Abstract
The diagnostic and treatment outcomes of intracranial germ cell tumors (ICGCTs) among low and middle income countries are limited. A total of 63 ICGCTs patients with a median age of 11.6 years were studied. A 30 (47.6%) and 33 (52.4%) patients were classified as pure germinomas and nongerminomatous germ cell tumors (NGGCTs), respectively. The concordances between serum and cerebrospinal fluid (CSF) alpha-fetoprotein (84.3%) and beta-human chorionic gonadotropin (68.4%) were addressed. The 5-year overall survival (OS) and event-free survival (EFS) rates of pure germinomas versus NGGCTs were 83.9 versus 69.1% and 74.6 versus 57.7%, respectively. Patients undergoing radiation had higher 5-year OS and EFS than those without radiation treatment with P < .001. Chemotherapy combined with radiation is a cornerstone treatment to achieve outcomes. Adverse prognostic factors included age <8 years, surgery, and nonradiation treatment. Either serum or CSF tumor markers were adequately required as a diagnostic test among patients with ICGCTs.
Keywords
Introduction
Primary central nervous system (CNS) tumors are the most common malignant solid tumors among children. 1 The World Health Organization classified CNS tumors, based on histology, immunohistochemistry, and molecular diagnosis of tumors. 2 Pediatric intracranial germ cell tumors (ICGCTs) were addressed with high incidence in the Asian population and constitute the third most common primary CNS tumors in Thailand. 3 The age-standardized incidence rate of ICGCTs ranged from 0.45 to 1.1 per million population, with the peak aged between 10 and 15 years3,4 in the Thai population.
Based on the heterogenicity of disease characteristics, histology, and the degree of tumor cells differentiation, ICGCTs are classified as pure germinoma or nonsecreting ICGCTs and non-germinomatous germ cell tumors (NGGCTs) or secreting ICGCTs.5,6 Evaluating tumor marker values of alpha-fetoprotein (AFP) and beta-human chorionic gonadotropin (β-HCG) in serum and/or cerebrospinal fluid (CSF) are essential to ensure the definite diagnosis of NGGCTs when combined with radiological diagnosis,6,7 especially when tissue diagnosis is limited due to the inoperability or limited surgical interventions in resource-constrained settings. However, less information is available regarding the validity, accuracy, and consistency between serum and CSF tumor marker values among patients with ICGCTs.
Multimodality treatment protocols, including platinum-based chemotherapy regimens and radiotherapy, are the backbone to achieving desirable outcomes in ICGCTs.5,7 Various strategies have been devised depending on the classification of ICGCTs.6,8 The Thai Pediatric Oncology Group (ThaiPOG) treatment protocols for ICGCTs (ThaiPOG-BT-13GCT for CNS pure germinomas and ThaiPOG-BT-13NGCT for CNS NGGCTs), which were officially established in 2013, included carboplatin-based regimens adapted from the Children’s Oncology Group studies. 9 These protocols were primarily used to treat pediatric patients with ICGCTs in Thailand since 2000 with effectiveness and lower ototoxicity over cisplatin-based regimens. 10
The present study aimed to describe the clinical characteristics, prognostic factors, and outcomes and determine the diagnostic accuracy, consistency, and tumor marker value distribution among pediatric ICGCTs at 3 oncology centers in Thailand. The results provide comprehensive information on children with ICGCTs in Thailand. Herein, this study might initiate management strategies in pediatric ICGCTs across Thailand and could be extended to Asia-Pacific neighbors where resources remain limited.
Methods
Patient Selection
The medical records of 63 pediatric patients with newly diagnosed ICGCTs and treated primarily at 3 tertiary pediatric oncology centers in Thailand between January 1, 2007 and December 31, 2019 were retrospectively reviewed. These centers were Chiang Mai University Hospital, Chiang Mai, Phramongkutklao Hospital and King Chulalongkorn Memorial Hospital, Bangkok, Thailand. The study follow-up endpoint was December 31, 2021.
The inclusion criteria included patients with ICGCTs aged <18 years whose diagnosis of ICGCTs was confirmed by histopathology or consistent radiological diagnosis combined with elevation of AFP and/or β-HCG in serum and/or CSF. Patients with uncertain diagnoses were excluded from the study.
Written informed consent and assent were waived. This study was approved by the Ethics Committee and Institutional Review Board of Phramongkutklao Hospital and Phramongkutklao College of Medicine, Bangkok, Thailand (reference number: IRBRTA 045/2562), and other institutional research ethics committees from 2 institutions following the ethics principles of the Declaration of Helsinki (1975) and its revision.
Clinical Definitions
Histological diagnoses of NGGCTs comprised endodermal sinus tumor (yolk sac tumor), choriocarcinoma, immature teratoma, mature teratoma with malignant transformation, and mixed germ cell tumors. Patients without histologic confirmation were eligible for the study if serum and/or CSF β-HCG was >50 IU/L, or AFP was >10 IU/L 11 with a confirmed radiological diagnosis.
