Abstract
The use of low lactose formula (LLF) in term and near-term infants in infants with neonatal abstinence syndrome (NAS) has been increasing recently. However, the clinical evidence of such use is limited. Our aim in this paper was to systematically review the current literature about the use of LLF in infants with NAS. We searched PubMed, Embase, Cumulative Index to Nursing and Allied Health Literature, and Cochrane Database of Systematic Reviews for articles published between 2015 and 2020. Only randomized controlled trials, prospective, and retrospective studies. The risk of bias was assessed by using published tools appropriate for the study type. The certainty of the evidence was evaluated using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE). Forty-one titles and/or abstracts were screened independently by 2 reviewers (MA and GA). After an indepth review, 4 studies answered the study question (1 randomized controlled trial (RCT), 2 retrospective studies, and 1 quality improvement study). A meta-analysis could not be completed due to the study type difference and how the outcomes were reported. The studies found no benefit to feeding LLF to infants with NAS regarding short-term outcomes (length of stay, duration, and need for pharmacological therapy and growth). Certainty in the evidence is low. In conclusion we found no beneficial effects regarding the need for pharmacological therapy, duration of pharmacological treatment, length of hospital stay, and growth of using LLF compared to the standard formula in infants with NAS.
Background
Newborns exposed to various opioids and other illicit substances in utero as part of a treatment program or addictive behavior are at risk of withdrawal and development of neonatal abstinence syndrome (NAS) due to discontinuation of exposure after birth. The type and intensity of withdrawal depend on many factors, including but not limited to the substance used, gestational age at birth, polysubstance abuse, breastfeeding, and genetic polymorphisms. 1 There has been a national increase in NAS incidence in the last decade. This incidence increase has resulted in a significant increase in the admissions to neonatal intensive care units and pediatric floors. A study that included 28 states reported a 300% increase in NAS diagnoses from 1999 to 2013. 2 The care and extended hospital stay for each patient with NAS can cost the health care system up to $53 400 US dollars. 3 The rise in incidence and the resulting medical and financial burdens have been substantial driving factors for studying this condition’s evaluation and management. 4
Treatment of symptoms associated with NAS is generally based on a combination of pharmacological and non-pharmacological measures. Multiple pharmacological treatment protocols are followed by using morphine, methadone, or other medications. 1 However, starting medications for NAS treatment significantly increases the length of stay (LOS). 5 For that reason, infants with mild to moderate NAS are most often managed with non-pharmacological measures, and most practices try to avoid pharmacological therapy. Research has recently focused on non-pharmacological factors, such as withdrawal scoring systems, rooming in with mother, minimizing surrounding environmental noise, nutritional management, gentle handling, on-demand feeding, swaddling, music and massage therapy, and skin-to-skin care with caregivers.1,6
The literature has clearly shown that maternal breastmilk is beneficial for infants with NAS and significantly decreases the need for pharmacological therapy and the LOS. 6 However, the evidence is less clear regarding the type of formula to be used if maternal breastmilk is unavailable or contraindicated. Given gastrointestinal (GI) system involvement of NAS, a recent trend has been observed of using low lactose formula (LLF) in infants with NAS.7,8 The non-evidence-supported use of these formulas is based on the hypothesis that partially hydrolyzed protein and low/free lactose carbohydrate decrease feeding intolerance and fussiness associated with NAS.
Methods
In this systematic review, we aimed to understand whether the use of LLF decreases the severity and the duration of NAS symptoms among term and near-term infants (≥35 weeks of gestation) with NAS compared with infants who received regular standard formula (RSF).
Inclusion Criteria
Studies that were eligible for inclusion included those that investigated the effect of using LLF in term or near-term infants with NAS diagnaosis. In the included studies, the authors compared the outcomes regarding the need for pharmacological therapy for NAS, duration or cumulative dose of pharmacological treatment, length of hospital stay, and effect on growth between the infants who received LLF and those who used RSF. Randomized controlled trials (RCTs), cohort, case-control observational, and quality improvement studies were included in the search. Case reports and case series were excluded. All written languages were accepted if an English abstract was provided.
