Abstract
This systematic review/meta-analysis aims to highlight the effect of vitamin D supplementation in deficient children suffering from obesity. Published clinical studies on vitamin D supplementation in obese children and adolescents with vitamin D deficiency were identified through a comprehensive MEDLINE/PubMed search (from July 1966 to November 2017). Outcomes intended after vitamin D supplementation were improvements in vitamin D status, BMI alterations and appetite changes. The inclusion criteria were children aged 2 to 18 years of both sexes in clinical trials that specified the oral and/or intramuscular dose of vitamin D supplementation. Ten studies were retrieved, but only 6 were relevant. First, supplemented obese children and adolescents were compared to non-obese controls; thereafter, supplemented obese children and adolescents were compared to matching obese peers given placebo. Pooled risks from the 2 studies that evaluated the number of obese and non-obese children and adolescents who improved upon vitamin D supplementation revealed that obesity poses a risk for not benefiting from the vitamin D supplementation regardless of the dose and the duration of supplementation. Pooled results from the 6 retrieved studies that compared supplemented obese children and adolescents to matching non-obese or obese peers given placebo revealed significantly lower vitamin D levels in obese participants than in non-obese peers. Vitamin D levels are significantly lower in obese children and adolescents with obesity, posing a risk for not benefiting from vitamin D supplementation regardless of the dose and duration of supplementation.
Introduction
Both childhood obesity and vitamin D deficiency are common in the Middle East. Additionally, the body adiposity index is significantly associated with 25 hydroxy vitamin D [25(OH)D] levels in Arab children. 1 Among the possible determinants of low vitamin D status in preschool children in the Makkah region were high BMI, inadequate vitamin D intake, and low levels of outdoor physical activity. 2
As early as intrauterine life, this problem may show; vitamin D deficiency in pregnancy may increase the risk of prenatal and early postnatal overweight offspring. 3 There is increasing evidence of an association between low vitamin D and a range of non-bone health outcomes; however, there is a lack of data from robust randomized controlled trials on vitamin D supplementation. 4 Williams et al 5 concluded that obese children should be considered for routine vitamin D screening.
Recently, it was suggested that vitamin D supplementation may enhance weight reduction and prevent bone loss in obese children with this deficiency. 6 Brinkmann et al 7 warned against weight excess, which interferes with the response to vitamin D supplementation, leading to an attenuated increase in 25(OH)D even with supplementation.
On the basis of a bi-directional genetic approach that limits confounds, it was previously concluded that a high BMI leads to low 25(OH)D, while any effects of low 25(OH)D increasing BMI are likely to be small. 8 The latter study further added that population-level interventions to reduce BMI are expected to decrease the prevalence of vitamin D deficiency.
Our systematic review/meta-analysis aims to highlight the effect of vitamin D supplementation in deficient children suffering from obesity. Comparing different results of supplementation between children and adolescents and between obese versus overweight individuals would be a secondary objective.
Methodology
Published clinical studies on vitamin D supplementation in obese children and adolescents with vitamin D deficiency were identified through a comprehensive MEDLINE/PubMed search (from July 1966 until November 2017) using a variety of Medical Subjects Headings and free text words: (vitamin D AND supplementation AND clinical trials AND children AND obesity). We conducted additional searches of Cochrane Central Register of Controlled Trials, Science Direct, Scirus, ISI Web of Knowledge, Trip Database, Google Scholar, previously published reviews, examination of cited reference sources, and personal contact and discussion with several investigative experts in the field. Outcomes intended after vitamin D supplementation were improvements in vitamin D status, BMI alterations and appetite changes.
The inclusion criteria were children over the age of 2 years and up to the age of 18 years of both sexes who participated in clinical trials that specified the oral and/or intramuscular dose of vitamin D supplementation. Exclusion criteria were studies conducted on children with chronic or metabolic diseases or those conducted on children receiving drugs, fortified foods, or vitamin D-enriched foods that could affect vitamin D status. Additionally, trials that did not specify the age of the participants or the dose for vitamin D supplementation were also excluded.
Abstracts of articles identified using the search strategies previously mentioned were viewed, and articles that appeared to fulfill the inclusion criteria were retrieved in full. Studies reporting data on at least one of the outcome measures were included in the study.
Each article identified was reviewed and categorized into one of the following groups:
included: randomized controlled clinical trials (RCTs) or controlled clinical trials (CCTs) that met the described inclusion criteria and those where it was impossible to tell from the abstract, title or MeSH headings;
excluded: non-RCT and non-CCT studies.