Pure germinoma is defined by histopathology confirmation of germinoma and serum and/or CSF β-HCG of ≤50 IU/L, and AFP of ≤10 IU/L.
Metastatic definitions are defined as: M0; no metastasis, M1; the presence of abnormal cell(s) from CSF cytology, M2; brain metastasis identified by magnetic resonance imaging (MRI), and M3; spinal cord metastasis identified by MRI.
Treatment
Surgery
The surgical approach was indicated using the surgical experience in the individual institutions with any surgical extent or ventriculoperitoneal (VP) shunt for correcting hydrocephalus, before or after completing chemotherapy. CSF studies for tumor marker levels and cytology were collected from ventriculostomy, VP shunt, or lumbar puncture before chemotherapy and radiotherapy procedures.
Chemotherapy regimens
ThaiPOG treatment protocols for ICGCTs including ThaiPOG-BT-13GCT for CNS pure germinomas (Supplemental Table 1) and ThaiPOG-BT-13NGCT for CNS NGGCTs (Supplemental Table 2) were initiated and primarily used in some oncologic centers between 2000 and 2012. Thereafter, these protocols have been officially used as national protocols for children with ICGCTs since 2013.
Radiation
After establishing ThaiPOG treatment protocols for ICGCTs in 2013, radiation was added to the treatment protocols for all children with ICGCTs.
Radiotherapy was initiated within 28 days after the initial 3 cycles of chemotherapy. Radiotherapies for pure germinomas M0 and M1 to 3 were whole ventricular irradiation (WVI) and craniospinal irradiation (CSI) 21 to 24 Gy, respectively. For patients having complete remission (CR) after 3 cycles of chemotherapy, the primary tumor bed would be boosted to 30.6 to 36 Gy while those without CR would be boosted to 40 to 45 Gy.
Radiotherapies for NGGCTs M0 and M1 to M3 were WVI 24 to 36 Gy and CSI 36 Gy, respectively. The primary tumor bed would subsequently be boosted to 54 Gy.
Nonetheless, between 2000 and 2012, radiation treatment for M0 pure germinomas and M0 NGGCTs might be considered whole-brain irradiation and CSI, respectively, instead of WVI, depending on institutional radiotherapy accessibility.
Outcome definitions
CR was characterized as no evidence of the primary tumor evaluated by computed tomography or MRI of the brain.
Overall survival (OS) was defined as the time interval from the date of diagnosis either to the time of death resulting from any causes or to the last follow-up for patients who survived.
Event-free survival (EFS) was defined as the time duration between the date of diagnosis and disease reactivation, progression, or death, whatever came first or the last follow-up for patients without events.
Statistical analysis
Overall demographic data, laboratory results and treatment from all participating patients were analyzed using descriptive statistics, presented as mean with standard deviation (normal distribution) or median with range (nonnormal distribution) for continuous variables, and calculated using frequency and percentage for categorical variables. Categorical and continuous variables were compared using Fisher’s exact and Mann-Whitney U tests, respectively.
Serum and CSF AFP and β-HCG levels were analyzed using Cohen’s Kappa Coefficient to determine the consistency and percent agreement.
The survival function was calculated using the Kaplan-Meier method and compared using Cox’s Proportional Hazard Model to evaluate the effect of covariates on hazard ratio (HR). Univariate and multivariate analyses were performed to analyze the impact of possible factors on defined outcomes. Statistical and survival analyses were performed using STATA/IC Software, Version 17.0 (Stata Corp, College Station, TX, USA), and P < .05 was considered statistically significant.
Results
Patient Characteristics
Among the 63 patients with ICGCTs enrolled in this study, patient characteristics including age, sex, primary sites, clinical manifestations, laboratories, and treatment, compared between pure germinomas and NGGCTs are summarized in Table 1. The patients’ age ranged from birth to 16 years with a median age of 11.6 years. The male to female ratio was 1.5:1. A 30 (47.6%) and 33 (52.4%) patients were classified as pure germinomas and NGGCTs, respectively.
Patient Demographic Data (n = 63).

Data are presented as mean ± SD and median (range) for continuous variables and number (%) for categorical variables. Comparison between 2 independent data sets was analyzed using Fisher’s exact or Mann-Whitney U test (age at diagnosis, serum, and CSF AFP and β-HCG, and surgical extent). Statistical significance was considered at P < .05.
Abbreviations: AFP, alpha-fetoprotein; β-HCG, beta-human chorionic gonadotropin; CSF, cerebrospinal fluid; NGGCTs, nongerminomatous germ cell tumors; SD, standard deviation.
The most common location of ICGCTs was the pineal gland (n = 20, 31.7%). Visual disturbance, headache, and weakness were the common presentations, in rank. More patients with NGGCTs had increased intracranial pressure (IICP) than patients with pure germinoma (P = .009). A 19 (30.2%) patients had metastatic disease at diagnosis. All pure germinomas and 29 (87.9%) NGGCTs patients had tissue diagnoses. All patients with ICGCTs were evaluated for serum AFP and β-HCG, while 45 (71.4%) patients had CSF tumor marker values.
