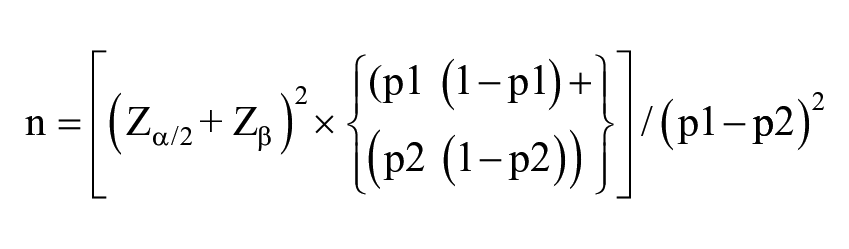Abstract
Background. Aflatoxin levels are very high in animals and humans in places where cereals are poorly stored. In this study, Novasil was evaluated for safety and efficacy in children. Methods. Children (200) aged between 2 and 9 years were put into Novasil and placebo group. Participants received either 1.5 g of Novasil or calcium carbonate in their food. Urine samples were analyzed for AFM1 by HPLC, blood samples were assayed for complete blood count and chemistries. Results. Aflatoxin M1 levels in the Novasil treated group, significantly reduced to 60% compared to an increase of urine AFM1 in the placebo group. Hematological parameters did not change except for an increase in hemoglobin level in the Novasil group. Biochemical parameters remained unchanged except calcium ions. Glutathione levels in the Novasil increased, compared group to the placebo group. Conclusion. Novasil is safe, reduce aflatoxin bioavailability in humans while improving GSH antioxidant capacity as well.
The trial has been registered with Pan African Clinical Trial Registry (www.pactr.org). A WHO registry for clinical trials with a unique identification number PACTR202202797930675.
Introduction
Aflatoxins are toxic secondary metabolites that are produced by molds, mainly Aspergillus flavus and A. parasiticus. The fungi grow in soil, decaying vegetations and various food crops such as maize, peanuts, wheat and rice. The fungal growth and development of the toxin are facilitated by a combination of inappropriate food storage conditions and hot humid climates. The toxins subsequently contaminate cereals and animal products such as eggs, meat, and milk. 1
Aflatoxins are mutagenic, cause immunosuppression and have severe detrimental effects on the human liver. 2 The most toxic aflatoxin compound, B1, can permeate the skin, 3 while the liver can convert the aflatoxin to a reactive epoxide intermediate or hydroxylated to form the less dangerous aflatoxin M1. Aflatoxin B1-8, 9-epoxide binds to reduced glutathione (GSH), which is essential in protecting tissues from the harmful effects of bioactivated aflatoxin. 4
One potential factor contributing to poor child health is increased exposure to aflatoxin-contaminated foods. 5 Cereal-based complementary foods for infants and young children may be contaminated with high levels of aflatoxin in Ghana.6,7 Besides the hepatotoxic and immunosuppressive effect of aflatoxin, the toxin has also been found to be an anti-nutrient and has been associated with growth disorders in infants and children. 8 Thus, it is necessary to conduct intervention studies that present therapeutic strategies to prevent aflatoxin bioavailability in children. Due to its high cation exchange, large surface area, mechanical stability, and layered structure, clay has the potential to be a strong absorbent. 9
The discovery of a healthy enterosorbent-based clay (Novasil), a natural clay containing calcium montmorillonite, presents an intervention strategy to reduce aflatoxin exposure and eradicate aflatoxicosis in animals and humans. Novasil reduces aflatoxin exposure by attracting molecules of positive charges due to the negative charge of the clay compound. 10 Human Novasil intervention trials in Ghanaian adults, previously recorded a reduction of aflatoxin bioavailability.11,12 In a study by Mitchell et al, 13 reported that Novasil inclusion in food was safe at a dose of 1.5 g/day and showed a 52% reduction in aflatoxin bioavailability compared to the placebo group in a 14-day safety and efficacy study in Ghanaian children. However, it is appropriate to broaden the scope of the study by Nicole and her group to assess the safety and efficacy of Novasil for a longer period in children. Therefore, we conducted the assessment for 60 days.
