Abstract
The prevalence of patent ductus arteriosus (PDA) in preterm infants is high. There is little information about the therapeutic effect and safety of rectal acetaminophen in the treatment of PDA. We aimed to compare the therapeutic effect and safety of oral and rectal acetaminophen on PDA in preterm infants. This study was a single-blind randomized clinical trial using 40 preterm infants. The cases were hospitalized in the neonatal intensive care unit of Mohammad Kermanshahi and Imam Reza hospitals of Kermanshah. Subjects were randomly divided into 2 groups, the first group was treated with oral acetaminophen and the second group was treated with rectal acetaminophen. The presence of PDA and response to treatment was assessed based on pre- and post-treatment echocardiographic criteria. The likelihood of complications or prohibition of acetaminophen use was assessed with paraclinical tests before and after treatment. The neonates were in the age range of 30 to 35 weeks. Twenty-one cases (52.5%) were boys and 19 cases (47.5%) were girls. Two cases in the oral-acetaminophen group and 1 case in the rectal-acetaminophen group needed the second round of treatment. There was no difference between the success of treatment and the type of treatment. The study showed that there was no difference between PDA treatment of preterm infants with oral and rectal acetaminophen. Also, no side effects were observed in treatment with any of the treatments. Therefore, it could be suggested that in infants who are intolerant to oral acetaminophen, the rectal form can be used.
Introduction
The ductus arteriosus (DA) is a vascular channel between the aorta and the pulmonary artery (PA) that diverts blood away from lungs and direct it to systemic circulation during pregnancy. 1 Therefore, its openness is necessary for the life of the fetus, but after birth its closure is necessary. 2 During pregnancy, the DA and the placenta produce vasodilators that keep the DA open, but, in term infants, as they are born, the amount of contractile factors increases and the sensitivity of DA to prostaglandins decreases, and sensitivity to oxygen increases which causes ductal constriction. In contrast, in premature infants, DA sensitivity to vasodilators increases, although this sensitivity decreases with increasing neonatal age. 3
Patent ductus arteriosus (PDA) is a major life-threatening problem in premature and low birth weight infants. 2 In 60% to 70% of preterm and low birth weight infants, the DA remains open. 2
Long-term PDA status in premature infants can be associated with significant complications such as ventilator dependency, respiratory distress syndrome (RDS), impaired blood supply to vital organs, edema and pulmonary hemorrhage, and long-term effects such as necrotizing enterocolitis (NEC), intraventricular hemorrhage (IVH), bronchopulmonary dysplasia (BPD), retinopathy of prematurity (ROP), and eventually mortality due to increased complications. 4 Due to the fact that spontaneous closure of this duct occurs in only one third of premature infants, most of them need medical treatment or surgical closure of the duct. 1
Currently, the first treatment for PDA includes nonspecific cyclooxygenase (COX) inhibitors (indomethacin, ibuprofen). These drugs work by inhibiting cyclooxygenase and stopping the synthesis of prostaglandins E2, F2a, I2, and thromboxane A2. This is followed by vascular smooth muscle constriction, local ischemia, angiogenesis, DA intima regeneration, and wall fibrosis and DA closure.1,5 Ibuprofen and indomethacin are standard treatments for PDA closure, but due to possible side effects in the gastrointestinal tract, kidney, chronic lung disease, thrombocytopenia, and hyperbilirubinemia, it is preferable to use acetaminophen with fewer side effects. 5
Surgery for PDA closure is performed when treatment with COX inhibitors is contraindicated or unsuccessful. 4 In addition, PDA closure may be associated with complications such as chronic lung disease, heart disease, neurodevelopmental disorder, and ROP. 1 Therefore, it is desirable to prescribe drugs with no contraindications and less side effects to close PDA. The effect of acetaminophen on PDA closure was first reported in 2011, 6 and extensive studies on its effects have been performed since then.5,7,8 Due to its properties such as safety, availability, low price, lack of side effects related to nonsteroidal anti-inflammatory drug (NSAIDs) and the fact that this drug has been used as an anti-inflammatory and analgesic treatment in infants for many years, acetaminophen will gradually replace NSAIDs for PDA medical closure. 9
Following our previous study on the efficacy of rectal acetaminophen in PDA closure, 10 here we aimed to further evaluate the therapeutic effect and safety of acetaminophen oral and rectal therapy on PDA in preterm infants.
Materials and Methods
This study is a single-blind, randomized clinical trial infants who were admitted to the neonatal intensive care unit of Mohammad Kermanshahi and Imam Reza hospitals, Kermanshah, Iran.