Surgical resection was performed more in patients with NGGCTs (n = 19, 57.6%) than in patients with pure germinoma (n = 7, 23.3%; P = .006) with a median surgical extent of 80%. Radiotherapy was administered among 54 (85.7%) patients.
Diagnostic Consistency and Agreement Between Serum and CSF Tumor Marker Values Among Patients With ICGCTs
In all, 45 patients with ICGCTs had obtained both serum and CSF AFP and β-HCG values. The percent agreement between serum and CSF tumor markers among pediatric patients with ICGCTs is shown in Table 2. AFP and β-HCG showed a moderate level of agreement (72% agreement; κ = .76% and 59.7% agreement; κ = .61, respectively) with P < .001.
Percent Agreement Between Serum and CSF Tumor Markers Among Pediatric Patients With ICGCTs (n = 45).
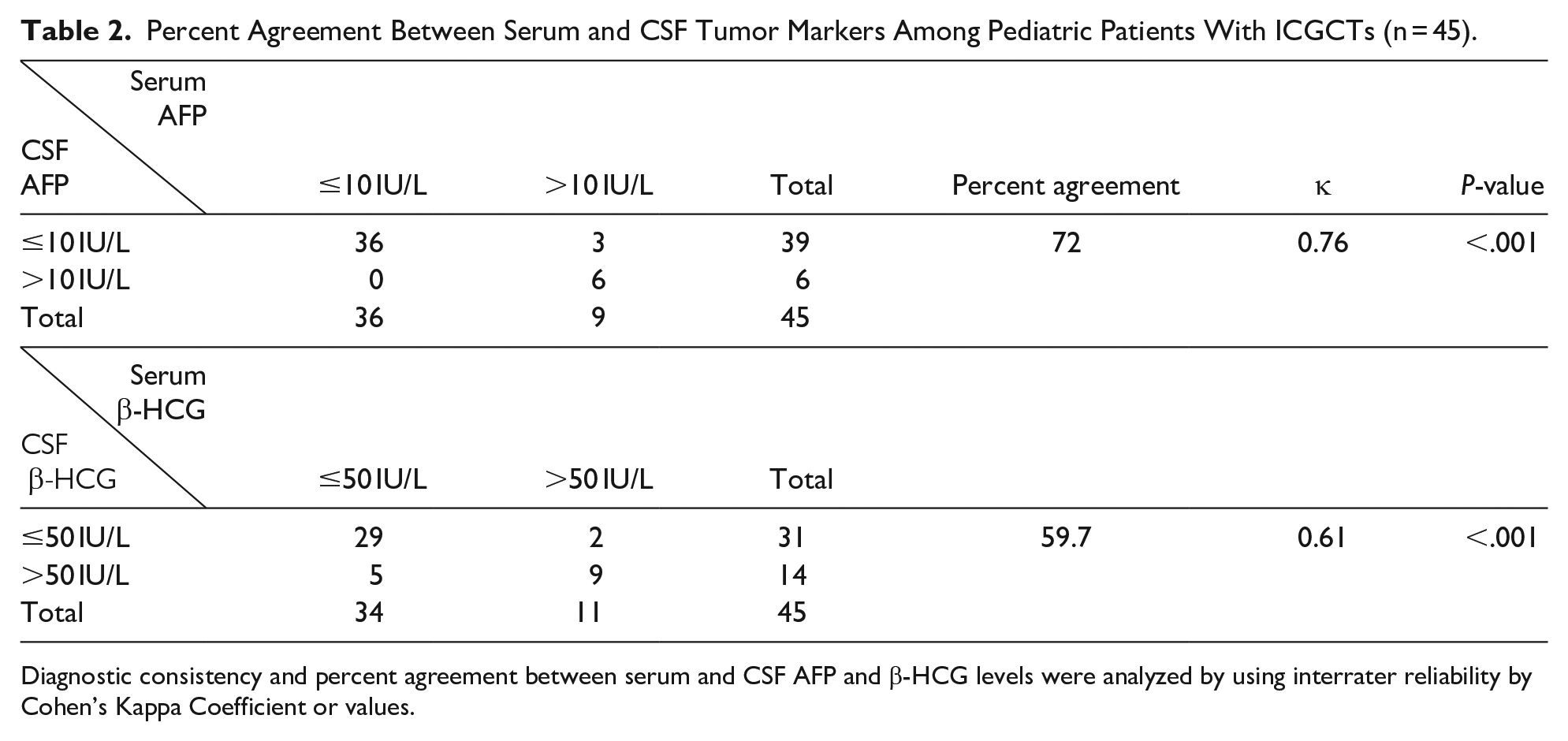
Diagnostic consistency and percent agreement between serum and CSF AFP and β-HCG levels were analyzed by using interrater reliability by Cohen’s Kappa Coefficient or values.