Methodology
Study Design, Study Participants, and Study Period
This study was done on children with detectable urinary aflatoxin in the Ejura-Sekyedumase District of the Ashanti Region of Ghana.
Participants’ Selection
The cluster sampling method was used to select qualified participants for the study. Ejura-Sekyedumase District was divided into 4 clusters (communities). A cluster was chosen using a simple random sampling technique, and all eligible subjects within households whose parents/guardians consented were included. Fifty-five (55) participants from each cluster were selected to obtain a total of 220 participants. Two hundred healthy children of both sexes (age: 2-9 years old) who fulfilled the inclusion criteria were selected for the intervention study and assign to 2 groups of 100 each for placebo and Novasil treatment groups.
Experimental Design Framework and Participants Inclusion Criteria
Figure 1 details the experimental framework of the study. Any child participant with urinary aflatoxin level of < 0.5 µg/kg was excluded from the study. Participants with detectable urinary aflatoxin (>0.5 µg/kg) were randomly placed on Novasil and placebo (calcium carbonate). Participants placed on either treatment arm were screened for full blood count, kidney and liver function tests, serum electrolyte assay (Na. K, Ca), urinary aflatoxin and GSH at baseline and at day 60.

Experimental and sampling framework for the study.
Preparing the Novasil Clay for the Experiment
Novasil used in this study was obtained from Engelhard/BASF Chemical Corporation (Jackson, MS, USA). The product was donated by Texas A&M University in the USA. Novasil clay was tested analogously for its effect on various environmental contaminants such as dioxins and heavy metals. The metals and dioxin analyses of Novasil were below the tolerable daily intake in accordance with the limit set up by the World Health Organization (WHO) and the Joint Food and Agriculture Organization/WHO Expert Committee on Food Additives. Novasil was sterilized by electron beam irradiation to prevent any possible bacterial or viral contamination before trial initiation.
Recruitment and Training of Field Assistants
The study recruited 5 field assistants. The field assistants included a Physician, 2 Nurses and 2 Laboratory Scientists. They were trained on sampling and data collection by the Principal Investigator.
Sample Size Estimation
The parameters for the sample size calculation were derived from a previous study which found that 52% of subjects treated with Novasil (drug A) was efficacious. 13 and a clinically significant difference of 19% when compared to a placebo is acceptable. Level of significance = 5%, Power = %, Type of test = two-sided.
The following formula was used for calculating the sample size:
Where:
n = sample size required in each group,
p1 = proportion of subject cured by Drug A = 0.52,
p2 = proportion of subject perceived to be cured by Placebo = 0.33,
p1−p2 = clinically significant difference = 0.19
The sample size required per group, according to the above formula, was 100. As a result, the total sample size required was 200, using a 2-tailed z-test of proportions between 2 groups with 80% power and a 5% level of significance.
Recruitment and Assigning Study Participants to Treatment Group
Study participants were recruited from the Ejura-Sekyedumase district of the Ashanti Region of Ghana using the cluster sampling techniques. The community is a rural area where residents are predominantly subsistence farmers. All recruited participants were between 2 and 9 years of age. Consent was obtained from the parents or legal guardians resident in the community. Consent documents were explained to each participant, and then signed by participant’s guardian before initiation of the study. Participants were randomly assigned to the 2 arms of study, namely, treatment with Novasil or placebo group (treatment with calcium carbonate). The treatment arm consisted of 1.5 g of Novasil and the placebo group took 1.5 g of calcium carbonate. Both arms of the study received 1.5 g of Novasil or 1.5 g of placebo once daily. Doses were weighed into identical packages at the Noguchi Memorial Institute for Medical Research (NMIMR) to ensure that monitors and participants were blinded to their treatment. Trained study monitors mixed each participant’s treatment into their normal maize porridge breakfast meal. Monitors visit participants each day to record any adverse effects and report to the study physician for treatment.
