Abstract
Objective. To summarize the characteristics and reliability of clinical practice guidelines (CPG) for diagnosing and managing common congenital heart diseases (CHD). Methods. We conducted a scoping review of CPG for CHD (ventricular septal defects, atrial septal defects, patent ductus arteriosus, and coarctation of aorta) published or updated from 2018 to April 2023. We searched in general search engines, CPG repositories, and webpages of organizations that elaborate CPG. To assess the reliability of CPG, we used the Appraisal of Guidelines Research and Evaluation II (AGREE-II) tool, with a score ≥70% considered acceptable quality. Results. Twenty-one CPG were identified. Only 6 (28.6%) conducted systematic reviews, and none achieved an acceptable AGREE-II score. “Scope and purpose” had the highest scores, while “stakeholder involvement” had the lowest. Conclusions. Most CPG for CHD lack systematic reviews and do not meet acceptable quality standards, underscoring the need for evidence-based guidelines to guide diagnosis and management effectively.
Keywords
Introduction
Congenital heart diseases (CHD) are cardiac malformations present at birth, exhibiting a wide range of severity. 1 Although significant advancements in diagnosis and treatment have been made in recent years, the prevalence of CHD remains at 1.8 per 100 live births, with notably higher mortality rates among infants aged one and under.2,3 Furthermore, from 1990 to 2017, the prevalence at birth of CHD cases increased by 4.2% and 18.7% in people living with CHD. 2 In low-income countries, significant challenges persist, including limited access to healthcare, diagnostic resources, and treatment options, which collectively contribute to a decreased quality of life for affected individuals.2,3 Previous studies mentioned that CHD with higher prevalence are ventricular septal defects (VSD), followed by atrial septal defects (ASD), patent ductus arteriosus (PDA), and coarctation of the aorta (CoA).4 -6 Given the prevalence of these CHD, the development of evidence-based clinical practice guidelines (CPG) is essential to ensure standardized and effective management approaches.
CPG are documents designed to assist in clinical decision-making. They are developed through systematic reviews, which serve as the basis for informed recommendations.4,7,8 These CPG have the potential to provide recommendations that can improve the quality of life and mitigate potential harm of CHD management approaches.
Scoping reviews of CPG are crucial for evaluating their characteristics, deficiencies, and gaps within these documents. 9 This process is essential for gaining a deeper insight into the variations in patient management and for suggesting potential improvements in how these guidelines are conceived or performed. Previous studies have focused on evaluating CPG for specific cardiology topics, including stroke and cardiac surgery10,11; however, no study has undertaken an assessment of guidelines concerning CHD.
Thus, we aimed to summarize the characteristics and reliability of CPG for the diagnosis, management, and prognosis of CHD.
Material and Methods
Design and Eligibility Criteria
We conducted a scoping review to identify CPG focused on CHD. Our literature search included CPG meeting the following inclusion criteria: (1) released or updated in the last 5 years (from 2018 to April 2023); (2) which provided recognizable recommendations (regarding the diagnosis, management, or prognosis) based on evidence; and (3) the four most-prevalent CHD: VSD, ASD, PDA or CoA. Our review had no language restrictions.
We excluded publications that exclusively addressed very specific cases (such as postoperative pain management), and those that did not include any information regarding the methodology used to reach their recommendations.
Information Sources
To identify potentially relevant CPG, we conducted a comprehensive search of electronic databases (PubMed, Embase, Web of Science, Scopus, Trip Database, Epistemonikos, Google, and Google Scholar) using predefined search terms and Medical Subject Headings (MeSH) related to “Practice Guidelines,” “Heart Defects, Congenital” and the specific conditions (VSD, ASD, PDA, CoA). Additionally, we searched in guidelines repositories (Canadian Medical Association Infobase, Guidelines International Network, National Guideline Clearinghouse, National Health and Medical Research Council, National Institute for Health and Care Excellence—UK, Scottish Intercollegate Guidelines Network, American College of Physicians Clinical Practice Guidelines, Registered Nurses’ Association of Ontario, New Zealand Guidelines Group). Also, the webpages of cardiology associations (European Society of Cardiology, American College of Cardiology, American Heart Association), and the webpages of other institutions that develop CPG (Centro Nacional de Excelencia Tecnológica en Salud—México, Guías de Práctica Clínica en el Sistema Nacional de Salud de España, IETS Colombia, IETSI Perú, Repositorio del Instituto Nacional de Salud del Niño San Borja—Perú). The search strategy for each of the consulted sources is detailed in Supplemental Table 1.
