Abstract
Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) Syndrome and Stevens-Johnson Syndrome (SJS) are severe cutaneous adverse reactions to drugs. Those reactions which are rare in children can be especially severe and challenging to diagnose and manage. Herein we present a 59-month-old male who presented with a rash, fever, and multiple organ dysfunction initiation of Phenobarbital for epilepsy. Diagnosis of ovelaping SJS and DRESS syndrome had been made based on clinical manifestations accompanied with skin biopsy according to RegisSCAR diagnostic criteria. A therapy with intravenous immune globulin (IVIG), corticosteroids and supportive care was given successfully for the patient. This case underscored the significance of promptly and effectively recognizing and managing these intricate reactions.
Keywords
Introduction
Aromatic anticonvulsants including phenobarbital, phenytoin, carbamazepine and oxacarbamazepin, have been known as the leading causes of drug-induced severe cutaneous adverse drug reactions, consisting of drug reaction with eosinophilia and systemic symptoms (DRESS), Stevens-Johnson Syndrome (SJS)/Toxic epidermal necrolysis (TEN) and Acute generalized exanthematous pustulosis (AGEP).
SJS/TEN manifest typically with skin detachment and mucosal erosions. The first symptoms onset approximately after several days of introducing the culprit medication (within 4 weeks). SJS/TEN typically present with a fever that is greater than 38°C, as well as ocular discomfort and a sore throat. Then, mucous membrane involvement happens and skin lesions with erythema, blisters and erosions appear and become more generalized by the time. These lesions can affect the eyes, nasopharynx, oropharynx, genitalia, or anus. In TEN, necrotic lesions expand rapidly with Nikolsky’s sign that characterizes with the epidermal sheet detached and sloughed by pressure. Differentiation between SJS and TEN is defined by the body surface area (BSA) involved in skin detachment, 10% in SJS; 10% to 30% in ovelaping SJS/TEN and more than 30% of BSA in TEN.
HSS/DRESS is characterized by exanthema and internal organs involvement. The new onset symptoms are flu-like symptoms accompanying fever greater than 38°C and variety of skin lesions such as maculopapular/morbilliform, exfoliative dermatitis and desquamation rashes. The index-day (time duration between taking medication to onset the first symptom is normally long, ranging from 2 to 6 weeks. This is high risk for misdiagnosis of HSS/DRESS. In HSS/DRESS, there are other signs that have been observed, including swelling of the face or extremities, palpable lymph nodes in more than one location and, in rare cases, involvement of the mucous membranes. When eosinophils or lymphocytes infiltrate tissues, internal organs can also be affected. The liver is commonly affected, which can be seen through elevated transaminase levels, as well as the kidneys, which can develop interstitial nephritis.
Overlapping between DRESS and SJS is rare but exists. Diagnosis of ovelaping SJS and DRESS syndrome had been made based on clinical manifestations accompanied with skin biopsy according to RegisSCAR diagnostic criteria. It is thought to involve a complex interplay between drug-specific immune responses and genetic factors. In some cases, an overlap between DRESS and SJS is challenging in the diagnosis and management.1-4 Recent reports have shed light on various aspects of these syndromes, including their epidemiology, clinical manifestations, pathogenesis, and management.1,5-8 For instance, Kardaun indicated a higher incidence of SJS and TEN in older adults with a priority suggestion for the age-specific preventive measures.1,7 Another study by Arici highlighted the importance of early recognition and prompt management of DRESS to avoid long-term complications. 9
The diagnosis and management of overlapping DRESS and SJS can be challenging, as it requires careful evaluation of the patient’s clinical features, medication history, and laboratory data. Treatment typically involves discontinuation of the offending medication, supportive care, and sometimes corticosteroids or other immunomodulatory therapies. In this context, updated reports of the overlapped clinical features of SJS and DRESS are crucial in early recognition and prompt intervention to improve patient outcomes and minimizing morbidity and mortality. Herein, we report the case of a 59-month-old male who developed an overlapping reaction between DRESS and SJS following treatment with Phenobarbital, an antiepileptic drug.