Outcomes from included trials were combined. Reasons for heterogeneity of studies were explored, and if necessary, sensitivity analysis was performed on the basis of methodological quality and random effects versus fixed effects modeling. Subgroup analyses were based on age (children or adolescents), obesity or overweight classifications and duration and dose of vitamin D supplementation.
All statistical analyses for pooling the studies were performed on STATA statistical software, release 14.0 (Stata Crop. 2015, College Station, Texas, USA).
Results
Ten studies were retrieved, but only 6 were relevant. Further classification was performed in meta-analysis I, where supplemented obese children and adolescents were compared to non-obese controls (Table 1), and meta-analysis II, where supplemented obese children and adolescents were compared to matching obese peers given placebo (Table 2).
Methodology of the 2 Studies that Compared Obese to Non-Obese Children and Adolescents in Meta-Analysis I.

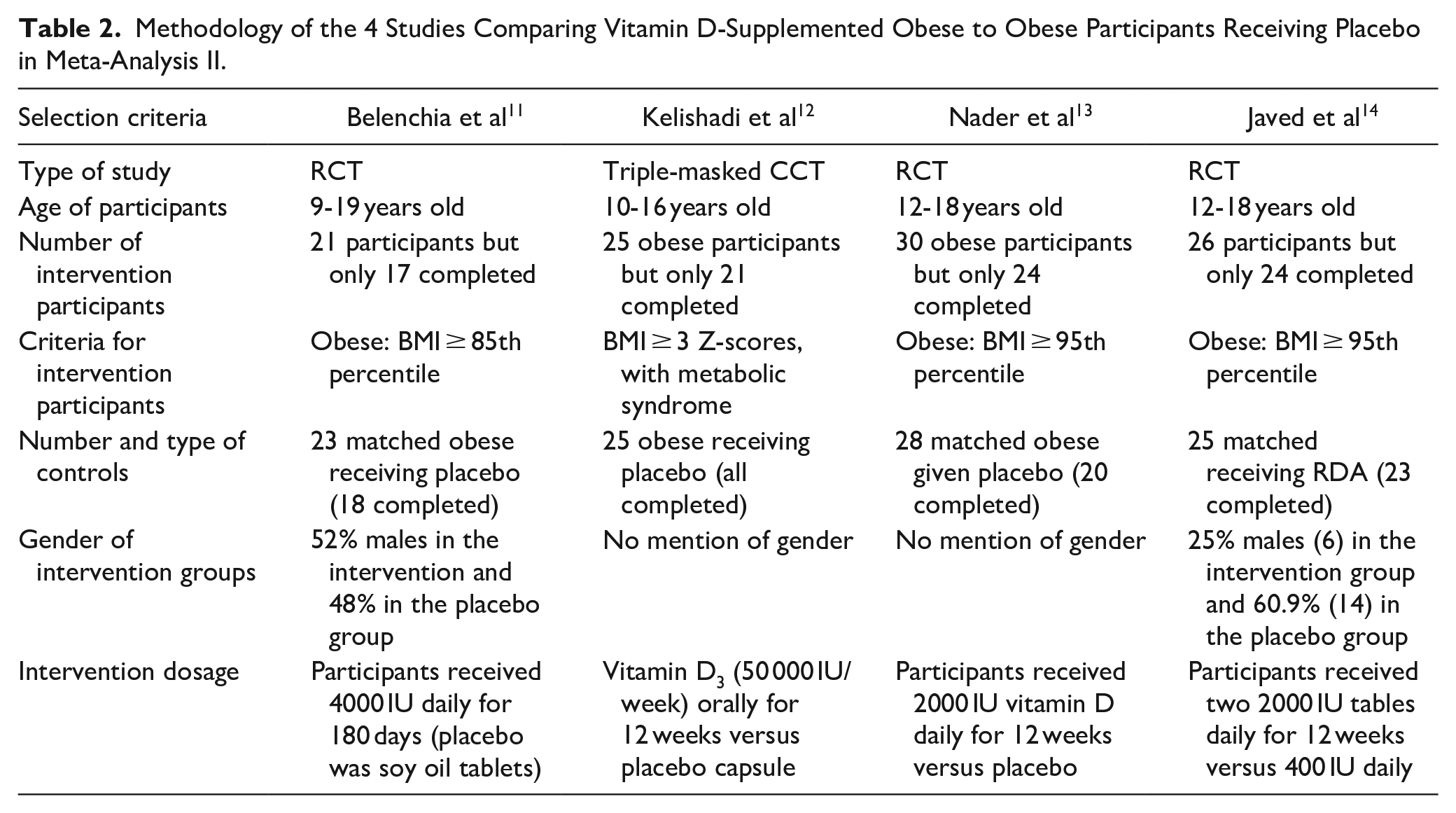
Methodology of the 4 Studies Comparing Vitamin D-Supplemented Obese to Obese Participants Receiving Placebo in Meta-Analysis II.