Selection of CPG and Data Extraction
We performed comprehensive searches within each source. We reviewed titles and abstracts, and where necessary, delved into the complete texts of the documents to ensure the inclusion of CPG meeting our predefined inclusion criteria.
Following a consensus, we designed a data-charting form to outline the specific data points for extraction. The data extracted encompassed a wide range of information, including document characteristics (such as title, country of origin, and the year of the most recent update), the age of the target population, the CHD addressed, the methodology employed, and the funding. Our aim was to replicate the experience of a typical CPG user, so we refrained from making additional efforts to acquire these variables if they were not already provided in the CPG.
Two reviewers (ORT and FACE) performed independently selection and extraction. In instances of any discordance or disagreement, a third author (ATR) was consulted. Any such disparities were diligently addressed and resolved through a consensus-based approach.
Critical Appraisal of Individual Sources of Evidence
We employed the Appraisal of Guidelines Research and Evaluation II (AGREE-II) instrument to conduct a comprehensive critical appraisal of the CPG that reported performing systematic reviews to inform their recommendations, since this represents a fundamental quality standard in the development of CPG. 12
The AGREE-II instrument assesses the methodological rigor and transparency in the development of guidelines across six key domains: (1) Scope and purpose; (2) Stakeholder involvement; (3) Rigor of development; (4) Clarity of presentation; (5) Applicability; and (6) Editorial independence. 11 To ensure a rigorous evaluation process, two authors (ORT and FACE) independently appraised the guidelines using a predefined assessment form. In cases where discrepancies emerged, a third author (ATR) was consulted.
We established that a CPG must attain a minimum total score of 70% to be considered of acceptable quality. This threshold aligns with prior studies that evaluated the quality of CPG.13,14 We consistently applied this same criterion to evaluate the quality of individual domains within the AGREE-II Instrument.
Synthesis of Results
We conducted descriptive analyses, presenting absolute and relative frequencies as well as means or medians when appropriate. All analyses were performed using the Microsoft Excel software (Microsoft 365).
Results
Selection of Sources of Evidence
The search strategy initially identified 4917 documents, of which 30 underwent a full-text assessment for eligibility, and ultimately 21 CPG related to CHD were included (Figure 1). A detailed overview of the search strategy can be found in Supplemental Table 1. Furthermore, the list of reports assessed in full-text and finally excluded can be located in Supplemental Table 2.

Flowchart of the selection of CPG.
Characteristics of CPG
The characteristics of the 21 included CPG are presented in Table 1. Among these, 8/21 were published between 2022 and 2023. The countries with the highest representation in the CPG were the USA, Australia and United Kingdom, with 4/21 each. A significant majority of the included CPG (14/21) focused on a population under 18 years of age, while 7/21 did not specify their target population (Tables 1 and 2).
Characteristics of the Included CPG.

Abbreviations: CPG, clinical practice guideline; CHD, congenital heart disease; CCHD, critical congenital heart disease; NR, not reported; VSD, ventricular septal defect; PDA, patent ductus arteriosus; CoA, coarctation of the aorta; AHA/AAC, American Heart Association/American Academy of Cardiology.
While these CPG reference the utilization of the GRADE methodology, none of them present Summary of Findings (SoF) tables.
Characteristics of Clinical Practice Guidelines on the Most Common Congenital Heart Diseases (n = 21).