Case Presentation
A 59-month-old male patient presented to our institution (National Children’s Hospital) with a history of mental retardation and epilepsy who developed SJS and DRESS after being treated with Phenobarbital. Patient condition at the time of admission was shown in Figure 1. Previously at our institution, the patient who had been treated for seizures had been seizure-free for nearly 3 years. However, at age 58 months, he presented with recurrent seizures and was then diagnosed with epilepsy. The patient was prescribed Phenobarbital, which effectively controlled the seizures. After the 25th day of treatment with Phenobarbital, he presented with fever, fatigue, cough and sore throat. Cutaneous examination of his limbs, trunk, and abdomen showed widespread and symmetrical skin lesions covering 80% to 90% of the body including dark erythema and scattered vesicles with positive Nikolsky’s sign. The patient presented mucosal involvement with mouth ulcers and conjunctivitis. Also he developed acute liver failure and bronchitis. Laboratory findings showed elevated levels of white blood cell count, eosinophilia, C-reactive protein (CRP) and liver enzymes (Table 1). A skin biopsy taken from a lesion on his skin showed epidermal detachment and inflammatory cell infiltration (Figure 2 & Figure 3). Microbiological tests showed negative result. The patient was positive for the HLA-B*15:02 and negative for HLA-B*57:01, HLA-B*58:01, HLA-A*31:01, CYP2D6, VKORC1, CYP2C9, TPMT, DPYD, and CYP2C19 (Table 1).