Abbreviations: AFP, alpha-fetoprotein; β-HCG, beta-human chorionic gonadotropin; CSF, cerebrospinal fluid; ICGCTs, intracranial germ cell tumors; κ, Cohen’s Kappa values.
Distribution of Serum and CSF Tumor Marker Value Categories in ICGCTs Diagnosis
The distribution between normal and abnormal serum and CSF AFP and β-HCG to diagnose pure germinomas and NGGCTs was calculated (Table 3).
Distribution of Patients With ICGCTs According to Tumor Marker Value Categories (n = 39).

Normal serum or CSF AFP is defined as serum or CSF AFP ≤10 IU/L. Abnormal serum or CSF AFP is defined as serum or CSF AFP >10 IU/L. Normal serum or CSF β-HCG is defined as serum or CSF β-HCG ≤50 IU/L. Abnormal serum or CSF β-HCG is defined as serum or CSF β-HCG >50 IU/L.
Abbreviations: AFP, alpha-fetoprotein; β-HCG, beta-human chorionic gonadotropin; CSF, cerebrospinal fluid; ICGCTs, intracranial germ cell tumors; NGGCTs, nongerminomatous germ cell tumors.
In total, 39 (62%) patients had obtained both serum and CSF tumor markers and histopathology diagnoses. For patients with pure germinoma, all (n = 20, 100%) patients had normal serum and CSF AFP and β-HCG. However, in the NGGCTs, 4 (21.1%) and 8 (42.1%) patients had abnormal both serum and CSF AFP and β-HCG, respectively. However, 12 (63.2%) and 5 (26.3%) patients had normal both serum and CSF AFP and β-HCG, respectively. The concordance between serum and CSF (normal serum and CSF or abnormal serum and CSF tumor marker levels) AFP (n = 16, 84.3%) and β-HCG (n = 13, 68.4%) were also addressed.
Overall Treatment Outcomes
Disease relapse or progression occurred in 13 (20.6%) patients, with a median relapse time of 1.6 years from diagnosis (range, 0.03-4.18 years). At the end of the study, 14 (22.2%) patients died, 7 (50%) patients died from disease relapse, 5 (35.7%) patients died from treatment-related infection and 2 (14.3%) patients died from respiratory failure, which was unrelated to treatment complications, with a median time to death of 1 year from diagnosis (range, 0.09-4.46 years). The median follow-up time was 4.08 years (range, 0.08-12.92 years).
Outcomes Between Pure Germinomas Versus NGGCTs
The 5-year OS of patients with pure germinoma and NGGCTs were 83.9% (95% CI, 61.5-93.9) and 69.1% (95% CI, 50.2-82.1; P = .128, HR, 2.46; 95% CI, 0.771-7.849; Figure 1A), and their 5-year EFS were 74.6% (95% CI, 53.7-87.1) and 57.7% (95% CI, 37.9-73.2; P = .213, HR, 1.794; 95% CI, 0.716-4.499; Figure 1B), respectively.

Overall survival (A) and event-free survival (B) between germinomas and NGGCTs. Overall survival (C) and event-free survival (D) according to age at diagnosis among pediatric patients with intracranial germ cell tumors.
Age at Diagnosis Impacting Outcomes
The 5-year OS of patients with ICGCTs according to age <8 and ≥8 years were 54.2% (95% CI, 20.4-78.9) versus 81.2% (95% CI, 66.9-89.8; P = .128, HR, 2.339; 95% CI, 0.783-6.986; Figure 1C), and their 5-year EFS were 53.9% (95% CI, 24.8-76) versus 68.9% (95% CI, 52.9-80.5; P = .172, HR, 1.951; 95% CI, 0.748-5.087; Figure 1D), respectively. On subsequent multivariate analysis, age <8 years at diagnosis became an additional independent risk factor significantly associated with death (P = .036, adjusted HR, 4.475; 95% CI, 1.103-18.157; Table 4).
Risk Factors Associated With Outcomes Among Pediatric Patients With ICGCTs (n = 63).