Sample Collection, Processing, and Analysis
Three milliliters of baseline blood and urine samples and another at day 60 of treatment were collected from each child. Blood samples were collected by trained phlebotomists at the Ejura District Hospital while early morning urine samples were at day 0 and day 60 by trained monitors. Whole blood samples were used for hematological analysis whereas serum samples stored at −20°C, were used for biochemical analysis. Urine samples were used for detecting aflatoxins.
Laboratory Analysis
Detecting of aflatoxin in the urine (AFM1)
Aflatoxin Vicam AflaTest kits were used according to the Association of Official Analytical Chemists’ method (AOAC) for aflatoxin analysis. AflaTest columns were purchased from VICAM (Watertown, MA, USA). Analysis of urinary AFM1 levels followed previously published method (Wang et al 11 ). After the immunoaffinity column (AflatestWB; VICAM) clean up, high-pressure liquid chromatography (HPLC) system with fluorescence detection was used to analyze urine aflatoxin M1 concentrations (Shimadzu Corp., Kyoto, Japan). Urinary aflatoxin concentrations were expressed as picograms per milligram creatinine to correct for variations in urine dilution among samples.
Determination of Blood Glutathione Levels
The blood samples were homogenized and diluted 200X with GSH buffer before use. The reaction mixture comprised 50 μL of GSH buffer (0.1 M NaH2PO4, 5 mM EDTA, pH 8.0 with 1 N NaOH) with 50 μL of the blood sample and 10 μL of 0.75 mM O-Phthaldehyde (OPT). The mixture was incubated for 15 minutes at room temperature in the dark, and fluorescence was measured at 340 nm for excitation and 460 nm for emission. 14 The experiment was carried out in triplicate. The reduced glutathione levels were calculated using the calibration curve and expressed as mg/mL glutathione equivalent.
Complete Blood Count
Complete blood count parameters analyzed included hemoglobin level (Hb), total white blood cell count (WBC) and total red blood cell count (RBC). White blood cell count analyzed were lymphocytes, neutrophils, monocytes, and eosinophils (Sysmex KX 21 hematology analyzer from Europe GmbH, Germany).
Kidney and Liver Biomarkers
The levels of kidney and liver biomarkers in serum were analyzed using an automated colorimetric method with Flexor E, endpoint automation (Vital Scientific, Netherlands). Liver markers analyzed included alanine aminotransferase (ALT), aspartate aminotransferase (AST), albumin, gamma-glutamyl transferase (GGT), total protein and total bilirubin. Kidney markers analyzed include creatinine, urea, sodium, chloride, and potassium. Serum calcium was also measured.
Statistical Analysis
Statistical analysis was conducted using Sigma Stat 3 (Jadel Scientific Software, 2003, USA). A descriptive analysis was employed to analyze for mean, standard deviation and the range of variables. Paired T-test was used to analyze AFM1 data, full blood count, biochemical parameters for the placebo and treatment group. The Signed Rank test was used to analyze significance between data where it was not normalized among the intervention group and the placebo group. Statistical significance was defined as a 2-tailed P-value < .05.
Ethical Consideration/Informed Consent
Ethical clearance for this study was obtained from the Institutional Review Board of the Noguchi Memorial Institute for Medical Research (NMIMR), University of Ghana (reference: IRB0001276) and the Institutional Review Board (IRB) of the University of Cape Coast (UCC) (reference: UCC/RB/CHAS/2019/109). Informed written consent were obtained from all participants in accordance with human experimental guidelines of the NMIMR (IRB) and UCC (IRB). Confidentiality was observed throughout the research process and the data made accessible only to the Principal Investigator and Study Supervisors.