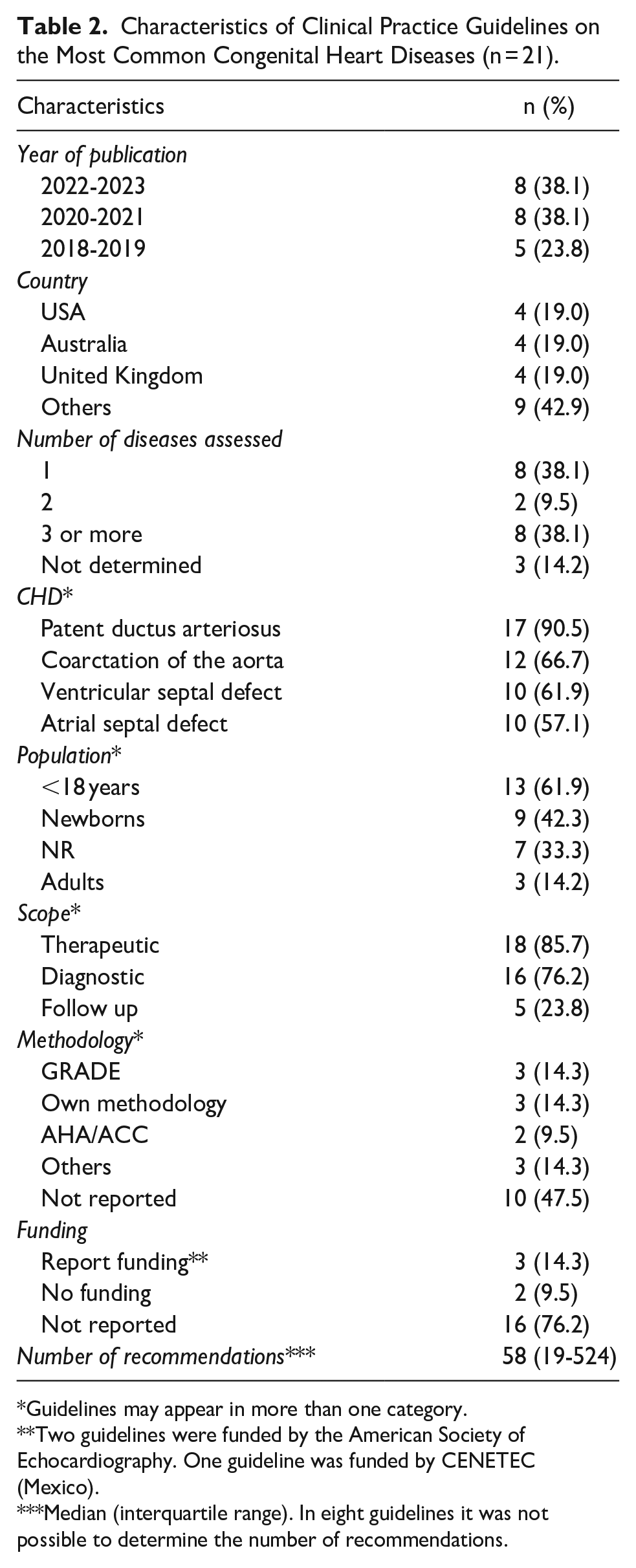
Guidelines may appear in more than one category.
Two guidelines were funded by the American Society of Echocardiography. One guideline was funded by CENETEC (Mexico).
Median (interquartile range). In eight guidelines it was not possible to determine the number of recommendations.
The majority of CPG (11/21) addressed multiple CHD. The most frequently addressed conditions among were PDA (19/21), CoA (14/21), VSD (13/21), and ASD (12/21). Regarding the methodologies used to formulate recommendations, the most commonly employed methodology was Grading of Recommendations Assessment, Development and Evaluation (GRADE) (3/21), although they reported the use of this methodology, any of them used a summary of findings (SoF) table and only one of them used an Evidence to Decision (ETD) table; however, 10/21 CPG did not report the specific methodology used (Tables 1 and 2).