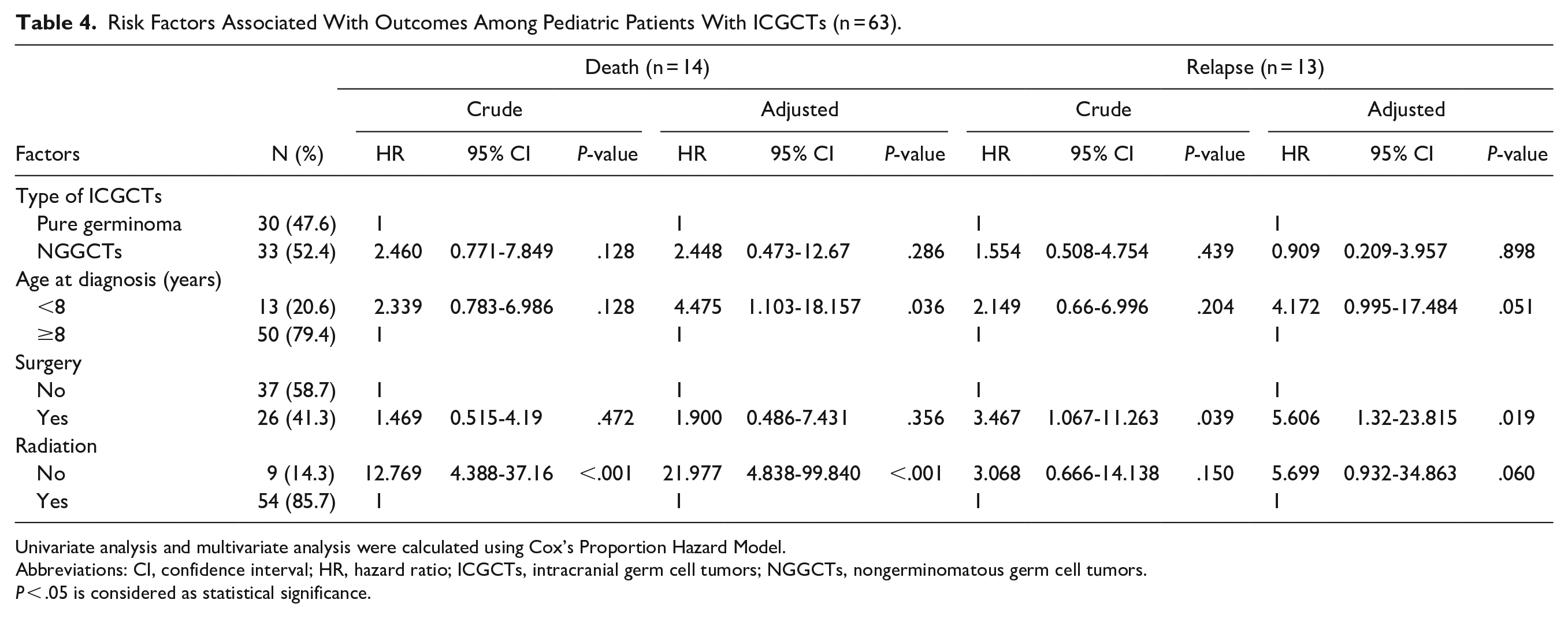
Univariate analysis and multivariate analysis were calculated using Cox’s Proportion Hazard Model.
Abbreviations: CI, confidence interval; HR, hazard ratio; ICGCTs, intracranial germ cell tumors; NGGCTs, nongerminomatous germ cell tumors.
P < .05 is considered as statistical significance.
Treatment Modalities Affecting Outcomes
The 5-year OS of patients with ICGCTs who had and had not undergone surgery were 69.2% (95% CI, 45.1-84.3) versus 80.7% (95% CI, 63.8-90.3; P = .472, HR, 1.469; 95% CI, 0.515-4.19; Figure 2A), and their 5-year EFS were 54.3% (95% CI, 32.4-71.8) versus 74.3% (95% CI, 56.2-85.8; P = .164, HR, 1.87; 95% CI, 0.775-4.513; Figure 2B), respectively. However, surgery was a significant risk factor related to relapse (P = .039) on univariate analysis. On subsequent multivariate analysis, surgery remained an independent risk factor significantly associated with relapse (P = .019, adjusted HR, 5.606; 95% CI, 1.32-23.815; Table 4).
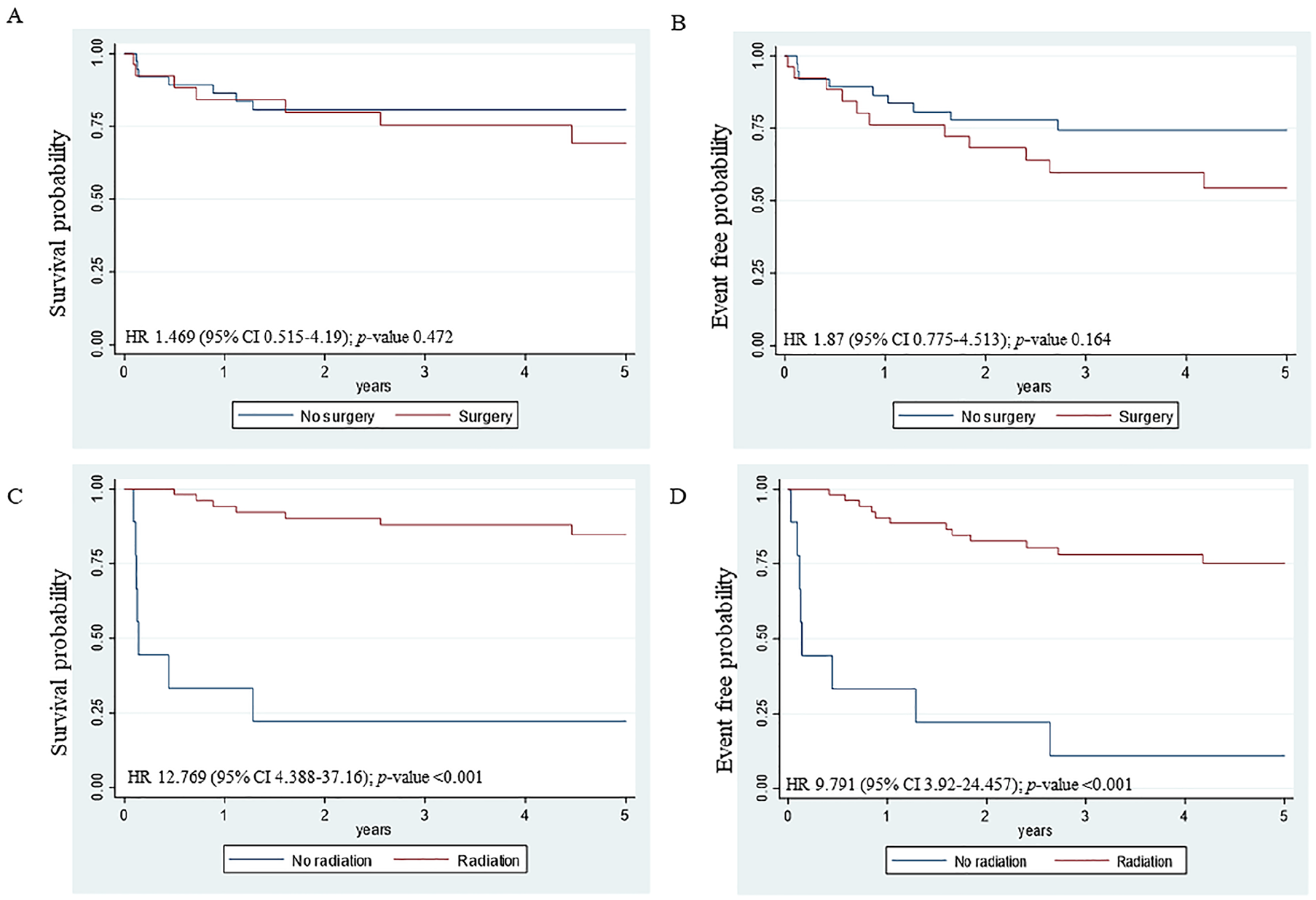
Overall survival and event-free survival according to treatment modalities among pediatric patients with intracranial germ cell tumors. Overall survival (A) and event-free survival (B) according to surgery.
Regarding radiotherapy, the 5-year OS of patients with ICGCTs had received versus had not received irradiation were 84.7% (95% CI, 70-92.6) versus 22.2% (95% CI, 3.4-51.3; P < .001, HR, 12.769; 95% CI, 4.388-37.16; Figure 2C), and their 5-year EFS were 75.2% (95% CI, 60.1-85.2) versus 11.1% (95% CI, 0.6-38.8; P < .001, HR, 9.791; 95% CI, 3.92-24.457; Figure 2D), respectively. On subsequent multivariate analysis, treatment without radiation remained an independent risk factor significantly related to survival (P < .001, adjusted HR, 21.977; 95% CI, 4.838-99.84; Table 4).
Discussion
Asian populations have a high prevalence of ICGCTs which are more predominantly diagnosed among male adolescents, similar to related reports.12,13 The clinical presentations of ICGCTs vary depending on tumor size and its locations. 5 ICGCTs are typically located in the midline and the most common locations are the pineal and suprasellar regions,12,14 which were also found in our study. The co-occurrence of tumors in both pineal and suprasellar regions, called bifocal tumors, is frequently found in pure germinomas.5,7 However, pineal gland and multifocal lesions were addressed as the common locations in our study, in rank, which were similar to the data from Japan. 13