Results
Table 1 indicates a total of 200 children (98 males and 102 females) between the ages of 2 and 9 years who were enrolled into the study. Only 2 participants (one from treatment and one from placebo) could not give blood at the post-intervention stage. Also, 2 participants within the placebo group were diagnosed and treated for malaria. All the participants completed the study. Table 1 also shows the average z score analysis of participants. The average z score for weight-for-age for the treatment group was −0.4 SD with a range of −2.35 to 1.9 SD, whiles the placebo had an average z score for weight-for-age as −0.3SD with a range of −2.35 to 1.9 SD. The average height-for-age z score was −0.4SD in a range of −3.6 to 2.5 SD in the treatment group. The placebo group also showed an average z-score value of −0.4 for height-for-age in a range of -3.6 to 2.5 SD.
Characteristics of Study Participants.

Data are represented as mean ± SD. All the data are baseline values. Significance: P < .05.
Adverse/Side Effects of Novasil
There were no significant side effects recorded with respect to Novasil administration during the 60-day intervention period. No major variations in the number of side effects were found between the treatment and control groups (P = .746) or differences in severity of symptoms reported (P = .700).
Analysis of Hematological Parameters
Table 2 shows an analysis of the hematology results obtained before and after each treatment group. With the exception of hemoglobin and platelets levels that were elevated after Novasil treatment, no other hematological parameter was significantly altered after both Novasil and placebo treatment.
Analysis of Hematology Parameters in the Treatment Groups.

Data is represented as mean ± SD.
Indicates an increased level of hemoglobin. N = 200 before intervention, N= 198 at post intervention.
Effect of Novasil and Calcium Carbonate on Liver and Kidney Parameters
Decrease levels in blood GGT levels were observed after Novasil treatment. In the placebo group, ALT was significantly elevated. Notwithstanding these observations, all other Day-60 parameters did not significantly differ from baseline levels (Table 3).
Analysis of serum liver and kidney injury biomarkers.

Data is showed as mean ±SD.
Indicates decrease levels within the treatment group, whiles b indicates increase in the placebo group. N = 200 before intervention, N = 198 at post intervention.
Analysis of Blood Glutathione and Urinary Aflatoxin M1
Baseline mean urinary aflatoxin M1 levels in the treatment group (AFM1 = 811.2 pg/mg creatinine) and the placebo group (AFM1 = 801.3 pg/mg creatinine) were statistically not different (P > .05). However, after a 60-day Novasil treatment, mean urinary level of aflatoxin significantly reduced to 329.1 pg/mg creatinine (P = .013) representing 60% reduction. On the other hand, Day-60 mean urinary level of aflatoxin increased to 1801.2 pg/mg creatinine increased within the placebo group representing 125% increase (Figure 2).
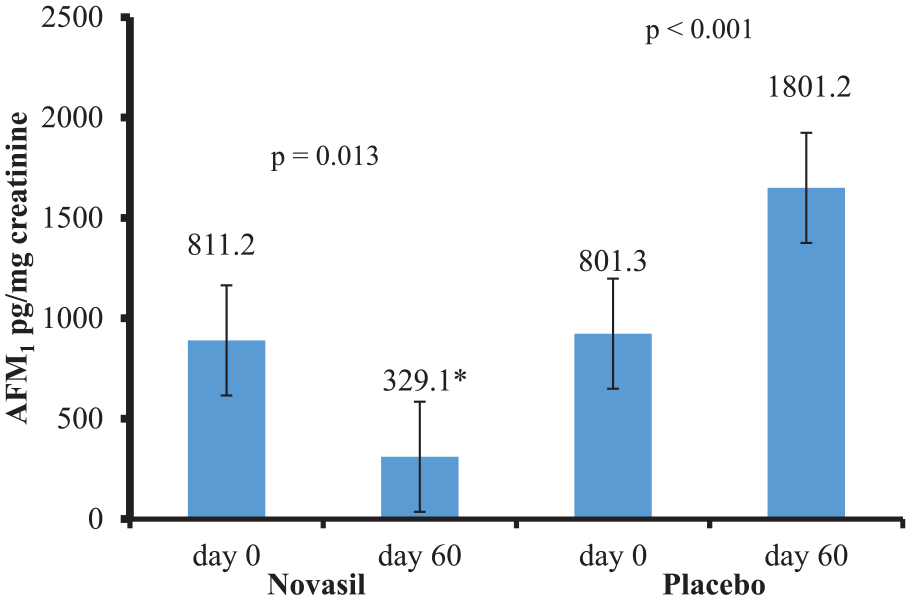
Mean AFM1 levels in pre- and post-treatment groups.
