Abstract
Recurrent infections and weak immunity were found to be linked with zinc deficiency especially in children. The goal of this study is to measure the level of blood zinc in children aged 6 months to less than 5 years old, and to assess the effect of 4 months of daily zinc supplements on the incidence and the severity of diarrheal morbidity. A randomized controlled trial study was conducted in Pediatric Outpatient Clinic in Ain Shams University Hospital on 140 children less than 5 years old who are apparently healthy. Random assignment of the sampled children to group receiving zinc (70 children) or group receiving placebo (70 children) was done. A questionnaire containing questions about occurrence of diarrhea was used during follow up. Serum zinc at baseline was measured. The mean age was 25.26 ± 15.7 months. Mean of serum zinc was 51.08 μg/dL, with 70% had low baseline serum zinc level. Cumulative incidence of diarrhea, number of diarrhea episodes per child and frequency of stools/day decreased significantly in zinc group (P < .05). Risk ratio was found to be 0.79, 95% CI: 0.64 to 0.97. we concluded that incidence of diarrhea and its severity reduced in children received zinc daily for 4 months.
This trial was registered at www.clinicaltrials.gov as NCT05002101.
Introduction
Many functions of the body systems and organs require the presence of adequate level of a micronutrient called zinc. Zinc is important for protein and lipid processing and metabolism, cell integrity and maturation. Moreover, besides its regulatory roles, zinc has an essential function in metabolism of different nutrients, for example it helps in catalysis, synthesis and breaking down of nutrients. 1 The role of zinc in protection and response of the body to infections is well documented especially in children. It is known that every cell in the immune system, either humoral or cell mediated, is influenced by zinc. Immune cells development and functioning require the presence of zinc. Moreover, zinc is an integral part of a thymic hormone, thymulin, which is required for maturation of T cell.2,3
Zinc is required to be included in everyday diet because it was not stored in the human body. 4
For children under 5 years, they should obtain about 3 to 5 mg of zinc from diet every day; that is to fulfill the dietary requirements of nearly all (97%-98%) healthy persons (Recommended Dietary Allowance [RDA]). The main food containing zinc includes meat, meat products, milk, dairy products, and eggs. Plant-based food as grains and cereals are rich in zinc also; but contain phytate as well which decrease the bioavailability of zinc in diet.5,6
In less developed nations, low zinc consumption in the diet is a common problem (affecting around 2 billions) where they relay mainly on grains and cereals in their diet, while consume animal products in modest quantities. 7
Repeated infections and weak immunity were found to be associated with zinc deficiency especially in under 5 years children. Common infectious diseases which are linked with zinc deficiency were diarrhea, pneumonia, and malaria which all can affect the growth of the children negatively.7,8
The World Health Organization reported in 2009 that zinc deficiency below 5 years resulted in 13% of lower respiratory tract infections (basically pneumonia), 10% of malaria bouts, and 8% of diarrhea. 9
Around the world, diarrhea is considered as one of the major killers of children under 5 years; it is the second common cause of death in this age group, killing about 525 000 children annually. 10
The passing of 3 or more loose or watery stools daily is considered diarrhea. From clinical point of view, acute diarrhea is considered if symptoms occur only for few days (less than 2 weeks); while in persistent diarrhea, symptoms last for long duration (more than 2 weeks). Passing 3 or more watery stools with blood is known as dysentery. 11
A meta-analysis published by Aggarwal et al 12 concluded that supplements of zinc markedly decreased diarrheal incidence, severity, and duration.
World Health Organization (WHO) recommended zinc supplements for children under 5 years who suffer from diarrhea as an adjuvant to their treatment regimens in developing countries. 11 On the other hand, there is limited evidence on the effects of zinc supplementation on prophylaxis against morbidities in this age group.
By searching results of the current reviews, it was found that the effect of zinc supplements on prevention of morbidities was not studied on Egyptian children under 5 years. Accordingly, the present study aimed to measure the level of serum zinc among healthy children between 6 months and 5 years attending Pediatrics Outpatient Clinic in Ain Shams University Hospital, and to assess the effect of daily zinc supplements for 4 months on the occurrence of diarrhea among these children.
Material and Methods
An interview questionnaire was conducted at the start of the study, before zinc administration, and then at end of each month for 4 months of zinc intervention. Laboratory investigations for serum zinc before start of intervention was also conducted.
