Abstract
Congenital hepatic arteriovenous malformations (HAVM) are rare and serious with a known high mortality rate. They are mostly described in the term population and, if symptomatic, present with early-onset congestive heart failure. To our knowledge, there have been no published cases of an affected very preterm infant. Prenatal diagnosis and preprocedural planning are paramount in these critically ill infants. Strategies for management of HAVM differ depending on the size of both the infant and HAVM as well as severity of symptoms. Management may include supportive pharmacologic therapy, transcatheter embolization, and surgical resection or ligation. In this report, we present the case of a 28-week gestation infant with high-output cardiac failure secondary to prenatally diagnosed hepatic arteriovenous malformation. On day of life 3, the infant underwent successful transcatheter occlusion. However, despite maximal medical management and partial embolization of his HAVM, he did not survive.
Introduction
Congenital hepatic arteriovenous malformation (HAVM) is a rare developmental vascular disorder characterized by direct arterial-to-venous connections within the liver. There are limited reports of HAVM in preterm infants. The mortality in HAVM is known to be high in term infants, with reports ranging from 50% to 90%. This number is likely to be higher in the very and extremely preterm infant, such as the one whom we describe in this case report.
Congenital HAVM most commonly present with early congestive heart failure. In-utero left to right shunting through the ductus arteriosus is limited given the high pulmonary vascular resistance (PVR) that exists at that time. At birth, decreased pulmonary vascular resistance leads to increased pulmonary blood flow, and increased systemic vascular resistance leads to increased flow through the HAV. These hemodynamic shifts often worsen the clinical condition. Neonatal presentation can also include hepatomegaly, consumptive coagulopathy, anemia, portal hypertension, hydrops fetalis, and persistent pulmonary hypertension of newborn.1-3 HAVM produce a low-resistance high-flow arterial to venous shunt (AVM “steal”). The physiology of postnatal circulatory transition with increased systolic arterial blood pressure after delivery leads to an increase in shunt volume. There is a resultant rise in cardiac output to compensate, which can bring about high-output cardiac failure. While hypotension and hypoxemia are common sequelae of symptomatic congenital HAVM, vasopressors and pulmonary vasodilators may worsen vascular steal, pulmonary over-circulation, and high-output heart failure, making these associations difficult to manage.
Prenatal diagnosis is important and usually achieved by ultrasound, although plain film radiography, computed tomography (CT)/CT angiography (CTA), magnetic resonance imaging (MRI), and angiography may also have utility. 4 Management options for infants vary depending on severity of symptoms and size of the HAVM. Prenatal diagnosis along with preprocedural planning can allow for prompt intervention in the infants that require more than pharmaceutical management. Most HAVM have been described in term infants, and in this report, we demonstrate feasibility of transcatheter occlusion as a management tool for very preterm infants.
Case Presentation
A 28-week gestation, 1.42 kg male newborn with prenatal diagnosis of HAVM and resulting fetal high-output cardiac failure was delivered via cesarean section due to concern for maternal Ballantyne (“mirror”) syndrome to a 41-year-old Caucasian mother. His HAVM was diagnosed via prenatal ultrasound and noted on fetal echocardiogram.
He appeared cyanotic and floppy at delivery and required intubation. Due to desaturations, he was placed on high frequency ventilation and inhaled Nitric Oxide (iNO), and surfactant therapy was administered. Initial chest X-ray revealed a very large cardiac silhouette occupying nearly the entire thoracic cavity with minimal lung volumes visible. Umbilical arterial and venous lines were placed. For the first few hours of life a profound metabolic and respiratory acidosis with incalculable pH and bicarbonate was present. Evidence of cell lysis was also present, with a hematocrit of 29, platelet count of 59, umbilical cord oozing, and elevated liver function tests. The infant was given multiple blood product transfusions and fluid boluses in the resuscitation room, including sodium bicarbonate, packed red blood cells, platelets, and cryoprecipitate.
An echocardiogram demonstrated a moderately dilated heart, hyperdynamic systolic function, atrial septal aneurysm, patent foramen ovale with moderate shunt, bidirectional inter-atrial shunt, and an unrestrictive PDA. The patient was discussed with pediatric cardiology, and recommendations were made to continue iNO, start milrinone to support right heart function, and give diuretic therapy as needed. Given the critical clinical status, the patient was determined to be unstable for transport to the level IV NICU at the local children’s hospital for further subspecialist intervention. His clinical status continued to decline the following day, with increased ventilatory needs, decreased perfusion and worsening metabolic acidosis. Renal failure persisting from birth had resulted in profound oliguria, hyponatremia, and hyperkalemia requiring treatment. The infant also became hypotensive, necessitating the initiation of multiple vasopressors.