Outcomes
The 4 outcomes analyzed in this systematic review included the need for pharmacological treatment, pharmacological treatment duration, length of hospitalization, and effect on growth if studied.
Literature Search
A medical librarian (KS) performed the search on December 22, 2020, of the available databases (PubMed, Embase, Cumulative Index to Nursing and Allied Health Literature, Cochrane Database of Systematic Reviews). A search method was performed from the year 2015 to 2020 utilizing the use of keywords and headings ((“Neonatal Abstinence Syndrome/diet therapy”) OR ((“Neonatal Abstinence Syndrome” OR “neonatal abstinence syndrome”) AND (“Infant Formula” OR “Enteral Nutrition” OR low lactose formula OR lactose-free formula OR protein partially hydrolyzed formula OR formula OR formula OR Feeds OR Feeding OR Enteral))) from the year of 2015 to 2020.
Study Selection and Extraction
Titles and abstracts were independently screened by 2 authors, MA and GA, who screened the full-text articles. Discrepancies were discussed and resolved after agreement with all investigators.
The Certainty of the Evidence, Risk of Bias Assessment, and Data Synthesis
GRADE was used to assess the overall certainty of the evidence. 9 The Cochrane Collaboration Risk of Bias Tool was used to evaluate the RCT. 10 A meta-analysis could not be completed due to the difference in the study type and statistical heterogeneity of the outcomes.
Results
Study Selection
Among 41 articles selected, 4 met the inclusion criteria: 2 cohort studies, 1 randomized controlled trial, and 1 quality improvement study, Figure 1.11-14 The inclusion of 1 other study was ruled out after discussion and agreement between the authors. 15 Study types, methodology, and limitations are shown in Table 1.

Prisma flow chart.
Summary of the Type and Methodology of Included Studies.
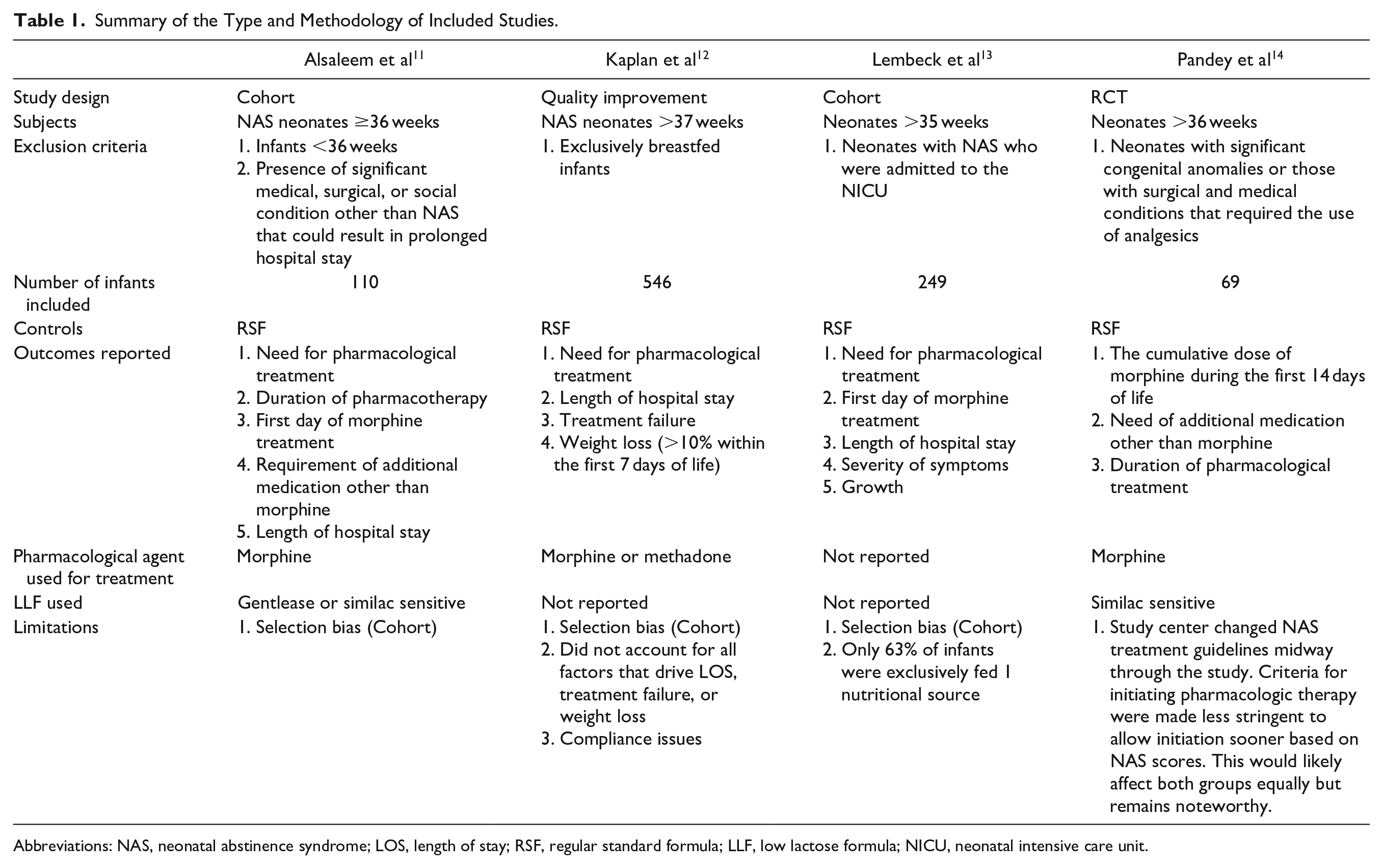
Abbreviations: NAS, neonatal abstinence syndrome; LOS, length of stay; RSF, regular standard formula; LLF, low lactose formula; NICU, neonatal intensive care unit.
The 4 studies were from different institutions. The studies used 3-arm comparisons between LLF, RSF, and human milk. For this review’s specific aim, we focused mainly on whether using LLF was more beneficial regarding short-term outcomes in infants with NAS. Alsaleem et al 11 and Pandey et al 14 reported the type of LLF (Similac Sensitive (Abbott, Columbus, OH), and/or Enfamil Gentlease (Mead Johnson & Co., Glenview, IL)) that was used in the analysis. The other 2 studies did not specifically mention the kind of LLF used in the study population.12,13