The participants were randomly assigned in a 1:1 ratio. In order to generate an allocation sequence, simple random allocation was applied using an Excel file; 40 eligible patients were enrolled in the study (20 individuals in each group). The Research Assistant allocated participants to the groups using the generated allocation sequence. For allocation sequence concealment, the study arm for each patient was contained in a sealed envelope labeled with a number from 1 to 40.
The inclusion criteria included gestational age less than 35 weeks and 6 days and postnatal age of more than 72 hours and less than 14 days, with PDA (confirmed by Doppler echocardiography based on the inner diameter of the arterial canal equal to or greater than 1.5 mm and the ratio of left atrial to aortic root diameter greater than 1.6), need for treatment from the third day onwards (72 hours after birth), no need for immediate action at the age of 1 to 3 days, and no contraindications to acetaminophen.
Neonates with the following criteria were excluded: rectosigmoid abnormalities, neutropenia less than 1500 cell/ml, platelets less than 30,000 per ml, NEC, IVH grade 3 and 4, liver failure or elevated liver enzymes, G6PD deficiency, hypovolemic or septic shock, gastrointestinal perforation or peritonitis, need for blood transfusion, renal failure and oliguria and creatinine clearance less than 10%, oral intolerance and vomiting, and respiratory rate more than 80 minutes.
After subjects’ selection, a checklist was prepared for recording of demographic profile and baseline clinical data of all cases including sex, weight, gestational age, and age at PDA diagnosis.
Patients were randomly divided into 2 groups. In the first group, treatment with oral acetaminophen at a dose of 25 mg per kg body weight was started and then continued with 15 mg per kg body weight every 8 hours for 3 days (10 doses in total) in infants weighing more than 1000. In the second group, treatment with rectal acetaminophen at a dose of 25 mg per kg body weight was started and then continued at a dose of 15 mg per kg body weight every 8 hours for 3 days (10 doses in total) in infants weighing more than 1000 g.
In both groups, if the infant weighed less than 1000 g, the initial dose was 15 mg/kg body weight, and the subsequent doses were 7.5 mg/kg body weight every 8 hours for 3 days (10 doses in total). These doses were considered based on the dose of NEOFAX with minimal side effects for infants. 11 The presence of PDA and response to treatment was assessed based on pre- and post-treatment echocardiographic criteria. The likelihood of complications or prohibition of acetaminophen use was assessed based on paraclinical tests before and after treatment. The criterion for starting treatment were age over 72 hours and less than 14 days and the internal size of PDA is higher than 1.5 mm and the ratio of left atrium to aortic root is more than 1.6.
The SPSS software was used for statistical analysis version 16 (SPSS Inc., Chicago, IL, USA). Continuous and categorical variables are presented as mean ± standard deviation (SD), number (%), respectively.
Independent-samples t-test was used for between-group comparisons as appropriate and paired t-test was used for within-group comparisons. variables were compared using chi-square test. The P < .05 was considered as statistically significant.
This study was registered in the Iranian Registry of Clinical Trials (IRCT registration code: IRCT2017020732449N1). Moreover, written informed consent forms were obtained from the neonates’ parents.
The protocol approval by the Ethics Committee of Kermanshah University of Medical Sciences (IR.KUMS.REC.1397.1035).
Results
In this study, 40 neonates with PDA were divided into 2 groups of 20 cases. One group, including 10 girls (50%) and 10 boys (50%), were treated with oral acetaminophen. The second group, which included 11 (55%) boys and 9 (45%) girls, were treated with rectal acetaminophen. Totally, 21 cases (52.5%) were boys and 19 cases (47.5%) were girls. Mean weight, gestational age and PDA diagnosis age were 1896.25 ± 2.73 g, 31.95 ± 2.679 weeks, and 3.42 ± 0.594 days, respectively. Most infants were in the gestational age range of 30 to 35 weeks. Regarding the type of delivery, most of the samples were through cesarean section (35 cases, 87.5%) and only 5 cases (12.5%) had normal delivery. The mean weight of neonates in the group treated with oral-acetaminophen was 1366.0 ± 206.51 and in the group treated with rectal acetaminophen was 1412.0 ± 196.91, which were not statistically significant different (Table 1).
Frequency distribution of treatment type in gender and type of delivery.
