Abstract
Background. Inflammatory bowel disease (IBD), which includes ulcerative colitis (UC) and Crohn’s disease (CD), is a chronic relapsing disease indicated by inflammation of the gastrointestinal tract. Celiac disease (CeD) is a chronic autoimmune disease of the small bowel. The prevalence of CeD in IBD patients is unknown. Some studies have described the coexistence of the 2 diseases in the same patient. This study aimed to investigate the prevalence of CeD in Saudi Arabian children with IBD. Methods. We used a retrospective study design because data can be collected immediately and is easier to analyze afterward. The study was conducted on IBD patients in the Pediatric Gastroenterology Department at National Guard Hospital, Jeddah, Saudi Arabia. We enrolled Saudi patients aged between 1 and 18 years who had been diagnosed with IBD and CeD based on positive biochemical serology and histology from January 2011 to January 2020. We excluded patients with immunodeficiency disorders. Results. Among the 46 enrolled patients with IBD, CeD was identified in 4, and they did not develop any relapses. We discovered that the weight at IBD diagnosis improved significantly compared to current weight (P-value < .0001). We also discovered that the height at diagnosis of IBD improved significantly compared to the current height (P-value < .0001). Additionally, we found no significant associations between UC and CeD (P-value = 1), or CD and CeD (P-value = .625). Conclusion. No significant associations were evident between the prevalence of CeD and IBD. More prospective multicenter studies are needed to clarify the prevalence of CeD in children with IBD.
Introduction
Inflammatory bowel disease (IBD), which includes Crohn’s disease and ulcerative colitis, is a chronic and relapsing disorder that is characterized by inflammation of the gastrointestinal tract.1,2 In addition to the impact of IBD on the gastrointestinal tract, it is responsible for many extra-intestinal manifestations, including arthritis, ocular involvement, dermatologic and pulmonary manifestations, biliary tree complications, anemia, and thromboembolism. Celiac disease (CeD) is a chronic autoimmune disease of the small intestine, which presents with symptoms such as mucosal damage induced by gliadin, malabsorption, anemia, diarrhea, and growth retardation.3-5
In the United States, the number of people who have adopted a gluten-free diet (GFD) even without having an underlying diagnosis of CeD has increased dramatically with at least 0.5% of the US population following a GFD without having a confirmed diagnosis of CeD. 6 Even in the absence of CeD, gluten is thought to be linked to symptoms of diarrhea, abdominal pain, fatigue, and nausea, which has led to the introduction of a new condition known as non-celiac gluten sensitivity. In 2014, a cross-sectional study was carried out with 1647 patients with inflammatory bowel disease (IBD) in the United States using a GFD questionnaire. 7 In general, 65.6% of the patients who adhered to a GFD reported an improvement in their gastrointestinal symptoms, and 38.3% described fewer or less severe IBD flares. In the patients attempting a GFD at the time of the study, excellent adherence was linked with a significant improvement in fatigue. Tissue transglutaminase (tTG) is an autoantigen that plays a major role in the pathophysiology of CeD. The pathophysiology of CeD involves the deamidation or crosslinking of the post-translational adjustment of ingested gliadin peptides, which are one of the main components of the gluten protein. Thereafter, mucosal damage and inflammation occurs because these peptides have become toxic. 8
CeD is among the most common immune-mediated diseases. In the United States and Europe, the prevalence currently of CeD is about 1 in 100 individuals.9,10 Although it has not yet been studied extensively, its global prevalence is most likely similar.11,12 In the Kingdom of Saudi Arabia, the prevalence of CeD is not known because no national epidemiological studies have been carried out; however, local data have shown that it is a common disease.13,14 The prevalence of CeD in IBD patients is also not known. Some instances of the simultaneous coexistence of both diseases in the same patient have been described in the literature,1,15-19 and physicians generally suspect a link between CeD and IBD; however, no clear association has been established to date. A study conducted in Isfahan in 2012 showed that IBD and CeD were linked in some adults, and the researchers surmised that this association may affect the prognosis of IBD. 3 This hypothesis can be supported for 2 reasons: the prevalence of IBD and CeD 5 and the theoretically similar pathogenesis of the conditions given the interaction of genetic, immunological, and environmental factors (eg, gut flora, gastroenteritis).