Morphine was the most common pharmacological therapy used in the studies included.11,12,14 Lembeck et al 13 did not specifically mention which medication was used for treatment. The quality improvement project reported the use of morphine in most intensive care units, with a small number of newborns treated with methadone. 12 The pharmacological therapy needs assessment was based on either the pharmacological therapy duration or the cumulative dose of morphine administered during the treatment duration. The risk of bias analysis for the RCT is shown in Table 2.
Risk of Bias Analysis for RCT Performed by Pandey et al 14 .

Results by Outcomes
Duration of pharmacological therapy
Three studies (Alsaleem et al, 2020; Lembeck et al, 2020; Pandey et al, 2020) measured the duration of treatment in infants with NAS (N = 428). Alsaleem et al measured the duration of morphine sulfate (MSO4) treatment in infants fed LLF versus RSF (n = 110). After adjusting for the type of drug used by the mother, regular maternal involvement in prenatal care, maternal smoking status, and maximum scores prior to MSO4 treatment, the intervention (LLF) was not different from RSF. 11 Lembeck et al 13 measured the duration of therapy in infants fed LLF versus RSF (n = 129). The authors found that the intervention (LLF) was not different from RSF. Pandey et al measured medication treatment duration in infants fed LLF versus infants fed RSF (n = 69). The mean difference of treatment duration in days indicated the intervention (LLF) was not different from RSF. 14
The certainty of the evidence for the duration of treatment
The certainty of the body of evidence was very low. The body of evidence was assessed to have a risk of bias, inconsistency, and imprecision. The risk of bias was assessed as serious as 2 of the studies (Alsaleem et al 11 and Lembeck et al 13 ) were retrospective cohorts, resulting in selection bias. Inconsistency was serious due to the heterogeneity of formulas used, calorie levels, and feeding type exclusivity. Imprecision was assessed as serious due to the low number of events.