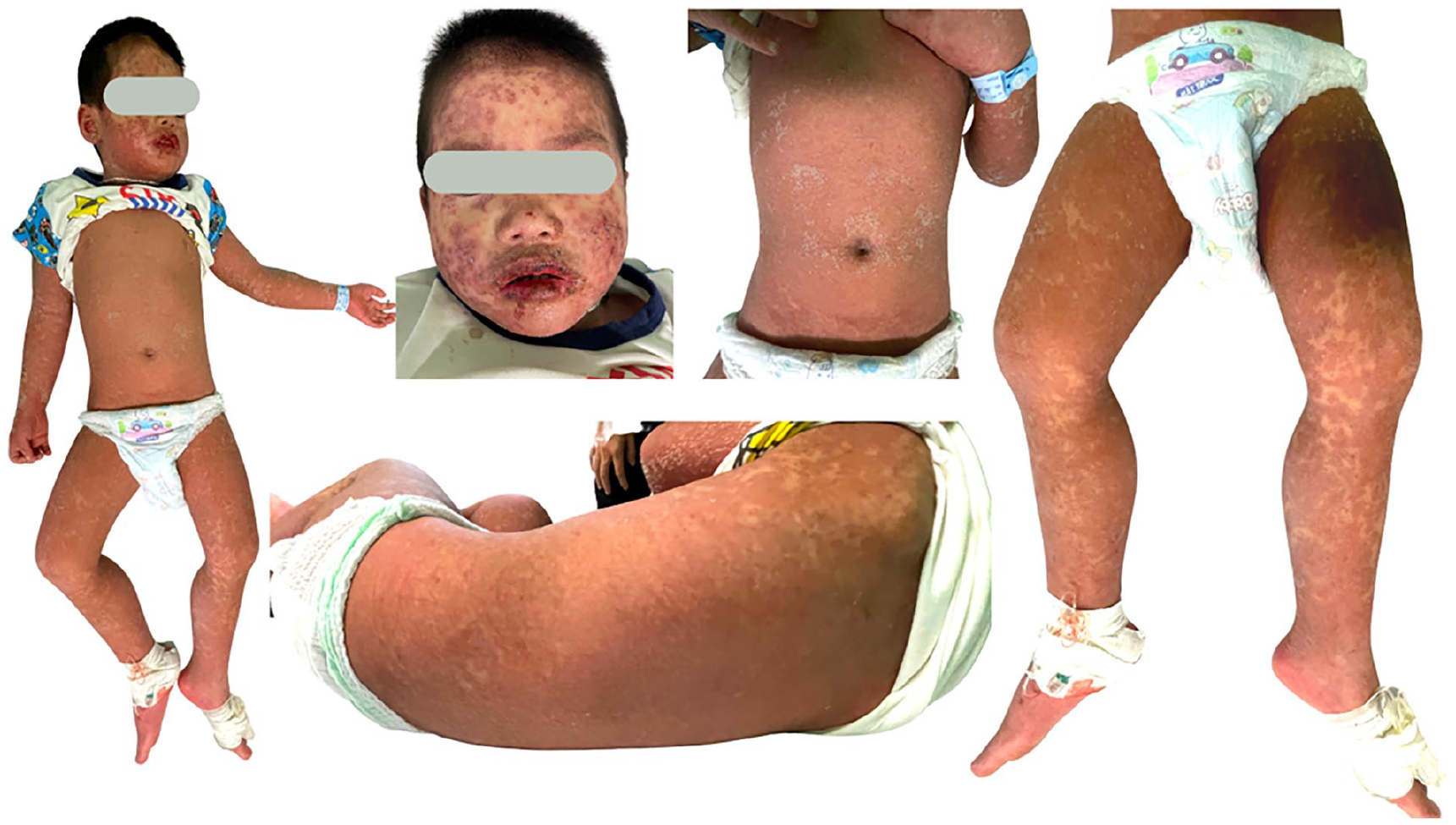
Patient condition at the time of admission.
Patient’s Blood Test Results.

Bold numbers indicate the values outside the reference range.

Biopsy of left thigh skin specimen, Immunofluorescence staining: IgA, IgM, IgG: Negative; C1q; C3c; Fibrinogen: Mildly positive.
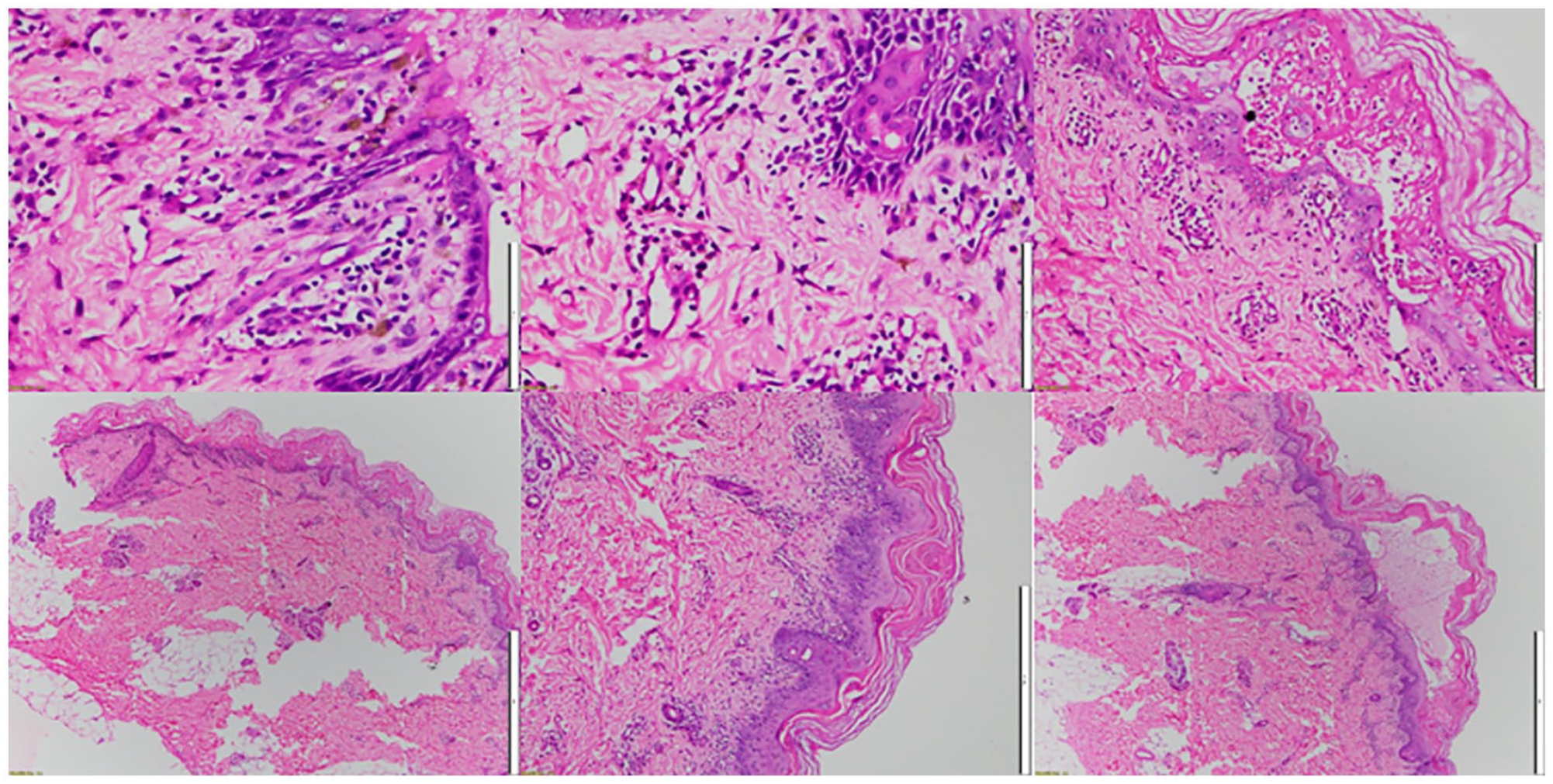
Biopsy of the left thigh skin specimen revealed the lesion to be concentrated in the epidermis at the basal membrane. The specimen exhibited areas of burns containing fluid and inflammatory cells, necrotic lesions, and epidermal surface dissection. Histopathology showed necrotic epidermis, with spongiosis and necrotic keratinocytes. There were also lymphocyte infiltrates present in the dermis.
According to the RegiSCAR criteria (Table 2),7,10 the clinical picture of this case could be classified as “probable” both for DRESS and SJS. Hence, an overlapping DRESS/SJS was a final diagnosis in our institution.
RegiSCAR DRESS Validation Score for Manifestations of Atypical/Typical DiHS.

Bold numbers indicate the scoring values of the patient.
Final score: 6 points => DRESS
<2: No DRESS;
2-3: possible DRESS;
4-5: probable DRESS;
>5: definite DRESS
During their 17-day treatment at our institution, Phenobarbital was discontinued and replaced by Levetiracetam as a means of seizure control. The patient was also received intravenous immunoglobulin (IVIG) (dose: 1 g/kg of body weight for 2 consecutive days), corticosteroids (1 mg/kg/day for 7 days) and antihistamines to manage SCAR symptoms. Besides supportive therapy for this patient involved several measures aimed at protecting and restoring the barrier function of the skin, preserving proper fluid balance, securing the airway, and addressing any infections. Additionally, we carried out monitoring and replacement of fluids and electrolytes, as well as providing nutritional support. It was typically necessary to ensure proper thermoregulation and adequate analgesia. Although the skin lesions of the patient had scabbed, with conjunctivitis and mouth ulcers resolved, systemic redness persisted. Cyclosporine was, therefore, added at a starting dose of 5 mg/kg/day in combination with a lower dose of corticosteroids for the next 4 weeks, resulting in significant clinical improvement in systemic redness (Figure 4). Periodic tests at follow-up were conducted to assess the patient’s liver and kidney function, as well as blood levels of cyclosporine. The results of these tests were consistently good. Following the initial treatment phase, the patient’s corticosteroid dosage was reduced to 0.5 mg/kg/day and continued at this lower dosage.