The prevalence of CeD in patients with IBD is not clear. Several studies have described the coexistence of both diseases in the same family or even in the same patient.18,19 Alper et al 4 found no increase in the incidence of CeD in pediatric patients with IBD compared to those who did not have IBD and noted that false-positive tTG tests can occur in children with IBD. In Saudi Arabia, El Mouzan et al 20 showed that the incidence of IBD in Saudi pediatric patients is lower than that suggested in the Western literature where the trend has increased considerably over time. No studies have been conducted to date on the prevalence of CeD in children with IBD in Saudi Arabia. The aim of the study was therefore to examine the prevalence of CeD in Saudi children with IBD.
Methods
We used a retrospective study design because data can be collected immediately and is easier to analyze afterward. The study was performed on IBD patients at the Pediatric Gastroenterology Department at National Guard Hospital (NGH), Jeddah, Saudi Arabia. National Guard Hospital in Jeddah which has a bed capacity of 751, provides medical care services to the Saudi Arabian population in the Western Region of the country. In addition, the Gastroenterology Department in particular provides care services to patients who suffer from a variety of health problems, including gastrointestinal and hepatobiliary diseases. Furthermore, the National Guard Hospital in Jeddah has various medical departments that offer both patient care and training in, for example, internal medicine, endocrinology, and neurology. 21 The total number of the study population was 160 Inflammatory Bowel Disease (IBD) patients. The study participants were Saudi patients aged between 1 and 18 years, who had been diagnosed with IBD according to the PORTO criteria and CeD based on positive biochemical serology (TTG and IgA) together with histology. The PORTO criteria comprise a combination of symptoms suggestive of IBD, such as abdominal pain, diarrhea, rectal bleeding, and weight loss. 22 In addition, patients with IBD should undergo an ileocolonoscopy and upper gastrointestinal endoscopy. 22 Moreover, inflamed and non-inflamed mucosa biopsies should be taken from each segment of the gastrointestinal tract. 22 After that, we excluded patients with immunodeficiency disorders from our study. Using a consecutive non-probability sampling technique, we collected the data from the patients’ physical and electronic files to ensure they met the inclusion criteria.
All the IBD patients were recruited for this study based on computer data from January 2011 until January 2020. The variables obtained from each patient included demographic data such as their code number, age, gender, date of diagnosis, weight at diagnosis of IBD and current weight, height at diagnosis of IBD and current height, other relevant data such as drug history, levels of anti-IgA, anti-IgG, anti-gliadin IgA, anti-gliadin IgG, and vitamin D, and finally a current diagnosis of Crohn’s disease or ulcerative disease and IBD complications. The patients’ blood was extracted into a purple-top ethylenediaminetetraacetic acid tube for storage and to prevent clotting. The anti-tTG and total IgA levels, which are both used to test for disease activity, were then determined. If the serology was positive, 4 biopsies were obtained from the duodenum and 2 biopsies from the duodenal bulb and placed in formalin. The histopathological findings were assessed using MARSH, which is a classification system used to describe the stages of damage and histological changes in the small intestine observed under a microscope and to diagnose CeD in those with IBD. 23 After obtaining informed consent from the patients, information regarding their demographics, IBD complications, drug history, and disease severity were collected and recorded. The total IgA levels of all the patients were also measured. In those with normal IgA levels, the IgA tTG antibodies were assayed using an enzyme-linked immunosorbent assay (ELISA), which is a test that detects and measures antibodies in blood. In those with abnormal IgA levels, the total IgG level and IgG tissue TG were measured using the ELISA method. Those patients with increased IgA and/or IgG levels underwent an endoscopy, and 4 standard biopsies were obtained from the duodenum. These biopsies were evaluated by a pathologist who was not aware of the serology and pathology results. After confirmation of CeD in the IBD patients, we assessed the effect of CeD on the IBD course (ie, the number of medications the patient was taking, the number of relapses, and any surgical interventions the patient had undergone).