Pineal ICGCTs commonly present with IICP with or without Parinaud syndrome, whereas endocrinopathies from hypothalamic/pituitary axis dysfunctions are common in suprasellar ICGCTs. 5 In our study, visual disturbance and headache were the common initial presentations observed in approximately half of the patients. IICP was more prevalent in NGGCTs because of the higher incidence of pineal tumors, likewise addressed in Germany. 15 The compression and obstruction of the cerebral aqueduct from pineal tumors cause obstructive hydrocephalus and IICP. 16 Diabetes insipidus (DI) was constituted in almost 30% of patients, which resembled related reports,17-19 of which DI determined the neurohypophyseal dysfunction from suprasellar invasion. 16
Pure germinoma is more prevalent rather than NGGCTs in the United States, 20 Canada, 14 , Japan, 12 and Korea. 21 Interestingly, the prevalence of pure germinoma was comparable to NGGCTs in the Thai population in our study, likewise reported in Singapore 17 and Malaysia. 19 This result might relate to the different diagnostic criteria between germinomas and NGGCTs among several institutions, either based on tumor histology,12,20,21 or histology plus tumor marker levels.14,19 However, integrating radiological and laboratory diagnostic criteria with histological findings remains essential to diagnose patients with ICGCTs in countries with limited resources. 17
The serum and CSF tumor marker levels are an essential evaluation to diagnose secreting ICGCTs or NGGCTs. The elevation of AFP >10 IU/L and/or β-HCG >50 IU/L is one of the criteria for diagnosing NGGCTs. 11 However, mild elevations of serum β-HCG or AFP could be observed in pure germinomas.5,22,23
Although serum and CSF AFP accounted for a higher percentage of agreement than β-HCG, the overall consistency between serum and CSF AFP and β-HCG levels was desirable with a moderate level of agreement. This could imply that assessing either serum or CSF is sufficient for diagnostic evaluation to lower the unnecessary lumbar puncture procedures among patients with ICGCTs.