At this point, the case was discussed with pediatric interventional radiology (IR) who agreed to partially embolize the hepatic AVM with a goal of slowing the venous return to the heart, if the infant could be stabilized for transport to the local children’s hospital. The family was counseled extensively regarding therapeutic options in relation to morbidity and mortality.
He was transferred later that evening and scheduled for HAVM embolization the following morning. Overnight, urine output was minimal, for which he was fluid restricted, and he continued to be anemic and thrombocytopenic requiring multiple blood product transfusions.
The patient was transported to the IR suite on day of life 3, and 2 IR physicians proceeded to partially embolize the patient’s HAVM. At that time, the patient’s weight was 1.74 kg. However, the increase in weight from birth weight was likely owing to significant volume administration as part of resuscitative efforts. Of note, this case was very complicated given the infant’s weight and vessel size, which greatly limited imaging options and contrast dosages. An initial angiogram was performed through the patient’s umbilical arterial catheter. This demonstrated a large and complex AVM involving the liver, which had a dominant draining vein at the porta hepatis (Figure 1). Venous access was obtained through the right internal jugular vein, and the large draining vein was accessed via ultrasound (Figures 2 and 3). Coils were then placed into the draining vein until there was stasis of blood flow (Figure 4). A final angiogram demonstrated markedly decreased flow through the AVM. During the final angiogram it was noted that the patient’s aorta was diminutive beyond the level of the AVM and the renal arteries were not seen well.

Arteriogram through the UAC showing a large AVM of the liver.

Venogram with catheter in the large draining vein of the AVM.
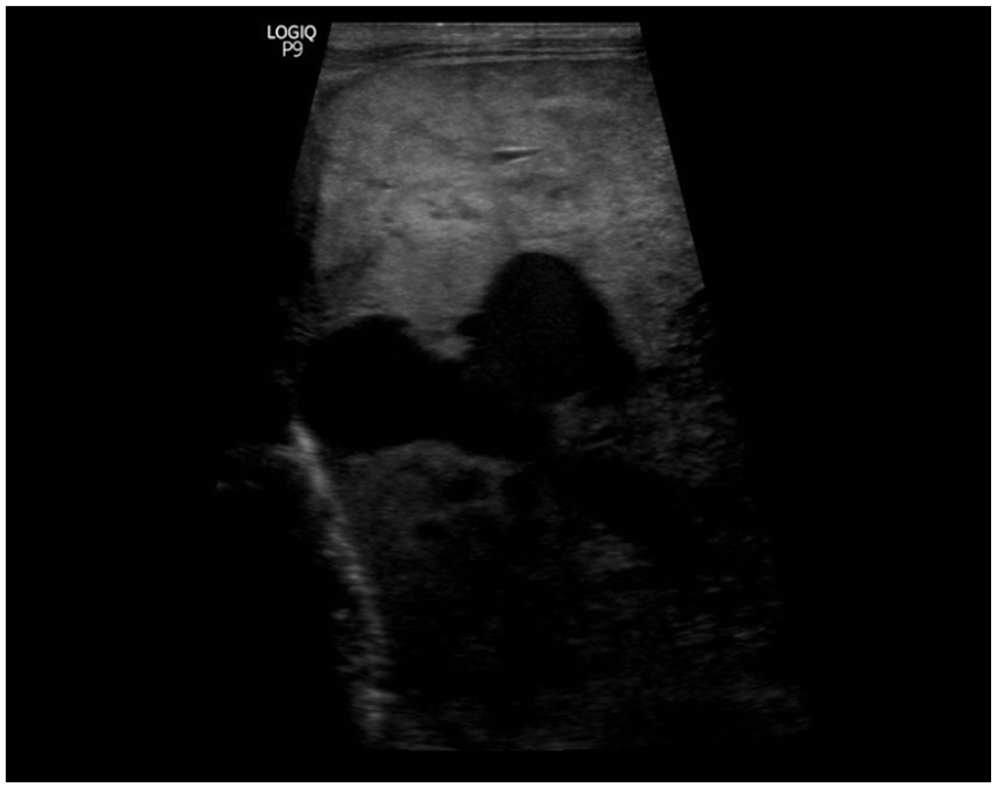
Ultrasound showing the large vein coming from the IVC.