Patient condition after 4 weeks of treatment.
Discussion
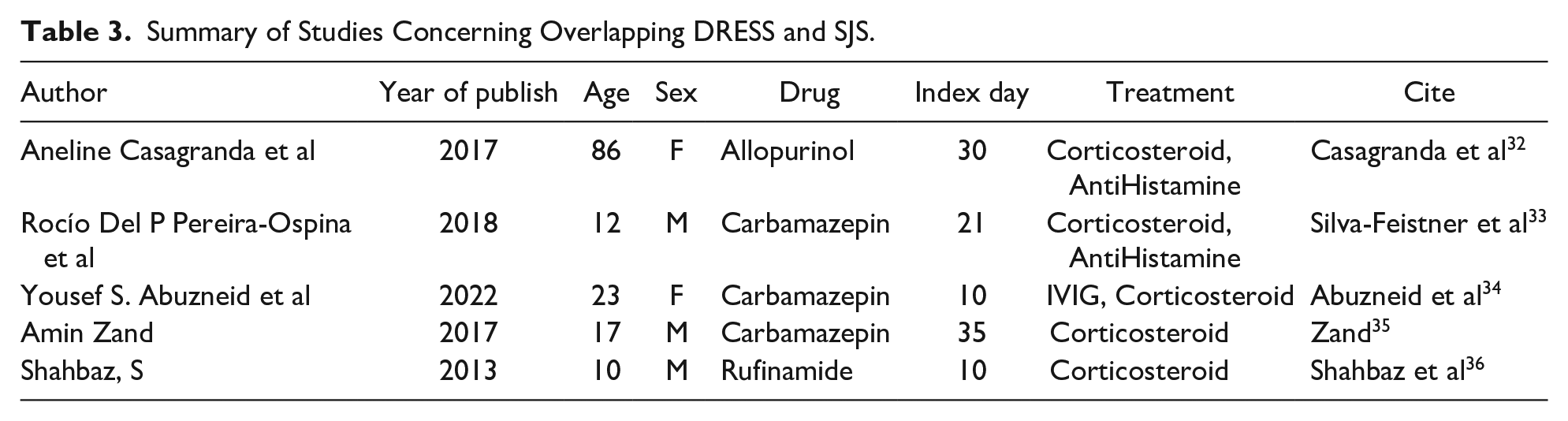
There have been several reported cases of overlapping DRESS and SJS in the literature (Table 3), and it is believed that both conditions may share similar immunologic pathways. Some authors suggested that the overlapping of these 2 conditions may represent a continuum of the same disease process, with different clinical expressions. 11 A study by Sassolas 12 reported that out of 223 patients diagnosed with SJS/TEN, 11 (4.9%) met the criteria for DRESS, indicating that overlapping of these 2 conditions is not uncommon. 8 As we discussed earlier, the histopathological findings of DRESS and SJS/TEN overlap in many cases, making it difficult to differentiate between the 2 conditions solely based on biopsy results. Up to 40% of patients with SJS/TEN have some features of DRESS on histopathological examination.7,13 Chen YC examined skin biopsy specimens from 23 patients with DRESS and 27 patients with SJS/TEN, finding that both conditions shared several common features including basal cell vacuolization, necrotic keratinocytes, lymphocyte infiltration and dermal edema. 1 However, this study also found that eosinophils were more common in DRESS than in SJS/TEN, while neutrophils and subepidermal bullae were more common in SJS/TEN. 1 Another study examined biopsy specimens from 33 patients with SJS/TEN and found that the presence of necrotic keratinocytes in the epidermis was the most important feature distinguishing SJS/TEN from other drug eruptions, including DRESS. 14 While there are several differences in histopathological findings between DRESS and SJS/TEN, there is also a significant amount of overlap, making it challenging to differentiate between 2 conditions based solely on biopsy results. Hence, a combination of clinical and laboratory findings is necessary to make an accurate diagnosis.