At Baseline
Other questions included in the questionnaire were about hospital admission during the diarrheal episode and treatment received especially the intake of oral rehydration solution. If the stool contained blood, the episode was classified as dysentery. 15
Children with serum zinc level between 60 and 100 μg/dL are considered normal. While children below 60 μg/dL were considered having low serum zinc. 16
The zinc was prepared such that in each 5 mL solution there was a 7 mg zinc sulfate. Children weighing >10 kg were instructed to consume 5 mL of syrup daily, while children weighing ≤10 kg were instructed to take 2.5 mL. A 150 mL bottle was stored in the child’s home and changed every month.
The placebo was similar to the zinc syrup in color, odor, consistency, and taste, but it was devoid from the vitamin or the mineral. The zinc and placebo syrups came in the identical bottles.
Follow Up
The follow up period was 4 months. Telephone calls were done on weekly bases to ensure compliance of the care givers. On monthly bases, each mother was asked about the occurrence of diarrhea in her child in the previous month during their visit to the Pediatrics Outpatient Clinic or via a telephone call. For standardization, questions asked at each month throughout the follow up duration were the same as in the baseline questionnaire.
For Descriptive statistics, quantitative data was analyzed by mean and standard deviation, whereas qualitative data was analyzed by frequency and its corresponding proportion.
For inferential statistics, based on the type of data collected for each variable, the appropriate statistical analysis was done. The statistical significance difference of a parametric variable between 2 independent means of 2 groups was determined using the independent sample t-test. The Chi-square test was conducted to examine the association between 2 qualitative variables, but when the expected count in more than 20% of the cells was less than 5; Fisher’s Exact test was the test of choice. Statistical significance was defined as a 2-sided P-value of less than .05.
For incidence density calculation, person day was used. The person-day for the total studied population was (140 × 4 × 30) = 16 800. The person-day for either zinc or placebo group was (70 × 4 × 30) = 8400.
- Cumulative incidence: probability of developing disease over a stated period of time. 19
- Incidence density: the number of diarrhea episodes per person-time. 19
- Risk ratio: the cumulative incidence in exposed group (zinc group) divided by the cumulative incidence in the unexposed group (placebo group). 19
- Rate ratio: the ratio of the incidence density in the exposed group (zinc group) to the incidence density in the unexposed group (placebo group). 20
- Risk difference: the difference between the cumulative incidence in the unexposed group (placebo group) and the cumulative incidence in the exposed group (zinc group). 20
- Rate difference: the difference between the incidence density in the unexposed group (placebo group) and the incidence density in the exposed group (zinc group). 20
- Preventive Fraction: the risk difference is divided by the cumulative incidence in the unexposed group. 20
Results
In the current study, apparently healthy 178 children were approached for participation and assessed for eligibility, and 28 children from them were excluded. Among those excluded, there were 13 children didn’t meet the inclusion criteria and 15 declined to participate in the study. From the 150 children who were allocated at random to the 2 groups, 10 children (5 in zinc group and 5 in placebo group) lost to follow up within the first month. Data from 140 children were included into the final analysis as presented by flow chart Figure 1.

CONSORT flow chart for the study.
Out of 140 studied children, (58.6%) of the studied children were 2 or less years old, while (41.4%) of the children were more than 2 years. The mean of the age of the studied children was 25.26 ± 15.701 months. Males represented (53.6%) of the children. Most of children (90.7%) were living in urban areas. About (12.1%) of children’s mothers were illiterate, while (37.1%) of them completed secondary school. Regarding breastfeeding history in the first 6 months of age, most of the children (84.3%) were breastfed. Among those who were breastfed, 65% were exclusive breastfed. Regarding birth weight, most of the studied children (79.3%) had normal birth weight with mean of 2.81 ± 0.53 kg (Table 1).
Socio-Demographic Characteristics of the Zinc and Placebo Groups of the Studied Children.

Chi square test was used.
The difference between children in zinc group and placebo group was not statistically significant as regard age, gender, residence, mother’s education, breast feeding, exclusive breast-feeding duration and weight at birth of the studied children (P > .05).