Whereas urinary levels of aflatoxin reduced in the treatment group, mean serum levels of GSH increased from 0.27 mg/mL (baseline) to 0.29 mg/mL (Day-60) representing 7.4% increase in blood GSH. However, the mean baseline and mean Day-60 levels of GSH in the placebo group remained unchanged (Figure 3).

Mean GSH levels in pre- and post-treatment groups.
Discussion
Aflatoxin-related health effects are widespread especially in developing countries that rely on food commodities such as cereals as their primary source of food. Aflatoxin exposure during infancy, through breast milk and complementary foods, may result in growth impairment, immune suppression and micronutrient deficiencies.15,16 Over the past years, high levels of aflatoxin exposure have been detected in people from the Ejura Sekyedumase District in the Ashanti Region of Ghana.11,12,17,18
Weanimix, made from groundnuts, beans and maize, is a popular semi-solid food used to wean children between the ages of 2 and 9 years in Ghana. The use of weanimix is intendent to correct malnutrition in children. However, weanimix has been found to be heavily contaminated with aflatoxin in 2 studies done in the Ejura Sekyeduamse District.7,18 It was not surprising to realize that close to 38% of children between the ages of 6 and 59 months were found to be stunted in the Atwima Kwanwoma District in the Ashanti region of Ghana 19 a district in the same region as this study district. It is noteworthy that farmers in the Ejura Sekyeduamse District are leading producers of maize, beans and peanuts in Ghana. 20 When cereals are contaminated with aflatoxins, some of the suggested harmful effects on humans are vomiting, diarrhea, hepatitis, cirrhosis, immunosuppression, miscarriages, mutagenic and teratogenic effects, and different cancers. 21 One of the most fundamental approaches to lowering the risk of aflatoxicosis in humans is to avoid eating foods contaminated with aflatoxin. However, many communities in developing countries simply are poor and ignorant about aflatoxin poisoning. As a result, strategies to reduce aflatoxin poisoning especially in children are required particularly in aflatoxin endemic areas. This will help to reduce the occurrence of aflatoxin related disease in children. Therefore, this study aimed at evaluating the safety and efficacy of Novasil in children who were at risk of developing aflatoxin related diseases in Ghana.