Summary of Studies Concerning Overlapping DRESS and SJS.
The occurrence of severe T-cell mediated reactions, such as abacavir hypersensitivity, SJS/TEN, and DRESS, have been strongly associated with specific HLA Class I genes. As a result, pre-prescription screening strategies have been implemented to identify individuals who may be at risk for these reactions.15-18 However, it is currently unclear whether exanthems (skin rashes) are solely caused by parainfectious events related to viral or bacterial antigens, or if they are a secondary cutaneous reaction to drugs stimulated by infectious agents. 19 Severe cutaneous adverse reactions (SCARs) are rare but can lead to significant morbidity and mortality. Biomarkers that are minimally invasive, biologically relevant, and linked to disease mechanisms are desired to aid in the diagnosis and management of these conditions. One promising biomarker for drug-induced hypersensitivity syndrome/drug reaction with eosinophilia and systemic symptoms (DIHS/DRESS) is the serum CCL17/TARC level, which is associated with Th2 skewing. Other potential biomarkers for SCARs include chemokines such as CCL22/MDC for DIHS/DRESS and CXCL9/MIG and CXCL10/IP-10 for SJS/TEN. 20 Large-scale clinical trials are needed to evaluate the usefulness of these biomarkers.
Phenobarbital is frequently used as the initial anticonvulsant treatment for children in Thailand, however, it is also one of the major culprits responsible for inducing hypersensitivity to aromatic anticonvulsants. Individuals who develop hypersensitivity may experience skin reactions such as maculopapular rashes or SCARs, which can manifest as Stevens-Johnson Syndrome (SJS), drug reaction with eosinophilia and systemic symptoms (DRESS) or toxic epidermal necrolysis (TEN). The occurrence of aromatic anticonvulsant hypersensitivity is given to be between 1/1000 and 1/10 000 21 and it has a high mortality rate, especially for patients with SCARs. 22 In Thailand, phenobarbital is also a common cause of SCARs. Recent evidence suggests that pharmacogenetics may play a role in predicting anticonvulsant-induced SCARs, but only a limited number of studies have investigated the association between human leukocyte antigen (HLA) genotypes and phenobarbital hypersensitivity. A small Japanese study demonstrated an association between HLA-B51:01 and phenobarbital-induced SJS/TEN. Eight patients who developed these reactions with the use of phenobarbital were recruited from the Japan Severe Adverse Reactions (JSAR) research group and RIKEN, with one patient having also received phenytoin. The onset of the reaction after drug initiation was slightly longer than those induced by phenytoin at 15.1 ± 7.1 days. In comparison to Japanese volunteers who were in good health, 8 patients were studied, out of which 6 were found to have HLA-B51:01. A significant association was observed between this allele and phenobarbital-induced SJS/TEN (OR 16.71, 95% CI 3.66-83.1). 23 In a Han Chinese population, 24 the HLA-B15:02 genotype has been linked to Carbamazepine-induced SJS; while in a Japanese population, 25 HLA-B58:01 has been associated with allopurinol-induced SJS/TEN. However, the association of HLA-B15:02 or HLA-B15:02 with phenobarbital-induced DRESS/SJS have been not reported in current populations.