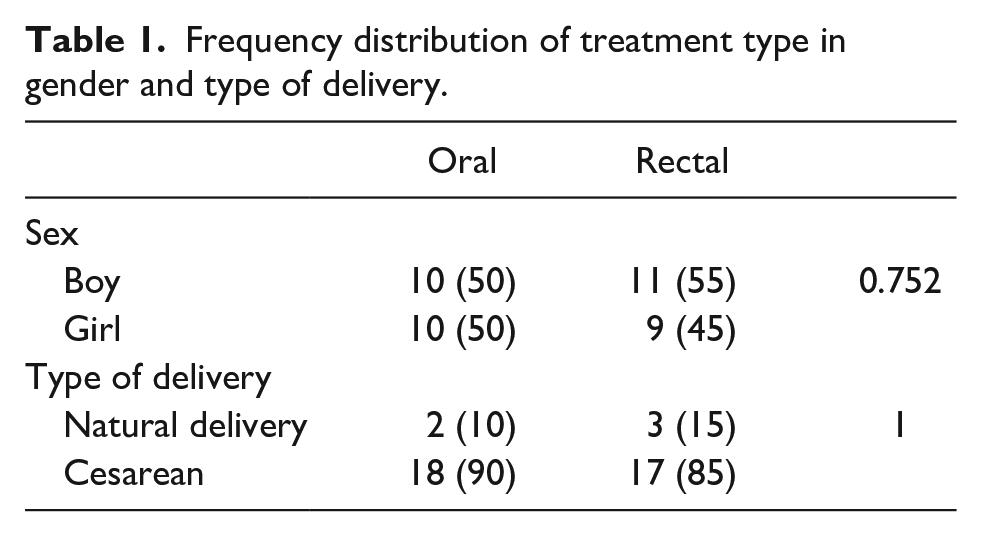
The age of diagnosis of PDA in the group treated with oral acetaminophen was 79.2500 ± 5.78451 hours and in the group treated with rectal acetaminophen was 77.6500 ± 5.00815 hours with no significant difference between them (P = .265).
The age of onset of PDA treatment in the group treated with the oral type was 81 ± 5.72161 hours and in the group treated with the rectal type was 79.4 ± 5.30541 hours, but no significant difference was observed between them (P = .365).
Comparison between the 2 groups showed that there were no significant difference between the plt, WBC, BUN, Cr, Bili Total, and Bili Direct with the type of treatment (Table 2).
Relationship between the desired variables and the type of treatment.

There was no difference between the PDA diameters in both groups, while there was the PDA diameter was lower in both groups after treatment compared to before treatment (Table 3).
Relationship between PDA inner diameter (mm) with type of treatment.

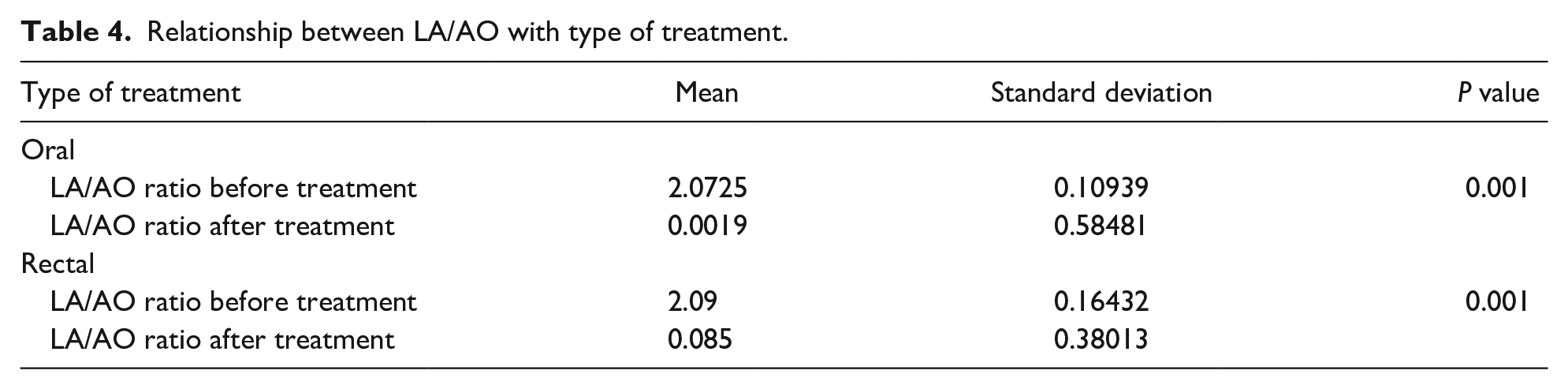
In both groups, the mean LA/AO (left atrial/aortic root) ratio was significantly lower after treatment than before treatment. There was no statistically significant difference between 2 treatment groups (Table 4).
Relationship between LA/AO with type of treatment.