Pooled risk from the 2 studies in meta-analysis I that evaluated the number of obese and non-obese children and adolescents who improved upon vitamin D supplementation revealed that obesity poses a risk for not receiving benefit from the vitamin D supplementation regardless of the dose and duration of supplementation (Table 3). Pooled results of baseline vitamin D levels in the non-obese and obese participants are illustrated in Tables 4 and 5, respectively. Additionally, Table 6 shows the comparison between the 2 groups of pooled results from the 6 studies, which revealed significantly lower baseline vitamin D levels in obese participants than in non-obese peers.
Pooled Results of the 2 Studies that Evaluated the Number of Obese and Non-Obese Children and Adolescents Who Improved Upon Vitamin D Supplementation.

Pooled Results for Baseline Vitamin D Levels in Non-Obese Children and Adolescents.

Pooled Results for Baseline Vitamin D Levels in Obese Children and Adolescents.

Comparison between Pooled Levels of Baseline Vitamin D in Obese and Non-Obese Children and Adolescents.

Regarding other outcome measures, that is, BMI or appetite changes, the data were minimal and inconsistent in the included studies, hindering further analysis. Subgroup analyses for age and gender could not be performed because neither of the 2 studies mentioned vitamin D values in these categories subgroups.
Regarding vitamin D levels in meta-analysis I, Castaneda et al 10 reported that vitamin D supplementation significantly increased vitamin D levels in non-obese compared to obese participants, which was not the case in the study of Rajakumar et al. 9 In meta-analysis II, there were reports of a significant increase in post-intervention vitamin D levels compared to the baseline only in the groups receiving vitamin D in the studies of Nader et al 13 and Javed et al, 14 Belenchia et al 11 reported significant differences when comparing the post-intervention vitamin D values between the supplemented and non-supplemented obese children and adolescents together with a significant increase in the post-intervention values compared to the baseline values within the supplemented group only. Regarding the fourth study, the only significant differences reported were those comparing the post-intervention vitamin D values between the supplemented and non-supplemented obese children and adolescents. 12
Discussion
Methodological concerns of any meta-analysis include limitations in the quality of the pooled data, as the usefulness of any systematic review largely depends on the quality of the individual included studies. Pooling results of interventional studies, as in the current study, provides more evidence than does pooling results from observational studies.
Our comprehensive search for vitamin D supplementation identified only 2 studies that performed supplementation trials in obese children and adolescents in comparison to non-obese matching controls (meta-analysis I). This small number of interventional studies does not usually permit meta-analysis studies to obtain reliable results. Nevertheless, the pooled risk revealed that while evaluating the number of obese and non-obese children and adolescents who improved upon vitamin D supplementation was close to the relative risk in each of the studies of Rajakumar et al 9 and Castaneda et al 10 when calculated separately. This result proves that obesity poses a risk for not receiving benefit from vitamin D supplementation regardless of the dose and duration of supplementation. This point further proves the conclusion of the clinical trial of Castaneda et al 10 who reported that higher doses of vitamin D are required to treat vitamin D deficiency in obese adolescents than doses required in their non-obese peers. This finding is also reinforced by Peterson, 15 who mentioned in her review that the vitamin D needs of obese children are greater than those of non-obese children.
The previously mentioned pooled risk result is consistent with Saliba et al, 16 who reported that BMI is inversely associated with the increase in serum 25(OH)D levels in response to vitamin D supplementation in the adult population. Additionally, Harel et al 17 reported that although there was a significant increase in mean 25(OH)D after the initial course of treatment with vitamin D, 25(OH)D levels were normalized in only 28% of their series of obese adolescents. The latter authors further added that repeat courses with the same dosage in the other 72% of participants did not significantly change their low vitamin D status.
Meta-analysis II comprised 4 clinical trials in which vitamin D supplementation was given to 1 group of obese children and adolescents in comparison to another obese group given placebo or the recommended dietary allowance (RDA) of vitamin D. Nevertheless, the dosage of vitamin D given as supplementation was not consistent among the 4 studies and neither was the supplementation period. The significant increases in vitamin D levels detected in meta-analysis II, whether within the supplemented groups or between the supplemented and non-supplemented groups reflect the ability of supplementary vitamin D doses to raise the 25(OH)D levels in obese children and adolescents, although the data cannot be pooled due to the diversity in supplementation methodology. However, the proper dose and duration cannot be deduced because several of these studies reported a portion of vitamin D-deficient obese children and adolescents at the end of their supplementation period. For instance, the study of Belenchia et al, 11 which performed the longest supplementation period, reported that only 93% of those receiving vitamin D supplementation became sufficient after 6 months. A more alarming number was reported by Javed et al, 14 who found that 75% of their vitamin D-supplemented children and adolescents did not reach vitamin D sufficiency by the end of the 12-week supplementation trial.