In the total population, measuring the zinc in the serum revealed a mean of 51.08, with no significant difference from statistical point of view between children in zinc group and placebo group (Table 2). A serum zinc level of 60 to 100 μg/dL was regarded normal. While children below 60 μg/dL were considered having low serum zinc. In the current study, 70% of the studied children had low baseline serum zinc level (below 60 μg/dL), whereas only 30% had normal baseline serum zinc level. There was statistically insignificant difference between zinc and placebo groups regarding baseline serum zinc level (P > .05). Higher percent of the children in both groups had low baseline serum zinc level (below 60), (67.1% in zinc group and 72.9% in placebo group).
Comparison Between Zinc and Placebo Groups Regarding Baseline Serum Zinc level (µg/dL).

Independent t test was used.
Chi square test was used.
Regarding the cumulative incidence of diarrhea, there was a statistically significant difference between children in zinc and placebo groups after the third and the fourth month of follow up period and throughout the whole follow up period (ie, throughout the 4 months of the study) (P < .05). In a 24 hours period, severe diarrhea was determined if there were 6 or more liquid or semi-liquid stools. It was found that there was a statistically significant difference between children in zinc group and placebo group regarding percent of children with severe diarrhea throughout all the follow up period (P < .05). Among zinc group, 8.6% got severe diarrhea, while among placebo group more children (25.7%) got severe diarrhea (Table 3).
Comparison Between Zinc and Placebo Group Concerning Cumulative Incidence of Diarrhea and Percent of Children With Severe Diarrhea at Baseline and During Follow Up Period.

Chi square test.
Fisher exact test were used.
In all studied children, mean number of diarrheal episodes per child throughout the period of follow up was significantly lower in zinc group (1.1 episode) than in placebo group (1.69 episode). This difference was also significant in children with low serum zinc, but not in children with normal serum zinc.
Regarding mean of duration of diarrhea episode, there was a statistically insignificant difference between children in zinc group and placebo group throughout the whole period of follow up (by summation of all durations of diarrheal episodes for each child in the 4 months of follow up) (P > .05). On the other hand, there was a statistically significant difference between children in zinc group and placebo group after the third and the fourth month of follow up period separately (P < .05).
Severity of diarrhea episodes was measured by the frequency of stools/day. Concerning mean number of stools/day, there was a statistically significant difference between children in zinc group and placebo group over the whole 4 months of the follow up period (for each child, the highest number of stools/day in any of the episodes occurred in the 4 months was considered). The mean number of stools per day was 2.77 ± 2.346 stools/day in the zinc receiving group and 3.66 ± 2.45 stools/day in the placebo group. Also, there was a statistically significant difference between children in zinc group and placebo group per day after the third and fourth month of follow up period (P < .05) (Table 4).
Comparison Between Zinc and Placebo Groups Concerning Mean Number of Diarrheal Episodes Per Child, Duration of the Diarrhea Episodes and Frequency of Stools/Day.

Independent t test was used.
The highest number of stools/days was considered.
Regarding oral rehydration solution, among children with diarrhea, there was a statistically insignificant difference between children in zinc group and placebo group regarding receiving oral rehydration solution throughout all the follow up period (P > .05).
Calculating measures of risk, association, and impact of the zinc on occurrence of diarrhea in comparison to the placebo was done. Cumulative incidence and incidence density were lower in zinc group than in placebo group, where zinc supplement is protective against diarrhea (risk ratio was found to be 0.7895, which means that children of zinc group had
Effectiveness of Zinc Supplements on Diarrhea.