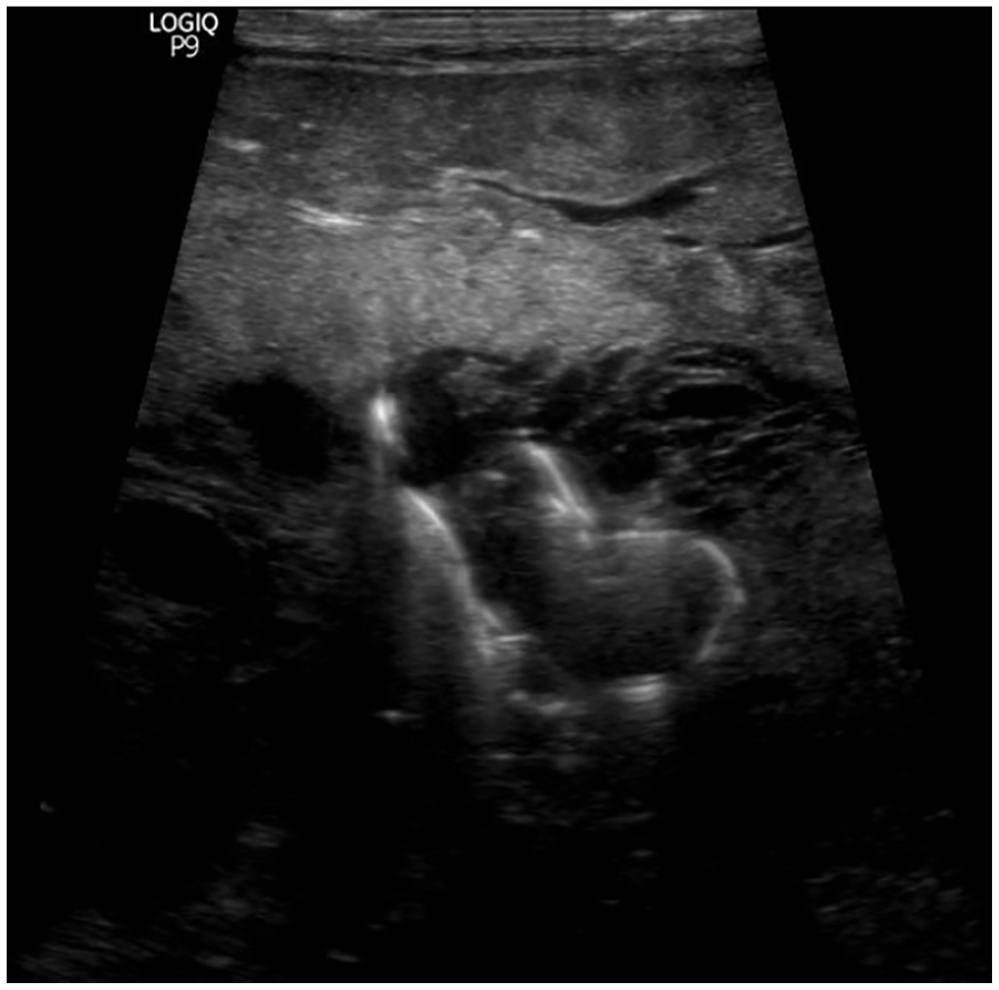
Ultrasound showing coil placement.
Following the procedure with IR, his direct hyperbilirubinemia continued to worsen. Anemia and thrombocytopenia persisted. He continued to require maximum respiratory support and multiple vasoactive agents for profound hypotension. His case was discussed among neonatology staff, and he was determined not to be a candidate for extracorporeal membrane oxygenation due to his size and gestational age.
An echocardiogram was repeated post AVM coiling with no significant changes appreciated. A head ultrasound was obtained at that time with no intraventricular hemorrhage noted.
The evening of day of life 3, he began to decompensate with worsening hypotension and hypoxia despite maximal support. He experienced a pulmonary hemorrhage after routine suctioning. He did begin to produce urine, but his serum potassium levels continued to increase, and he remained hypoglycemic and coagulopathic.