However, diagnosing NGGCTs from tumor marker values is challenging due to some discrepancies in tumor marker values. In our study, the concordance between serum and CSF AFP (84.3%) and β-HCG (68.4%) were relatively high. The discrepancy between serum and CSF AFP (15.8%) and β-HCG (31.6%) were acceptable with one or another representing abnormal levels. β-HCG was found with more divergence than AFP likewise found in a related study of CNS germinomas, in which most patients had elevated CSF but normal serum β-HCG levels. 24 Therefore, serum may be less informative and sensitive than CSF β-HCG levels because the blood-brain barrier may postulate the effect of higher CSF β-HCG compared with serum levels. 25 Contrarily, a higher serum than CSF AFP level was observed among patients from China. 26
Interestingly, NGGCTs who had normal AFP (63.2%) and β-HCG (26.3%) levels in both serum and CSF were determined. This might be due to the diversity of histology variants among patients with NGGCTs, of which some cell types may secrete tumor markers below the cut-off level. Shin et al 27 defined nonsecreting germinomas as β-HCG level <3 IU/L and serum β-HCG ranged from 39 to 260 IU/L among 6 secreting germinomas. With this cut-off level, clinical characteristics and survival outcomes did not significantly differ between both groups of patients. 27 In our study, 33.3% of patients with germinoma were secreting germinomas and were defined as NGGCTs. Given the variety of cut-off levels of β-HCG in different circumstances, the appropriate cut-off level in specific ICGCTs subtypes is warranted to be more applicable for diagnostic classification and treatment selection. For these reasons, tissue diagnosis remains a crucial investigation, particularly in non-secreting ICGCTs.
The therapeutic strategies and treatment outcomes of ICGCTs in Thailand have gradually improved. From 1990 to 2000, using radiation treatment with or without surgery, 5-year OS was 81%. 28 From 2000 to 2008, adding chemotherapy to the treatment protocol, 5-year OS had improved to 88%. 18 Currently, combining chemotherapy and irradiation are the mainstay treatment modalities among pediatric patients with ICGCTs. 8 Patients with pure germinoma exhibited excellent outcomes, of which minimizing late effects was the key therapeutic approach. On the other hand, treatment modification to improve outcomes remains challenging in NGGCTs. 6 Concerning long term treatment effects from irradiation, chemotherapy alone among patients with pure germinoma was conducted with unsatisfactory outcomes.29,30 Platinum-based chemotherapy regimens, using cisplatin31,32 and carboplatin,11,33 have been acknowledged to achieve outcomes among patients with ICGCTs. Carboplatin-based regimen instead of cisplatin followed by irradiation has been employed as a standard regimen for childhood ICGCTs in Thailand since 2013. Worawongsakul et al 10 reported an excellent OS of 96% and EFS of 94% for patients with ICGCTs treated with a carboplatin-based regimen with lower ototoxicity effects. They found that germinomas had higher OS and EFS but without statistical significance, similar to our report. However, many studies reported significantly superior outcomes in patients with germinoma over NGGCTs.20,21 Age at diagnosis was not associated with outcomes in a related report. 17 However, in our study, age <8 years became an independent risk factor related to mortality. The outcome disparity may be an effect of treatment variation among studies.
Residual tumor resection should be considered to achieve desirable outcomes in NGGCTs. 6 The SIOP-CNS-GCT 96 trial 31 reported a superior 5-year progression-free survival (PFS) in NGGCTs, but not for patients with germinoma without the residual disease. Matsutani et al 34 reported a superior survival outcome in extensive tumor removal compared with partial removal or biopsy. In our study, patients with NGGCTs (58%) underwent surgery significantly more than patients with pure germinoma (23%), but did not differ in surgical extent. Interestingly, OS and EFS between patients with pure germinoma and NGGCTs did not exhibit statistically significant differences. Otherwise, patients undergoing tumor resection had a higher risk of relapse from multivariate analysis. The possible reason may be due to a serious post-operative CNS infection, 19 which may have delayed treatment and affected outcomes. Therefore, tumor resection may not be required as a standard treatment and should be consider among patients with failure from chemotherapy and radiation.