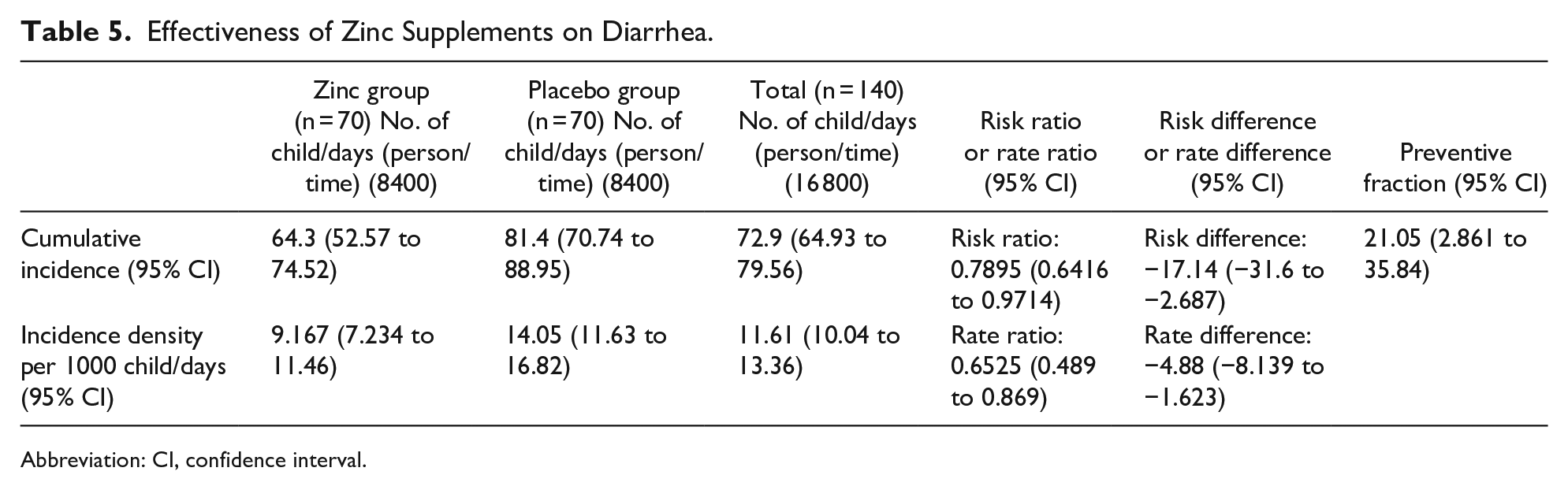
Abbreviation: CI, confidence interval.
Discussion
In the present study, serum zinc level was measured before the intervention and the calculated mean was 51.08 ± 23.37 μg/dL in all studied children, with no statistically significant difference between zinc and placebo groups. Accordingly, there would be a fair comparison between the intervention and control groups during the follow up. Low baseline serum zinc level was found in 70% of the studied population. This result was similar to Barffour et al 14 who found that 71% of the studied children had low serum zinc at the baseline.
There are some Egyptian studies measured serum zinc in different populations and revealed different concentrations. The mean of serum zinc concentration in these studies ranged from 57 up to 117.4 μg/dL.21-23
These differences and wide variations between studies regarding serum zinc level may be due to its normal physiological fluctuations; serum zinc level fluctuate by about 20% throughout the day, mostly because of the ingestion of food. After meals, there was immediate increase in serum zinc concentration, after which this concentration decreases for 4 hours and then increase again. However, the fluctuation pattern is changed during fasting, whereby it decreased from morning for some hours and then increase again. Moreover, serum zinc concentration was affected by presence of several conditions like acute infections inflammation, hemolysis, malnutrition, dehydration, and fever.24,25
In the present study, zinc supplementation was examined to evaluate its impact on diarrhea morbidity.
In the present study, the cumulative incidence of diarrhea in zinc group was significantly lower than that in placebo group after the third and the fourth month of follow up period and throughout the whole follow up period. Liberato et al 17 also concluded that prophylactic zinc was effective in decreasing incidence of diarrhea.
Risk of developing diarrhea in the zinc group was lower than that in placebo group, thus, risk ratio was found to be 0.7895; 95% CI: 0.6416 to 0.9714. Also, the risk difference was found to be −17.14%, which means that children of zinc group had 17.14 fewer cases of diarrhea per 100 children compared to children of placebo group. These results agreed with Aggarwal et al 12 who found that supplementation of zinc decreased the incidence of diarrhea significantly by 14% with a relative risk of 0.86. In addition, Brown et al 26 found that zinc supplementation caused a significant 20% lower incidence of diarrhea (relative risk = 0.80). The pooled analysis by Yakoob et al 27 revealed a significant reduction of 13% on the incidence of diarrhea in children receiving zinc in comparison to children in the control group, and the relative risk was found to be 0.87; 95% CI: 0.81 to 0.94.