The findings of this study show that giving healthy children (ages 2-9) dietary Novasil powder at a concentration of 1.5 g/day for 60 days was safe and also reduced the bioavailability of aflatoxin metabolite in urine. These findings are consistent with other studies in Ghana and elsewhere.11-13,22,23
Novasil exhibited binding capacity to aflatoxin by reducing urinary aflatoxin levels by 60% while increasing the glutathione levels in the children. Novasil clay has been found to have high affinity for aflatoxin due to the opposite charges they possess. Due to the electronegativity of the Novasil clay, it has high binding capacity in the gastrointestinal tract for aflatoxin. 10 Novasil has been found to absorb aflatoxin in both animal models and human studies, reducing the bioavailability of the blood toxin. 17 Additionally, the use of Novasil on humans did not affected the utilization of vitamins and trace elements in the body.24,25
Glutathione, a potent antioxidant is critical in protecting tissues from the damaging effects of toxicants such as bioactivated aflatoxin. 4 In an animal study, aflatoxin induced reduction in the level of blood GSH. 26 The present study observed an increase in blood GSH levels in the treatment group at the end of the Novasil intervention. This study also confirms the potential cytoprotective nature of Novasil. The systemic safety of Novasil has been demonstrated in this study. Novasil did not significantly alter the blood levels of WBC and its sub-cells, lymphocytes, monocytes and neutrophils. Platelets levels were also not affected however, at day 60 of Novasil use, hemoglobin levels were increased. With the exception of calcium levels that were elevated, all liver and kidney parameters did not change after the 60-day treatment period. The efficacy of Novasil has been previously determined in human and animal models.13,27 Elevation of total serum Ca2+ levels post-Novasil treatment supports the studies by Afriyie-Gyawu et al 17 , and Marroquín-Cardona et al. 28 The elevation could be due to the breakdown of calcite and exchangeable Ca2+ from montmorillonite clay as previously explained17,28 Since Novasil clay commonly acts as a calcium supplement, it was likely that the increase in total serum calcium levels seen in the treatment group in this study was due to the addition of Novasil to the food. Calcium levels in the placebo group did not see any change in levels although the placebo group also received calcium carbonate. Possibly due to the lack of supplementation in calcium levels in the placebo group. However this development needs to be further investigated.
Conclusion
This study has shown that the addition of Novasil to cereal-based weaning foods is beneficial in reducing aflatoxin bioavailability in children. This has the potential to reduce the negative effects of aflatoxin exposure. Therefore, we recommend that a longer safety and efficacy trial be establish to ascertain whether Novasil could reduce urinary aflatoxin level to less than 0.5 µg/kg. Furthermore, it is essential to evaluate the impact of Novasil on nutrients, growth and anthropometric indicators for improvement in children’s health in a longer study. Furthermore, a longer safety trial is necessary to establish whether Novasil could affect micronutrient absorption, while controlling for critical dietary nutrient intake. The study showed that levels of aflatoxin exposure in children at Ejura-Sekyedumase continues to be a major problem. Thus, parents of the study participants were educated on the dangers of aflatoxin exposure to the development of their children. Parents were encouraged to feed them on uncontaminated cereals.
Footnotes
Acknowledgements
We appreciate the unmeasurable support from BASF SE food fortification, Human Nutrition Germany, for providing financial support for the field work. We are grateful to Ghana Education Trust Fund (GETFUND) for providing financial sponsorship for the academic work. To parents who voluntary agreed for their children to be part of the study we say thank you. We sincerely appreciate the children who took their time to be part of the research, and also the kindness of the Nurses and Laboratory Scientist who helped in data and sample collection. To Dr. Seidu Zakari of Ejura-Sekyedumase Hospital, we say thank you.
Author Contributions
Justice Kumi, contributed to the concepion and design, drafted the manuscript.
Regina Appiah-Opong, contributed to the design and critically reviewed manuscript.
Dietrich Rein, contributed to interpretation and critically reviewed manuscript.
Godfred Egbi, Contributed to conception and manuscript review.
Enoch Aninagyei, contributed to analysis and critically reviewd manuscript.
Alex Boye, contributed to interpretation and manuscript review.
Desmond Omane-Acheampong, contrbuted to conception and design, critically reviewed manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author(s) received financial support from BASF SE Food Fortification, Human Nutrition, Ludwigshafen, Germany for the research.
Ethical Approval
Ethical Approval for the study was obtained from the Institutional Review Board of the Noguchi Memorial Institute for Medical Research (NMIMR), University of Ghana (reference: IRB0001276) and the Institutional Review Board (IRB) of the University of Cape Coast (UCC) (reference: UCC/RB/CHAS/2019/109).
Consent
Informed written consent were obtained from all participants through their parents or guardian in accordance with human experimental guidelines of the NMIMR (IRB) and UCC (IRB). Confidentiality was observed throughout the research process and the data made accessible only to the Principal Investigator and Study Supervisors.