CPG That Performed Systematic Reviews to Reach Their Recommendations
Of the included CPG, six performed systematic reviews to reach their recommendations: the CPG for CHD by the European Society of Cardiology (ESC 2020), 1 the CPG for CHD by the American College of Cardiology and American Heart Association (ACC/AHA 2018), 15 the CPG for CoA by the ACC/AHA (ACC/AHA CoA 2022), 16 the CPG for CHD by the Canadian Cardiovascular Society (Canada 2022), 17 the CPG for PDA by the Canadian Paediatric Society (CPS PCA 2022), 18 and the CPG for VSD and ASD by the National System of Health of Mexico (Mexico 2018) 19 (Table 1).
These six CPG reported the methodology used to reach their recommendations: three used the GRADE methodology (Canada 2020, CPS PCA 2022, and Mexico 2018),17 -19 although any of them displayed SoF tables. The CPG made by AHA/ACC 2018 and AHA/ACC CoA 2022 described its own methodology in a separate document15,16; similarly, ESC 2020 1 described its own methodology in another document.
The six CPG that performed systematic reviews to inform their recommendations were assessed using the AGREE-II (Table 3 and Supplemental Table 3). None of these guidelines achieved an overall score ≥ 70% required for considering them of acceptable quality (median: 56%, range: 45%-69%). The domain of “scope and purpose” received the higher score (median: 75%, range: 53%-89%). In contrast, the lower scores were obtained in the domains of “stakeholder involvement” (median: 45%, range: 22%-53%) and “applicability” (median: 43%, range: 33%-50%).
AGREE-II Assessment of the Included Guidelines That Have Performed Systematic Reviews to Inform Their Recommendations.

Domain 1 = scope and purpose; Domain 2 = stakeholder involvement; Domain 3 = rigor of development; Domain 4 = clarity of presentation; Domain 5 = applicability; and Domain 6 = editorial independence.
Discussion
Main Findings
We identified 21 CPG relevant to CHD, published or updated between 2018 and April 2023. The most frequently addressed CHD in these CPG were PDA and CoA. The methodologies employed varied; while some adhered to standardized approaches like GRADE or the AHA/ACC methodology, approximately half of the CPG lacked information regarding the methodology used. The United States, Australia, and the UK reported the highest numbers of CPG. Among the included CPG, only six reported the conduct of systematic reviews, none of which achieved an AMSTAR-II score ≥70% or displayed SoF tables.
Other authors have emphasized the difficulty of conducting RCT due to the heterogeneity present in the population of patients with congenital heart disease, which can lead to little evidence being generated and therefore the CPG not having evidence to issue solid recommendations and which depend on the consensus of experts. 20
Type of CHD
The most common CHD considered in the CPG were PDA and CoA, despite not being the most frequent CHD in the general population, with approximate frequencies of 8% to 11% and 3.2% to 3.8% respectively.21,22 Conversely, VSD, one of the most frequent CHD with an approximate frequency of 33% to 37%, 2 was less frequently addressed in the CPG. Additionally, a large majority of the occasions in which a CPG assessed PDA it did not include other CHD, while CPG that evaluated ASD or VSD often encompassed other CHD.
This pattern can be explained by the sense of urgent necessity for recommendations on interventions in conditions like PDA, which require prompt treatment due to the heightened risks they pose, such as the development of bronchopulmonary dysplasia, 23 while CHD like VSD and ASD can exhibit spontaneous closure of the congenital defect. 24 In cases of ASD and VSD that do not receive treatment or present spontaneous closure, depending on the size of the defect, they may begin to present very mild symptoms from approximately the age of 30 and may even develop Eisenmenger syndrome. 25
Population and Age
Most of the CPG in our study are focused on individuals younger than 18 years of age. However, as an increasing number of cases demonstrate improved survival rates for certain CHD and some are diagnosed in adulthood, 2 a current challenge emerges in the long-term management of these patients. Furthermore, addressing the diagnosis and treatment of adult patients with CHD, along with their associated complications, is a critical aspect that encounters obstacles due to the limited availability of evidence and experts.4,7,26 Hence, it is imperative for CPG to adopt a rational methodology for dealing with the uncertainty of evidence and the differing opinions among experts.
