samples from the pre-intervention and post-intervention groups, respectively (P = .351). Most infants from both groups were admitted to the neonatal intensive care unit during treatment initiation (66% and 91% of newborns from the pre-intervention and post-intervention groups, respectively; P = .164).
Furthermore, newborns in the pre-intervention group were treated with antibiotics longer than those in the post-intervention group (median difference: 2.00 days; 95% CI: 1.00-3.00; P = .003) (Table 3).
Comparison of Newborns Commenced on Antibiotics According to Study Period.

Abbreviation: GA, gestational age.
Discussion
This study observed a significant reduction in antibiotic use during the first days of life among infants born at ≥35 weeks GA. To our knowledge, the observed frequency of antibiotic utilization in infants post-intervention (0.6%) is among the lowest in the existing literature.15-20 Low antibiotic utilization could be beneficial for many reasons, including decreasing the exposure of uninfected infants to antimicrobials and reducing pain and stress related to repeated blood tests and maintaining venous access. Consequently, the novel approach used in this study could also reduce parental anxiety and mother-infant separation, which negatively affects bonding and breastfeeding initiation. Another possible advantage of this approach is the decreased cost of care resulting from the reduced use of antibiotics, disposable medical supplies, and laboratory tests. The main disadvantage of the approach might be the greater workload during night-shift hours.
The observed significant reduction in antibiotic use might be attributed to several factors. First, an unambiguous clinical status became the primary indicator of newborn well-being and the absence of systemic infection. Second, adequate and verified volumes of blood samples sent for cultures were the main test for ruling out EOS. Third, using CRP levels as a diagnostic tool was avoided in most common clinical scenarios.
The authors suggest that other clinical teams abandon CRP testing as part of routine sepsis workups because of its low positive predictive value. Accordingly, clinicians will constantly encounter a subset of uninfected infants with elevated CRP concentrations more often than infected infants. 8 Furthermore, infected infants with elevated serum CRP concentrations probably will be symptomatic as well. This suggestion, however, is inconsistent with the current Polish Neonatal Society guidelines, which advocate CRP testing. Interestingly, the guidelines do not recommend antibiotic treatment due to abnormal results if the blood culture result remains negative and the infant is asymptomatic. 21 The underlying principle of this recommendation is indicated by our findings that CRP testing adds little or no confidence for the clinical status of asymptomatic infants with normal results. However, unnecessary uncertainty can be introduced in frequently occurring cases of false-positive results, which often lead to prolonged antibiotic treatment, even if the blood culture result is negative.
SCO or SPE is well-established strategy originating from Italian centers16,19 with the lowest antibiotic utilization rate among all 3 aforementioned clinical strategies for managing infants at risk of EOS. This study added point-of-care verification of the volume of submitted blood culture samples to this simple and efficient concept. This was added because potentially inadequate sample volumes are the most critical reason why clinicians consider negative culture results unreliable. This concern is not speculative because surprisingly low inoculant volumes have been reported by several studies.22,23 The efficacy of real-time bedside verification of blood volumes sent for culture may cause negative culture results to be more trustworthy. Thus, patients treated during the post-intervention period of this study were more likely to be administered antibiotics for <72 hours. The approach used during this study may help restore confidence in the diagnostic potential of blood cultures.
This study had several limitations. First, this was a single-center study with a limited number of participants. Analyzed 12-month periods were arbitrarily chosen and no sample size calculations were done. Data of infants managed without antibiotic treatment during either period were not collected. Consequently, the frequency of risk factors or more appropriate antibiotic utilization metrics of both groups could not be reported. Second, EOS is a disease with a relatively low incidence; hence, not many affected patients were included in both study periods. Many asymptomatic newborns were discharged immediately after 48 hours. Thus, there is a possibility that EOS symptoms might have developed after discharge. Although most newborns with health problems are readmitted to the same hospital, this cannot be assumed.
These limitations suggest that further studies with larger sample sizes and more comprehensive data collection would be useful for verifying the effects of our approach.
Conclusions
This study found a significant reduction in antibiotic use among infants born at ≥35 weeks GA during the first days of life using a strategy of SCO complemented with point-of-care verification of the blood culture volume. Point-of-care verification of the volume of every blood sample seems to be a complementary add-on to SCO, which could enhance the process of ruling out sepsis.
These findings advocate the abandoning of CRP testing and support the importance of the clinical status and possible role of the point-of-care verified blood culture volume in diagnosing EOS.
Footnotes
Acknowledgements
The authors thank all the nursing staff and physicians of the Division of Neonatology and Neonatal Intensive Care for their cooperation. We also want to thank Urszula Malinowska for statistical analysis and Paweł Krajewski for general support.
Author Contributions
TW was responsible for study conception, data collection, analysis and interpretation, drafting the article, critical revision of the article and final approval of the version to be published. JR was responsible for study conception, data analysis and interpretation, critical revision of the article and final approval of the version to be published. Both authors agree to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