Inconsistent with the previous studies, Luabeya et al reported that supplementation with zinc did not reduce diarrhea morbidity in rural South African children. The authors of this trial suggested that the cause of this inconsistency was that studies that found an effect on diarrheal morbidity hade often included poor nourished children. Children enrolled in Luabeya et al 28 trial had better anthropometric indices than those reported on in most other studies. Also Barffour et al 14 found no benefits of daily preventive zinc supplements on occurrence of diarrhea.
The current study revealed that the zinc supplements significantly decreased the overall number of diarrheal episodes in children of zinc group in comparison to the children of placebo group. This impact was more prominent in children with low baseline serum zinc level. This result agreed with Bhandari et al 29 who found a significant decline in the number of children who had recurrent diarrhea with significant decrease in the number of diarrheal episodes in children of zinc group in comparison to the children of placebo group. Moreover, Umeta et al 30 found that, in response to zinc supplements, there were better improvements in the incidence of diarrhea in children whose serum zinc was low at the beginning of the study, in comparison with children with higher baseline serum zinc levels.
On the other hand, Bhandari et al 29 found the observation in the opposite direction, such that the trend of lower diarrhea incidence was prominent only in children who had serum zinc higher than 60 μg/dL. Moreover, a clinical trial was performed by Rahmati et al 31 reported that, in terms of the mean number of diarrheal episodes, there was no substantial difference present between zinc and control groups.
Regarding duration of diarrhea episodes, there was no statistically significant difference between children in zinc group and placebo group throughout the whole period of follow up (by summation of all durations of diarrheal episodes for each child in the 4 months of follow up) (P > .05). Consistent with the current study, a review conducted by Brown et al 26 found that, in community-based trials, there was no substantial effect of prophylactic zinc supplementation on the duration of diarrhea. Similarly, Rahmati et al 31 found no apparent difference between the groups in terms of the duration of diarrheal episodes.
On the other hand, Garenne et al 32 revealed shorter length of diarrheal episodes in boys who get supplements of zinc. Meta-analysis conducted by Aggarwal et al 12 found that supplementation of zinc shorten the duration of diarrhea episodes.
Concerning frequency of stools/day, the present study found that the zinc group had significantly lower frequency of stools/day than that in the placebo group. These results are inconsistent with the clinical trial of Rahmati et al 31 who found that there was no difference of statistical importance between zinc receiving group and the other group regarding the number of stools/day.
Number of stools/day reflects the severity of the diarrhea episode, such that, in a 24 hours period, if the number of liquid or semi-liquid stools equals to or exceeding 6 times daily, it was considered as severe diarrhea. The present study found that percent children with severe diarrhea in zinc group was significantly lower than that in placebo group throughout all the follow up period. This result was inconsistent with the randomized, double-blind placebo-controlled trial conducted by Barffour et al 14 who found that there was no effect for the preventive zinc supplements on the severity of the diarrhea episodes.
Generally, the diversity in effects of zinc on prevention of diarrhea could be due to a varied patient population, the underlying zinc status of the population, study designs (community based or hospital based), the dose and form of zinc administrated, the methods of monitoring diarrhea, the outcome measured and possible confounding effects of child feeding practices.
Conclusions and Recommendations
The study found that daily zinc supplementation for 4 months to children aged 6 months to 5 years is beneficial in reducing diarrhea and may be used as a suitable preventative supplement. Zinc reduced the number of diarrheal episodes and its severity but had no effect on the duration of each episode in the current study. Thus we recommend the following: (1) Nationally nutritional assessment surveys need to be conducted to assess the population zinc status, (2) Further studies to be conducted to assess the effect of preventive zinc on different age groups, and (3) Prompt measures to be done to improve the zinc status of the population as: fortification of foods with zinc, cultivation of zinc rich plants, supplements of certain children with zinc and health education to the parents about foods containing zinc and proper weaning practices
Footnotes
Author Contributions
Conceptualization and design: A.E.-S., S.A.-H. and Y.G.-A.; acquisition, analysis, or interpretation of data: Y.S.-A., Y.G.-A. and M.M.-W.; assembled the data under Y.G.-A. supervision; writing—original draft preparation: Y.S.-A. and M.M.-W.; writing—review and editing: A.E.-S., Y.S.-A., Y.G.-A and S.A.-H.; supervision: A.E.-S. and S.A.-H. All authors read and approved the final version of the manuscript. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