Discussion
The DA in most term infants closes functionally on the a few days after birth, and the permanent duct opening is abnormal. 12 In about 30% to 60% of premature infants, the DA is open and only one third of infant lees than 1000 g will be closed spontaneously. Therefore, the vast majority of PDA infants are potential candidates for drug or surgical interventions. Although drugs such as indomethacin and ibuprofen are commonly used to duct close but they are also associated with side effects. 2
In 2011, Hammerman et al 6 first reported that oral administration of 15 mg/kg acetaminophen arterial closure in 5 PDA infants. Since then, most studies have used oral or intravenous acetaminophen as an alternative to arterial closure in PDA patients who have not responded to ibuprofen/indomethacin therapy.8,13 The mechanism of action of acetaminophen in arterial canal closure is still unknown. However, there are some researchers have reported that acetaminophen is effective in inhibiting prostaglandin synthase that may be related to the duct closure. 14
In the present study, acetaminophen was started at a dose of 25 mg/kg and then administered at a dose of 15 mg/kg every 8 hours for 3 days, of which 37 out of 40 infants were treated in the first stage. In a study by Asadpour et al 15 using a dose of 10 mg/kg acetaminophen at 6-hour intervals for 72 hours, in 23 of 25 infants (92%) achieved vascular duct closure. 15 Yurttutan et al 7 showed that oral dose of acetaminophen 15 mg per kg body weight every 6 hours for 3 days in preterm infants caused PDA closure in 4 out of 5 infants (80%). In a study in 2013 by Dang et al, 16 80 PDA infants with a gestational age of less than 34 weeks were treated with acetaminophen 15 mg/kg at 6-hour intervals for 3 days, in which the duct was closed in 81.2% cases. In the study by Akbari et al, 17 administration of acetaminophen at 15 mg/kg at 6-hour intervals for 48 hours resulted in vascular duct closure in 12 (75%) neonates while in 4 (25%) cases arterial ducts remained open. The low rate of arterial canal closure in the Akbari et al study compared to our study may be due to the shorter treatment period.
It has recently been reported that intravenous (IV) acetaminophen may be an alternative treatment in premature infants with malnutrition. 8 However, IV administration of acetaminophen is an invasive procedure. 18
In our study, in the group treated with rectal type of acetaminophen, 19 infants (95%) out of 20 infants the PDA was successfully closed. A number of studies have reported the effectiveness of the rectal method. In Van Lingen et al 19 study the rectal acetaminophen dose of 20 mg per kg body weight at intervals of 8 to 12 hours in preterm infants was effective in pain and fever. 19 A case series study of rectal acetaminophen was 95% effective in treating PDA. 10 Therefore, the use of rectal acetaminophen can be used to treat PDA cases.
In comparison between the therapeutic and rectal effects of acetaminophen, in the present study, the oral treatment of acetaminophen was not superior to the rectal type. Furthermore, the changes in DA diameter after treatment compared to before treatment in both groups had a decreasing trend. Therefore, there was no significant relationship between DA diameter and type of treatment. The results cannot be compared with other studies, because only in this study, a comparison was made between the oral therapy of acetaminophen and its rectal type.
In the present study, there was no significant relationship between the types of treatment with following parameters: gender, type of delivery, birth weight, gestational age, age of onset of PDA treatment, age of diagnosis of PDA, and treatment success. Similarly, in the study of Mehralizadeh et al 18 in evaluating the effectiveness of oral and IV acetaminophen in PDA closure, there was no significant difference in mean gestational age, age at diagnosis, weight, sex distribution, and frequency of PDA symptoms between the 2 groups.
In our study in both treatment groups, the mean LA/AO ratio decreased after treatment compared to before treatment. Both treatments were similar. In the study of Mehralizadeh et al, 18 it was reported that both IV and oral acetaminophen significantly reduce the mean LA/AO ratio.
The results of the present study showed that rectal acetaminophen had no therapeutic side effects. Oral and rectal use of acetaminophen had no effect on Platelets, Blood urea nitrogen, Creatinine, WBC, and bilirubin. Yang et al 20 showed that oral administration of ibuprofen and acetaminophen had no significant effect on platelets, serum creatinine and glutamic-pyruvic transaminases, and also no NEC was observed.
Conclusions
This study showed that there was no difference between PDA treatment of preterm infants with oral and rectal acetaminophen. Also, no side effects were observed in treatment with any of the treatments. It is suggested that in infants who are intolerant to oral acetaminophen, the rectal route can be replaced.
Footnotes
Author Contributions
MV designed the study. HD, HB, AA, RH, and RCL recorded the data of the patients. MR analyzed and interpreted the data. KGH have drafted the work. All authors read and approved the final manuscript
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