The assistant pathologist was prevented from seeing the early serology and pathology results to avoid introducing any form of bias. We also used the hybrid prevalence estimator formula created by Hedt and Pagano to help us best utilize the data from the convenience population. The improved efficiency translated into decreased sample sizes, which helped increase the frequency of the data collection to support the data and ultimately minimize any potential convenience sampling bias. 24
Data Analysis
The data were analyzed using JMP®, version Pro 15 (SAS Institute Inc., Cary, NC, 1989-2021). For precise results and inferences, some tests were applied using statistical measurements. In this study, several variables were included in the analysis. The mean and standard deviation were used for the parametric numerical variables, and the median, minimum, and maximum were determined for the non-parametric numerical variables. Percentages were used to obtain precise categorical and outcome variables. A confidence interval of 95% with a P-value of .05 was applied. To evaluate the association between IBD patients and CeD, a univariate analysis was conducted using Karl Pearson’s chi-squared test. Finally, a paired t-test was used to observe the association between the patients’ weight at diagnosis and their current weight and between their height at diagnosis and current height.
Ethical Approval and Informed Consent
The ethics committee of King Abdullah International Medical Research Center approved the study (SP20/119/J). Consent was also obtained for enrollment because this was a retrospective study. Informed consent was a requirement due to the retrospective nature of the study; however, the confidentiality of the data extracted from the patients’ files was assured. To maintain the patients’ privacy, all the patients’ medical record numbers were converted into serial numbers using a coding sheet that was accessible only by the principal investigator and saved in a separate document. The patients’ new serial numbers were subsequently used throughout the data analysis process. In addition, authorization to access the patients’ data in the spreadsheet was limited to the study supervisor, co-supervisor, and the research group members, all of whom were willing to maintain the confidentiality of the data. Finally, the principal investigator ensured that all the data (both the hard and soft copies) were stored within the premises of the National Guard Health Affairs and accessed by the research team only.