CYP2C19 is a crucial cytochrome P450 enzyme that metabolizes aromatic anticonvulsants. It maintains the balance between bioactivating and detoxifying pathways, as an imbalance can lead to an increase in hapten formation, which triggers immune responses. 26 Genetic polymorphisms of CYP2C19 affect metabolic rates 27 and are highly prevalent in Asians.28,29 Common variants like CYP2C19*2 and CYP2C19*3 result in poor metabolizers. Following the research conducted by Wiparat Manuyakorn, patients in Thailand who had the CYP2C19*2 genetic variation were 4 times more likely to experience SCARs from phenobarbital compared to those without this variation. 30 CYP2C19 genotype has been shown to play a significant role in the metabolism and elimination of phenytoin and phenobarbital. 31 However, we did not find the significance of CYP2C19 in our patient following treatment with Phenobarbital. Further investigation through larger studies is necessary to assess the association between the CYP2C19 gene and the development of SJS and DRESS in Vietnamese patients undergoing treatment with Phenobarbital. We also tested several more genotypes such as CYP2D6, VKORC1, CYP2C9, TPMT, and DPYD, but the results are not relevant to the present Vietnamese case.
Overlapping DRESS and SJS/TEN is a rare but well-recognized phenomenon in clinical practice. Several reports have investigated the clinical features, laboratory findings, and outcomes of patients with overlapping DRESS and SJS/TEN. One study reviewed 22 cases of overlapping DRESS and SJS/TEN from the literature and found that allopurinol, carbamazepine, and sulfamethoxazole/trimethoprim were the most common causative drugs. 37 Patients with overlapping DRESS and SJS/TEN had a longer duration of drug exposure, a higher incidence of eosinophilia, and a higher mortality rate compared to patients with DRESS or SJS/TEN alone. 37 Kardaun analyzed data from the EuroSCAR study, a large international registry of patients with severe cutaneous adverse reactions, and identified 37 cases of overlapping DRESS and SJS/TEN.4,7 Patients with overlapping DRESS and SJS/TEN had a higher incidence of liver involvement, lymphadenopathy, and eosinophilia compared to patients with SJS/TEN alone.4,7 In addition, a case series by Ponzo et al 38 reported on 5 cases of overlapping DRESS and SJS/TEN and found that all patients had a history of antiepileptic drug use, with carbamazepine being the most common culprit. 38 The study also reported that all patients had a delayed onset of symptoms and a prolonged duration of hospitalization. 38 Overall, these studies suggested that overlapping DRESS and SJS/TEN was associated with a higher morbidity and mortality rate, and might be characterized by a longer duration of drug exposure, higher incidence of eosinophilia, and more severe liver involvement. Early recognition and prompt management of these severe cutaneous adverse reactions are crucial in improving patient outcomes.32,36,37
Conclusions
An overlapping DRESS/SJS is a rare but serious cutaneous adverse drug reactions that can occur in response to certain medications. This updated case report highlights the significance of distinguishing the diagnosis and the management plan between SJS and DRESS. Also, we showed the successful management of SCAR in a pediatric patient using a combination of pharmacological and supportive care measures. Incorporating cyclosporine into the patient treatment plan resulted in significant improvement in systemic redness.
Footnotes
Acknowledgements
We would like to express our gratitude to the patient and their family for their unwavering support. We would also like to acknowledge the nursing staffs, treating physicians, and supporting staffs at Vietnam National Children’s Hospital, VinBigData, and Vinmec Times City International Hospital for their valuable contributions to the management of our patient.
Author’s Note
Dinh Van Nguyen is alsoaffiliated to Department of Medicine, Penn State University, USA.
Author Contributions
KVN and DVN contributed to the conception and designed and reviewed the manuscript. QVV, MHT, HQN, CQL, BCTD and HCC contributed to the writing of the manuscript. All authors contributed to the article and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Khiem Van Nguyen was funded by PhD Scholarship Programme of Vingroup Innovation Foundation (VINIF), code VINIF.2022.TS059.
Data Availability Statement
The data used in this article are available upon request from the corresponding author (K.V.N. Email:
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of Vietnam National Children’s Hospital.
Informed Consent Statement
The patient’s family was informed about our intention to involve him in a case study, and they agreed to partake in the study. A written informed consent was obtained from the legally authorized representative of patients for their anonymized information to be published in this article.