Our study confirmed that irradiation combined with chemotherapy was a fundamental treatment to achieve outcomes,6,29,30 and an independent risk factor for survival outcomes in pediatric ICGCTs. Worawongsakul 10 and Villano et al 20 reported significantly inferior outcomes among patients with ICGCTs treated without irradiation compared with those who undergoing irradiation. In the Western population, the 3-year PFS and OS of NGGCTs were 88% and 93%, respectively. 9 Appropriate radiation dosages and techniques should be explored to optimize disease control and minimize the toxicity from treatment.
The present study constitutes a multicenter study exploring clinical characteristics, factors associated with outcomes using the ThaiPOG-ICGCTs protocols and consistency of serum and CSF tumor markers of Thai pediatric ICGCTs. A national protocol with multimodality treatment by multidisciplinary professionals was well established to facilitate the standard of care and improve outcomes. Further studies exploring the long term effects and biological research in ICGCTs would be informative and may facilitate the development of novel therapies for enhancing outcomes.
Limitations
This study employed a retrospective design concerning some essential but unavailable data. Moreover, the study included patients from 3 institutions that might not represent the entire population of Thailand. Furthermore, the reliability and sensitivity of tumor marker levels from lumbar or ventricular CSF were uncertain. Legault et al 35 reported the lower β-HCG level from ventricular CSF compared with lumbar CSF. The variation of surgical techniques and extent, radiation dose, and techniques could also have affected distinct outcomes.
Conclusion
The outcomes of pediatric ICGCTs in Thailand were comparable to those of developed countries, and both pure germinomas and NGGCTs are similar. The fundamental treatment of ICGCTs was chemotherapy combined with radiation to achieve outcomes. Unfavorable prognostic factors comprised age <8 years, surgery, and nonradiation treatment. Tumor marker values, with either serum or CSF, were adequately required and useful to classify patients with ICGCTs. However, tissue diagnosis remains a crucial investigation, particularly in nonsecreting ICGCTs.
Supplemental Material
sj-docx-1-gph-10.1177_2333794X221141765 – Supplemental material for Clinical Outcomes and Diagnostic Consistency of Serum and CSF Tumor Markers in Pediatric Intracranial Germ Cell Tumors in Thailand: A Multicenter Study
Supplemental material, sj-docx-1-gph-10.1177_2333794X221141765 for Clinical Outcomes and Diagnostic Consistency of Serum and CSF Tumor Markers in Pediatric Intracranial Germ Cell Tumors in Thailand: A Multicenter Study by Lalita Sathitsamitphong, Chalinee Monsereenusorn, Piti Techavichit, Kanjana Shotelersuk, Piradee Suwanpakdee, Piya Rujkijyanont and Pimlak Charoenkwan in Global Pediatric Health
Supplemental Material
sj-docx-2-gph-10.1177_2333794X221141765 – Supplemental material for Clinical Outcomes and Diagnostic Consistency of Serum and CSF Tumor Markers in Pediatric Intracranial Germ Cell Tumors in Thailand: A Multicenter Study
Supplemental material, sj-docx-2-gph-10.1177_2333794X221141765 for Clinical Outcomes and Diagnostic Consistency of Serum and CSF Tumor Markers in Pediatric Intracranial Germ Cell Tumors in Thailand: A Multicenter Study by Lalita Sathitsamitphong, Chalinee Monsereenusorn, Piti Techavichit, Kanjana Shotelersuk, Piradee Suwanpakdee, Piya Rujkijyanont and Pimlak Charoenkwan in Global Pediatric Health
Footnotes
Acknowledgements
The authors gratefully acknowledged Dr. Chusana Khaiman, Integrative and Innovative Hematology/Oncology Research Unit, Department of Pediatrics, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand for initially collecting data, Mrs. Supak Cae-Ngow, Office of Research and Development, Phramongkutklao College of Medicine for statistical analysis, the English Editing Service, Research Affairs, Faculty of Medicine, Chulalongkorn University for English language editing assistance and the ThaiPOG for supporting this study.
Authors’ Note
Kanjana Shotelersuk is now affiliated with Division of Radiation Oncology, Department of Radiology, Faculty of Medicine, Chulalongkorn University, Bangkok, Thailand. Piradee Suwanpakdee is now affiliated with Division of Neurology, Department of Pediatrics, Phramongkutklao Hospital and Phramongkutklao College of Medicine, Bangkok, Thailand.
Author Contributions
LS collected and interpreted data, drafted, and edited the manuscript. CM conceptualized and designed the study, accumulated, analyzed, and interpreted data, managed the program overall, and was a major contributor to composing the manuscript. PT assisted in developing the study concepts and interpreting data. All authors contributed to patient care and critically reviewed and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The funding to conduct the study was obtained from the Phramongkutklao College of Medicine, Royal Thai Army.
Ethical Approval and Consent to Participate and for Publication
Written informed consent and assent forms were waived. This study was approved by the Institutional Review Board, Royal Thai Army Medical Department according to the ethics principles of the Declaration of Helsinki (1975) and its revision (reference number: IRBRTA 045/2562)
Data Sharing Statement
The datasets generated or analyzed during the current study are not publicly available due to privacy or ethics restrictions. The data are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