Results
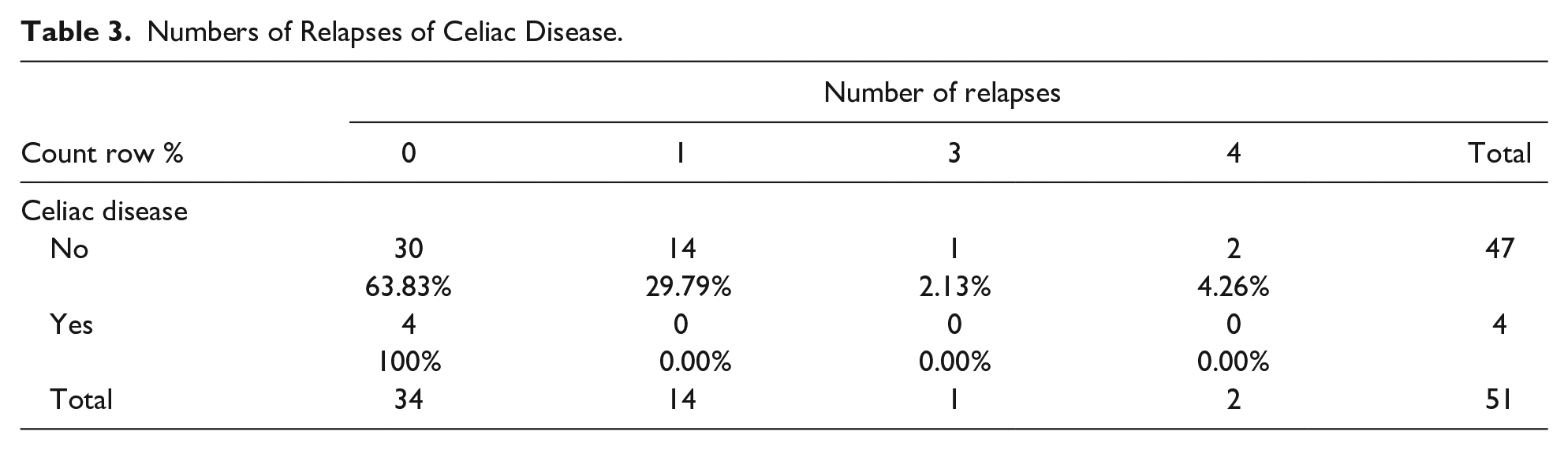
In total, 51 pediatric gastrointestinal patients were included in this study. The patients’ median age was 14 (5-18) years, current weight 36 (13.6-94) kg, and current height 147 (103.3-170) cm. The patients’ median vitamin D levels on first admission were 41.8 (20-225.6). The rest of the patients’ characteristics are shown in Table 1. About 31 (60.8%) patients were male. Among all the patients, 29 (56.9%) had Crohn’s disease, and 17 (33.3%) had ulcerative colitis. Of the 51 pediatric gastrointestinal patients included in this study, 19 patients had the anti-tTG test, and 7 tested positive. Of the 46 patients diagnosed with IBD, the highest number were diagnosed in 2016, 2017, and 2019 at 8 (17.4%) patients each year, while the lowest number were diagnosed in 2011 and 2014 at 2 (4.3%) each year. In terms of relapses per year, 34 (66.7%) patients had no relapses, 14 (27.5%) had 1 relapse, 1 (2%) had 3 relapses, and 2 (3.9%) had 4 relapses of the disease. The rest of the characteristics are shown in Table 2. Among the 51 patients, 4 patients had CeD and did not develop any relapses (Table 3). With respect to medication, azathioprine was being taken by the majority of patients with 33 (64.7%), while methotrexate was being used by the lowest number with 3 (5.9%). The other medications in use are presented in Graph 1. The patients’ current weight was significantly increased compared to their weight at the time of IBD diagnosis (P value < .0001). Similarly, their current height was significantly increased compared to their height at the time of IBD diagnosis (P value < .0001) (Table 4). Furthermore, ulcerative colitis was not significantly associated with CeD (P value = 1), Crohn’s disease was not significantly associated with CeD (P value = .625), and gender was not associated with age (P value = .322) (Table 5).
Quantitative Patient Characteristics.

Abbreviations: y, years; kg, kilogram; cm, centimeter; TTG, tissue transglutaminase antibody; CeD, celiac disease; IgA, immunoglobulin A; IgG, immunoglobulin G.
Qualitative Patient Characteristics.

Abbreviations: No, number; CeD, celiac disease; EGD, esophagogastroduodenoscopy.
Numbers of Relapses of Celiac Disease.
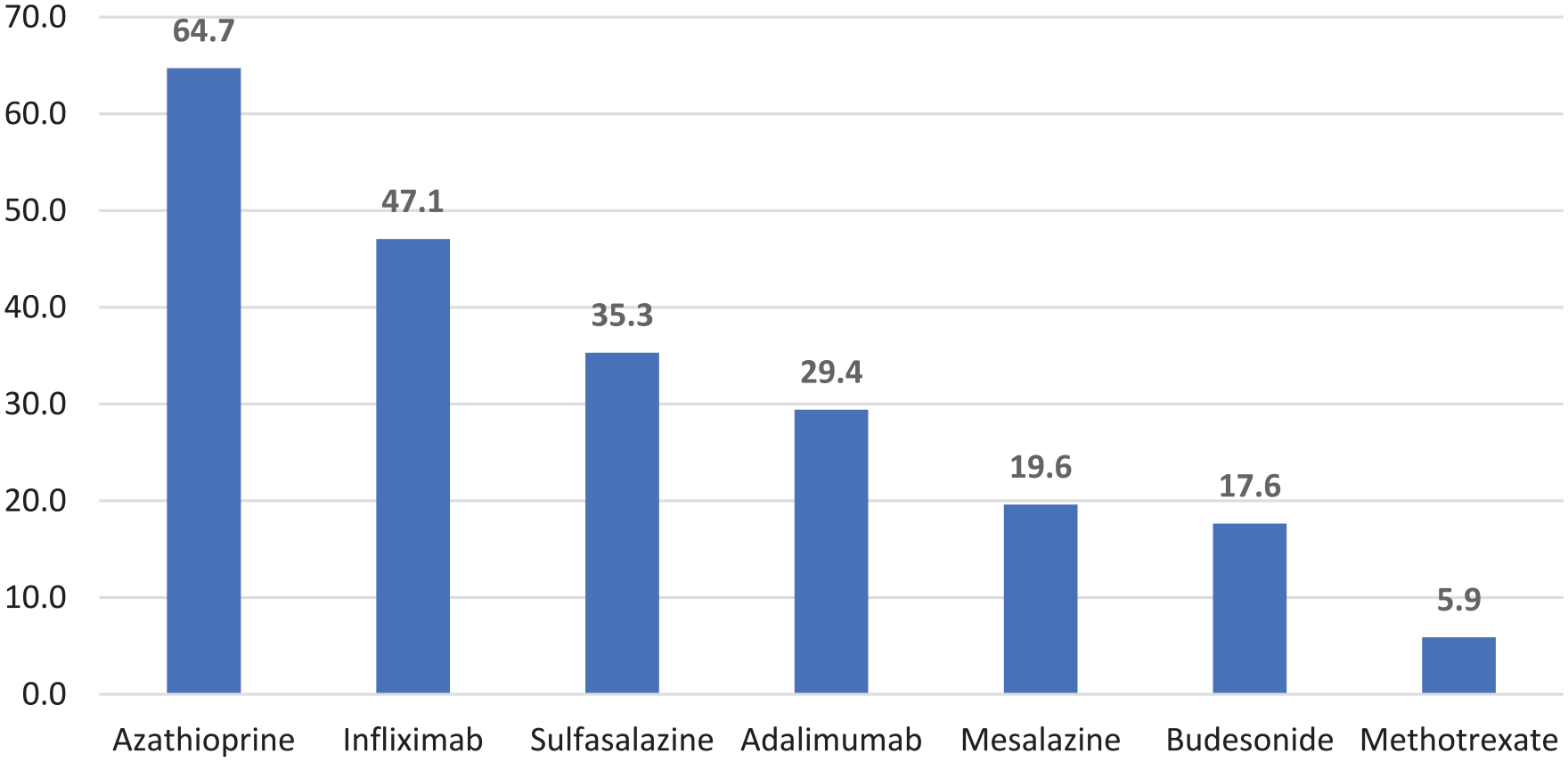
Medication taken for inflammatory bowel disease (%).
Comparison of Weight and Height at Diagnosis of IBD and Current.