Of the 6 included studies, only 310,13,14 gave vitamin D supplementation doses according to the guidelines of 1200 to 2000 IU daily for obese pediatric patients (Cediel et al, 2018). 18
The pooled values of baseline vitamin D in non-obese versus obese children and adolescents showed significantly lower baseline values of vitamin D in obese children and adolescents than in non-obese controls. This result is contrary to both studies of meta-analysis I, which reported non-significant differences in baseline vitamin D levels among the non-obese and obese children and adolescents.9,10 This pooled result, however, is consistent with many previous worldwide studies that noted the decreased 25(OH)D levels that occur in obese adolescents compared to those normal weight controls.19-21 Lower concentrations of serum 25(OH)D levels in obese subjects may be explained by enhanced uptake by adipose tissue and increased metabolic clearance, and it is suggested that the sedentary lifestyle of obese subjects could be associated with less outdoor activity and less exposure to sunlight (Wortsman et al, 2000 22 and Bolland et al, 2007 23 ).
None of the studies in meta-analyses I or II mentioned any changes in BMI over the supplementation period. This finding may explain the decreased response to vitamin D supplementation in obese children and adolescents if we consider the hypothesis of vitamin D sequestration in adipose tissue because of its lipid-soluble nature, as proposed by Wortsman et al, (2000). Although the clinical trial of Mason et al 24 deduced that vitamin D3 supplementation during weight loss did not increase weight loss or associated factors compared with placebo, Mason et al 25 reported that a greater degree of weight loss, achieved through either a reduced-calorie diet or increased exercise, is associated with increased circulating 25(OH)D concentrations in older women. Thus, it is prudent to hypothesize that if the studies in both meta-analyses I and II had performed simultaneous weight adjustment strategies, whether by diet and/or exercise, more of the enrolled children and adolescents would have reached vitamin D sufficiency by the end of the supplementation trial coupled with the expected and favorable changes in BMI.
Subgroup analyses for age and gender could not be performed because neither of the 2 studies of meta-analysis I nor the 4 studies of meta-analysis II mentioned vitamin D values in these subcategories of studied participants. Nevertheless, a British study that included 711 788 children aged 0 to 17 years reported that diagnosis rates of vitamin D deficiency in children aged <5 years were higher in boys than girls, whereas in children aged ≥10 years, they were higher in girls. 26 Similarly, Al-Sadat et al 27 studied vitamin D deficiency among 1361 Malaysian students with a mean age of 12.9 ± 0.3 years and found that the deficiency was significantly higher in girls. The latter authors added that vitamin D deficiency in females showed an odds ratio of 8.98 (95% CI 6.48-12.45), and in adolescents with a greater waist circumference, the odds ratio was 2.64 (95% CI 1.65-4.25).
In conclusion, the pooled results of this meta-analysis reveal that vitamin D levels are significantly lower in obese children and adolescents compared to non-obese matching peers. The pooled risk result proves that obesity poses a risk for not receiving benefit from the vitamin D supplementation regardless of the dose or duration of supplementation. Vitamin D supplementation given to these vulnerable groups results in a significantly attenuated increase in hydroxy vitamin D levels with supplementation, suggesting a need for higher doses in this population. However, the proper dose and duration cannot be deduced because several of the studies in the current meta-analysis reported a portion of vitamin D-deficient obese children and adolescents at the end of their supplementation period. It is prudent to hypothesize that if the studies in the current meta-analysis had performed simultaneous weight adjustment strategies, more of their enrolled children and adolescents would have reached vitamin D sufficiency by the end of the supplementation trial coupled with the expected and favorable changes in BMI.
Based on the results of the current meta-analysis, we recommend that obese children and adolescents be screened for vitamin D status and that their supplementation should follow the guidelines. Simultaneous weight adjustment strategies are also advised during vitamin D supplementation for obese children and adolescents to better achieve vitamin D sufficiency.
Footnotes
Author Contributions
MFN: Substantially contributedto conception or design; Contributed toacquisition, analysis,or interpretation of data; Drafted the manuscript; Critically revisedthe manuscript for important Intellectual content; Gave final approval; Agree to be accountable for all aspects of the work in ensuring that questions relatingto the accuracy or integrity of any part of the workare appropriately investigated and resolved.
EKE: Contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
MFA: Contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted manuscript; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