Length of Stay
Three studies measured LOS in infants with NAS diagnosis (N = 905).11-13 Alsaleem et al 11 measured LOS in infants fed LLF versus RSF (n = 110). After adjusting for the type of drug used by the mother, regular maternal involvement in prenatal care and inborn status, smoking status, and maximum scores prior to MSO4 treatment, the intervention (LLF) was not different from RSF.11,13 Lembeck et al 13 measured LOS in infants exclusively fed LLF versus those who were fed RSF. The intervention (LLF) was not different from RSF. Kaplan et al measured LOS in infants fed low lactose high calorie (LL + HC) versus high lactose high calorie (HL + HC) versus low lactose low calorie (LL + LC) versus high lactose + low calorie (HL + LC) formula. The mean days indicated that the intervention (LL + HC) was less effective compared to HL + HC; however, the significance was not reported. LOS for LL + HC versus HL + HC was 16.6 and 14.8 days, respectively. 12
The certainty of the evidence for LOS
The certainty of the body of evidence was very low. The body of evidence was assessed to have a serious risk of bias, serious inconsistency, and serious imprecision. The risk of bias was assessed as serious as 2 of the studies (Alsaleem et al 11 and Lembeck et al 13 ) were retrospective cohorts, resulting in selection bias. Also, the risk of bias was serious as Kaplan et al’s 12 study was a quality improvement study that did not report significance. Inconsistency was serious due to the heterogeneity of formulas used, calorie levels, and feeding type exclusivity. Imprecision was assessed as serious due to the low number of events.
Need for pharmacological therapy
Three studies (Alsaleem et al, 11 Lembeck et al, 13 and Pandey et al 14 ) measured the need for pharmacologic treatment in infants with NAS (N = 428). Pandey et al 14 indicated that the intervention (LLF) was not different compared to RSF. Cumulative morphine for the first 14 days was 20.7 ± 19.8 and 23 ± 23.5 mg for LLF and RSF, respectively. Alsaleem et al measured the need for pharmacological therapy in infants fed LLF versus standard term formula (n = 110). After adjusting for the type of drug used by the mother, regular maternal involvement in prenatal care, smoking status, and maximum scores prior to MSO4 treatment, the intervention (LLF) was not different RSF. 11 In Lembeck et al, 13 the authors found that the intervention (LLF) was not different from RSF.
The certainty of the evidence for pharmacological therapy
The certainty of the body of evidence was very low based. The body of evidence was assessed to have a serious risk of bias, serious inconsistency, and serious imprecision. The risk of bias was assessed as serious, as 2 of the studies (Alsaleem et al 11 and Lembeck et al 13 ) were retrospective cohorts, resulting in selection bias. Inconsistency was serious due to the heterogeneity of formulas used, calorie levels, and feeding type exclusivity. Imprecision was assessed as serious due to the low number of events.
Growth
One study (Lembeck et al) measured growth in infants exclusively fed LLF versus RSF (n = 129). The authors reported that the intervention (LLF) was not different from RSF. 13
The certainty of the evidence for growth
The certainty of the body of evidence was very low. The body of evidence was assessed to have a serious risk of bias and serious imprecision. The risk of bias was assessed as serious, as the study (Lembeck et al) was a retrospective cohort, resulting in selection bias 13. Imprecision was considered serious due to the low number of events. As only 1 study (Lembeck et al 13 ) was identified to assess the effect of LLF on growth, consistency could not be assessed. More detailed information and statistical analysis of the included studies’ outcomes are shown in Supplemental Table 1.
Discussion
Our goal was to evaluate the possible short-term beneficial effects of using LLF in term or near-term neonates with NAS in this review of the current literature. Our systematic review of 4 studies found no benefit regarding short-term outcomes (the need for pharmacological therapy, duration of pharmacological treatment, LOS, and growth) using LLF compared to RSF in infants with NAS. Overall, the use of LLF did not improve the outcomes in neonates with NAS.