Association Between CeD and IBD.

Discussion
In our study, 7.8% of the patients with IBD had CeD that was confirmed by biopsy. Furthermore, 3 (75%) patients had Crohn’s disease, and 1 (25%) patient had ulcerative colitis. Our findings suggest that the relationship between IBD and CeD was not significant. Likewise, neither ulcerative colitis nor Crohn’s disease were found to be associated with CeD in a statistically significant way. An increase in the patients’ weight and height following a diagnosis of IBD was noted. In our study of IBD patients at the National Guard Hospital in Jeddah, we found that ulcerative colitis was not significantly associated with CeD (P value = 1), and Crohn’s disease was not significantly associated with CeD (P value = .625). Similarly, Alper et al 4 did not find an increased prevalence of CeD among the IBD patients in their study. However, our results were contrary to those of Tavakkoli et al 3 who suggested a higher prevalence of CeD among IBD patients than in the general population. While Alper et al 4 enrolled 130 IBD patients in their study, we had only 46 IBD patients, yet our results showing no significant association between CeD and IBD were similar. Notwithstanding, the differences between our results and those of other studies could be accounted for by the participants’ origin. The participants in our study were from Saudi Arabia as no studies had yet been conducted to determine the association between CeD and IBD in this patient population. Moreover, our study type was cross-sectional, which is not as strong as the cohort that was used, for example, by Yang et al 1 According to our results, neither ulcerative colitis nor Crohn’s disease was significantly associated with CeD, so the existing treatment of the patients with CeD was not modified. IBD patients should take anti-inflammatory drugs, and CeD patients should follow a gluten-free diet (GFD).
Our methodological choices were constrained by our use of non-computerized medical records as they did not include all 63 variables. We could not assess the effects of immunomodulatory therapies for IBD, which are also used to ameliorate CeD symptoms and may result in CeD becoming undetectable. Our study may have been further limited by its retrospective design, the small number of IBD patients, the absence of a control group of non-IBD children, and the lack of long-term monitoring following the introduction of a GFD.