On day of life 4, his high-output cardiac failure persisted, and he became unresponsive to pressors, fluids, and blood products. His head ultrasound was repeated and was now significant for bilateral intraventricular, thalamic, right occipital, and cerebellar hemorrhages with mild ventriculomegaly present. Multi-organ failure was noted to be progressing, and family was notified that no further interventions were possible. His family was counseled that due to his coagulopathic status with newly noted intracranial hemorrhages, there was a very low chance of survival with likely poor neurodevelopmental outcomes if he were to survive. Parents agreed to consider transition to comfort care measures. He remained profoundly hypoxic for the hours leading up to his death. He proceeded to have a rapid decline in his heart rate likely secondary to a cardiac arrythmia from rising serum potassium.
Parents consented to an autopsy which was significant for hepatomegaly involved by a large vascular malformation, numerous organ infarctions, the intracranial hemorrhages, and diffuse alveolar damage.
Discussion
Fetal and neonatal hepatic arteriovenous fistulas are rare, with incidence of congenital HAVM <1:100 000. To our knowledge, there have been no published cases of an affected very preterm infant, with the most premature reported patient having a gestational age at birth of 34 weeks. 5 The mortality rate is very high, and large lesions with multiple feeder vessels are typically associated with poor outcomes.
In the workup of HAVM, including diagnosis and preprocedural planning, plain film radiography, ultrasonography, computed tomography (CT)/CT angiography (CTA), magnetic resonance imaging (MRI), and angiography can all provide different degrees of information. 4 Ultrasonography is the recommended first step in diagnosis, revealing dilated vascular channels in the parenchyma accompanied by distended hepatic arteries and veins. Arterial spectral Doppler interrogation will often show increased systolic and diastolic flow with resultant low resistive indices. Venous spectral Doppler analysis may show arterialization of venous waveforms because of intralesional arterial-to-venous shunting. 6 Fetal diagnosis is also possible with prenatal ultrasound when multiple engorged vascular channels are detected in the fetal liver. 5 Should this be detected on prenatal ultrasound, fetal MRI has come to play a key role in further evaluation of the anomaly.
Strategies for management of HAVM include supportive pharmacologic therapy of congestive heart failure with digoxin and diuretics, transcatheter embolization, surgical resection, and ligation, and, in cases of diffuse HAVM, possibly liver transplantation.7-11 Surgical intervention is typically reserved for more simple and smaller HAVM which can be resected and has a high mortality risk in critically ill infants. The transarterial approach depending on HAVM composition can be a stop gap measure to curative. Regarding the former, the goal is to close the largest, highest flow fistulae to acutely stabilize hemodynamics. This allows time for the patient to grow and potentially receive additional staged embolizations later to completely close the lesion. Embolization is not without risk and may be unsuccessful due to collateral circulation, 12 and successful embolization can be complicated by variceal bleeding, cholangitis, hepatic insufficiency, and sepsis.13-15
In our case, one point of discussion is the fact that the goal of coil placement is to cause blood to clot around the coils and then ultimately turn to scar. In a preterm infant with such a large dominant draining vein, it is conceivable that by inducing formation of this clot we caused the child to have a consumptive coagulopathy by depletion of clotting factors. In future cases, it may be prudent to empirically administer clotting factors and platelets during and after such a procedure. Additionally, this patient required inhaled pulmonary vasodilatory therapy and systemic vasoactive agents for management of hypoxemia and hypotension, which as previously stated may further contribute to high-output cardiac failure.
Conclusions
The literature on congenital HAVM is very limited, particularly in the preterm newborn population. While the mortality rate of HAVM is high even in full term infants, it stands to reason that survivability is still lower in the preterm population. Our very preterm newborn patient did not survive the complications of a large congenital HAVM despite successful transcatheter occlusion. In unique cases such as these, directing efforts toward comfort care measures may be a reasonable alternative to intensive care.
Footnotes
Acknowledgements
Not applicable
Authors’ Contributions
JJ drafted the abstract and introduction and edited the entirety of the report. AL drafted the discussion and conclusion and edited the entirety of the report. KW drafted the case presentation and discussion. MA drafted the case presentation.
Availability of Data and Materials
Not applicable.
Consent for Publication
Written parental consent for publication obtained and will provide if requested.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Reviewed by University of Arkansas for Medical Sciences IRB and determined that this project is NOT human subject research. Number: 275545.