LLFs are different from RSF by having reduced or absent lactose content in addition to the presence of partially hydrolyzed protein components. Different brands are available on the market, such as Similac Sensitive, Enfamil Gentlease, and Gerber Good Start Gentle. 16 The carbohydrate source in these formulas is usually derived from corn syrup solids compared to the standard human or cow milk that is mostly lactose derived. The reduced amount of lactose in these formulas can potentially interfere with the normal biological functions in neonates because lactose is an essential energy source in neonates and plays a role in calcium absorption and innate immunity.17-20 Another potential drawback of these formulas is derived from the fact that the partially digested proteins do not have a similar trophic effect on the bioactive peptides and enzymes required for the normal digestive function in comparison to intact proteins present in humans or cows milk.21,22 Studies have shown that, in addition to the interference with these important biochemical functions, LLF has a bitter taste due to the partially hydrolyzed proteins when compared to RSF. 23
The LLF is usually an alternative to RSF for infants who experience feeding intolerance, severe colic symptoms, or reflux episodes. As NAS symptoms involve the GI system, these formulas have become an attractive option for infants with NAS to decrease GI symptom severity. Although evidence of such use lacked before 2019, the increasing trend of using these formulas has allowed researchers to evaluate this intervention’s evidence-based practice. We found 2 cohort studies, 1 quality improvement project, and 1 double-controlled randomized trial performed recently that have shed light on the use of LLF in infants with NAS. We speculate that the results reported in this review originated from the pathophysiological mechanism associated with GI withdrawal symptoms seen in infants with NAS. Holzer showed that the effects of opioids on the GI tract are caused primarily by altering the acetylcholine neurotransmitter and receptor interaction. The reduced amount of lactose or the partially hydrolyzed proteins in LLF is unlikely to affect the signs and symptoms associated with the disturbance of the acetylcholine neurotransmitter pathway. 24
Although assessing the effect of maternal breast milk (MBM) on NAS outcomes was not the purpose of this review, we found that the authors of the 4 studies included in this systematic review found a consistent beneficial effect of MBM regarding the short-term outcomes (need for pharmacological therapy, duration of pharmacological treatment, and hospital LOS). This observation is consistent with established evidence from previous studies showing favorable outcomes of infants with NAS who received human milk.25,26
The strengths of this systematic review include the detailed literature search and assessment of the certainty of evidence using GRADE and the detailed analysis of the specific outcomes targeted in this review. Limitations include the small number of eligible studies and the significant heterogeneity between the included studies regarding outcomes reported. The lack of details about the percentage for the specific formula intake and the slightly different treatment protocols followed in each of the studies were additional limiting factors for which we could not control in this systematic review. Given the theoretical adverse effects of using LLF that may compromise calcium absorption and digestive function, we remain concerned about the lack of clear evidence for the use of LLF in infants with NAS.
Conclusion
From the limited evidence currently available, we did not find beneficial effects regarding the need for pharmacological therapy, duration of pharmacological treatment, LOS, and growth of using LLF compared to other standard formulas in infants with NAS. A conditional recommendation is made against LLF, as no beneficial effects were found in patients with NAS. This recommendation is based on expert opinion and a review of the current literature. The overall certainty of the evidence is low.
Supplemental Material
sj-pdf-1-gph-10.1177_2333794X211035258 – Supplemental material for Effect of Low Lactose Formula on the Short-Term Outcomes of Neonatal Abstinence Syndrome: A Systematic Review
Supplemental material, sj-pdf-1-gph-10.1177_2333794X211035258 for Effect of Low Lactose Formula on the Short-Term Outcomes of Neonatal Abstinence Syndrome: A Systematic Review by Mahdi Alsaleem, Jarrod Dusin and Gangaram Akangire in Global Pediatric Health
Footnotes
Acknowledgements
The authors would like to extend a sincere thanks to the following: K. Swaggart, MLIS, AHIP, for performing the literature search. Office of Evidence-based Practice at Children’s Mercy Hospital for performing the data analysis. Medical Writing Center at Children’s Mercy Hospital for editing this manuscript.
Author Contributions
MA: Contributed to design, research, acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
GA: Contributed to conception, design, analysis, and interpretation; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work, ensuring integrity and accuracy.
JD: Contributed to analysis and interpretation; critically revised manuscript; gave final approval; and agrees to be accountable for all aspects of work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