When comparing our study to others, we found some similarities in the way the data were collected and the final results. For example, Jandaghi et al 25 used the same methods to confirm the diagnosis of CeD. They also assessed the total IgA and IgA tTG antibodies to diagnose CeD in IBD patients, and in the case of IgA deficiency, the total IgG and IgG tTG were measured instead. 25 The results of Jandaghi et al’s 25 study also matched those of our study, which showed no significant increase in CeD among IBD patients compared to the general population. However, different studies from Spain and Turkey showed a significant association between the 2 diseases.26,27 This could be explained by the fact that those studies examined adult rather than pediatric patients and were conducted in different geographical locations.26,27 Compared to adults, the occurrence of CeD and IBD together in children is rare. 28 In a systematic review and meta-analysis of 9791 studies, patients with CeD were found to be at an increased risk of IBD and patients with IBD at an increased risk of CeD compared to other patient populations. 29 The actual prevalence of CeD based on serological and histological findings was reported to be 0.5% in 1711 adults with IBD in a multicenter study from Italy. 30 Similar to our findings, a study from Sweden reported a prevalence of 2.2% of CeD in 396 adults with IBD. 31 Virta and Kolho 32 studied 596 children with IBD in Finland and noted the prevalence of CeD in 2.2% of children with IBD compared with 0.7% prevalence in the non-IBD control group. In our study, CeD was present in 7.8% of the children with IBD. Tse et al 33 conducted a retrospective case-control study of adults with IBD and CeD. The patients with IBD and CeD had higher rates of primary sclerosing cholangitis (19.3% vs 5.7%; odds ratio, 4.4; 95% confidence interval, 2.1-9.4; P value < .001), extensive ulcerative colitis (78.1% vs 59.0%; odds ratio, 2.8; 95% confidence interval, 1.5-5.5; P value = .002), and a family history of CeD (10.5% vs 3.5%; odds ratio 3.2; 95% confidence interval, 1.3-8.2; P value = .01) compared to the patients without concomitant CeD. 33 We could not conduct a similar comparison in our study because of the absence of control patients. We used tTG to screen for CeD because it is the most specific test for the diagnosis of CeD. Although a high correlation exists between tTG and EMA antibodies, the highest specificity has been observed for EMA because tTG can be present in individuals with other conditions, including Crohn’s disease and ulcerative colitis. However, it is difficult to determine the frequency of tTG in IBD patients because studies developed with that aim have yielded different results, likely at least partially caused by the wide variety of commercial kits used.4,34 The main limitations of our study were thus the small sample size and the retrospective analysis. Additionally, the nature of our study did not allow us to examine the false-negative rates of tTG or the effects of a GFD on IBD. Theoretically, we may have missed children with IBD who also have CeD in the presence of normal tTG levels.
Conclusion
Our study showed that 7.8% of patients with IBD had biopsy-confirmed CeD, but the prevalence of CeD was not significantly associated with IBD. Similar to a number of studies on adults with IBD, we did not note an increased incidence of CeD among the pediatric patients with IBD in our study. Larger prospective multicenter studies are required to elucidate the prevalence of CeD in children with IBD.
Footnotes
Acknowledgements
We would like to thank the following doctors for helping us in our research paper: Dr. Mohammed Anwar Khan, Department of Medical Education, College of Medicine, King Saud bin Abdulaziz University for Health Sciences, Jeddah, Saudi Arabia. Dr. Ali Al-Zidan, Pediatric Gastroenterology Department, Ministry of the National Guard—Health Affairs, Jeddah, Saudi Arabia.
Author Contributions
Mohammed Hasosah contributed to the conceptualization and methodology of the study. Abdullah AlMansour, Anas Alamoudi, Mohammed Alghamdi, Faisal Alzahrani, Mamdouh Qadi, Sultan Alzahrani, and Bader Khawaji collected the data. Anas Alamoudi, Abdullah AlMansour, Mohammed Alghamdi, Faisal Alzahrani, Mamdouh Qadi, and Sultan Alzahrani wrote the first draft of paper. Mohammed Hasosah and Anas Alamoudi reviewed and edited the manuscript before submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