Quality of CPG
In our study, none of the CPG that employed systematic reviews were able to attain the AGREE-II threshold of ≥70% to be designated as an acceptable quality CPG. While the AGREE-II domain with the higher score was “scope and purpose,” the ones with the lower scores were “stakeholder involvement” and “applicability.” This may be because patient groups with these conditions are not always easily accessible, and this can still be stated in the CPG, however, details about the CPG developers, as well as details of the external reviewers, should still be included. Moreover, the “Rigour of development” domain had a mean score of 56% (from 40% to 71%), signaling relevant shortcomings in the methodological aspect of the guidelines. This is closely related to the fact that only 29% of the CPG had performed systematic reviews and around half of the CPG did not report the methodological framework used to reach their recommendations.
Financial support details were provided in just 5/21 of the included CPG. The disclosure of funding is significant as it plays a crucial role in promoting transparency. This transparency, in turn, facilitates the evaluation of potential biases that could impact the formulation of CPG and their recommendations. Ultimately, the explicit reporting of funding sources enhances the credibility and reliability of the guidelines. One explanation for the lack of details on the financial support may be that it was self-funded and therefore not declared, however, even if that was the case, it should still be declared that it was self-funded.
Furthermore, the precision of recommendations from specific guidelines, including those from the AHA and ESC, has faced scrutiny due to concerns about their methodology. The formulation of recommendations in some instances lacks clarity, and there have been instances where recommendations based on weak evidence leaded to strong recommendations.27,28 This raises questions about the reliability and robustness of the guideline development process. Moreover, it’s worth noting that even guidelines explicitly claiming to employ the GRADE methodology may fall short in transparency by not providing essential components such as SoF tables.29,30
Our study is not without limitations. Despite conducting a comprehensive review across multiple search engines (including Google and Google Scholar), it is conceivable that certain CPG published exclusively on the websites of their creators, such as medical societies or government pages, might have eluded our search efforts. Furthermore, even though numerous CPG lacked essential information concerning their methodology, target audience, or funding sources, we refrained from making additional efforts to gather these variables. This decision aligns with our goal of mirroring the experience of an ordinary CPG user.
Conclusions
We identified a total of 21 CPG. Methodology was not reported in near half of these CPG. Only six CPG performed systematic reviews to inform their recommendations, none of which reached the cut-off point of an overall score ≥70% in the AGREE-II tool, to be considered as of acceptable quality. The domain with the highest score was scope and purpose, while the ones with the lowest score were stakeholder involvement and applicability. For the development of future clinical practice guidelines (CPG), we recommend providing detailed information on the CPG developers (including their names, specialties, and institutions), the intended target users of the guidelines, and the sources of funding (or specifying the absence thereof). Additionally, we suggest including a summary of the recommendations along with a flowchart to facilitate the practical implementation of these guidelines. Finally, we recommend that future guidelines be based on systematic reviews of the literature and use appropriate decision-making methodologies, such as GRADE. 31
Supplemental Material
sj-docx-1-gph-10.1177_30502225251315623 – Supplemental material for Clinical Practice Guidelines for Common Congenital Heart Diseases: A Scoping Review
Supplemental material, sj-docx-1-gph-10.1177_30502225251315623 for Clinical Practice Guidelines for Common Congenital Heart Diseases: A Scoping Review by Oscar Rivera-Torrejón, Fabian A. Chavez-Ecos and Alvaro Taype-Rondan in Sage Open Pediatrics
Footnotes
Authors’ Note
Fabian A. Chavez-Ecos is also affiliated to Red de Cardiología y Salud Pública (RCSP), Ica, Peru.
Author Contributions
All the authors contributed to the conception and design of the study. ORT and FACE acquire, analyze, interpretated the data and wrote the initial draft of the manuscript. ATR offered critical revisions. ORT and FACE finalized the manuscript. All the authors reviewed and approved the final version of the manuscript.
Data Availability Statement
All relevant data of this scoping review are reported in the document.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
There are no human participants in this article and informed consent is not required.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
