Abstract
Pediatric cancer is a life threatening disease known to create multi-organ complications that further compromise medical management affecting patient morbidity and mortality. Pulmonary hypertension (PH) is becoming more recognized as a complication of cancer and its therapies but has not been well characterized in pediatrics. Cancer pathophysiology can be uniquely set up to promote pulmonary vascular injury and remodeling that is similar to PH patients without cancer. This highlights the need to evaluate for PH clinically and with routine testing such as echocardiography during the course of a patient’s care even into adulthood. This review article will discuss the direct, indirect and therapy related aspects of cancer which can promote PH in these patients. This understanding is essential to target effective treatment options in a potentially fatal complication. Diagnostic and treatment algorithms are presented in relation to the most recent pediatric PH management guidelines.
Introduction
According to the American Cancer Society, pediatric cancer constitutes just 1% of all US cancers numbering about 16 000 cases a year with an annual incidence of about 190 cases per million children/adolescents. 1 Fortunately, the survivability of all cancers has improved to an average of 80% after 5 years, 1 however acute/early complications can occur as a direct result of the cancer itself or from early treatment side-effects involving any organ system. Long-term consequences of pediatric cancer are well documented and show a higher tendency to involve cancer reoccurrence, cardiovascular, and pulmonary sequela. 2 Pulmonary hypertension (PH) is a cardiovascular complication that has been associated with cancer especially during the treatment phase in adult 3 and pediatric4,5 patients with an estimated prevalence in 1 adult study of 15.4% 6 but the prevalence in pediatric cancer is not known. This review article will discuss the classification, pathophysiology and evaluation of PH in pediatric cancer which is needed to guide PH treatment in this population.
Pulmonary Hypertension Definition and Classification
PH is a general term for the abnormal elevation of pulmonary artery (PA) pressures which can lead to right heart failure. The yearly incidence and total prevalence of PH in pediatrics is about 4 and 20 to 50 cases, respectively per million children.7,8 With current PH therapies, the survivability of pediatric PH by the REVEAL registry is about 74% at 5 years for pulmonary arterial hypertension (PAH). 9 PH is generally considered a chronic and sometimes insidious disease that is progressive in nature without a cure. 10 However there are circumstances that can lead to more rapid progression of PH such as pulmonary embolism or interstitial lung disease. PH has been classified into 5 sub-categories in terms of mechanism of disease to aid in diagnosis and treatment (Table 1). 11 The latest classification system was updated in 2018 and includes a new hemodynamic definition of a mean PA pressure of >20 mmHg (Table 2). 11 There are several registries of pediatric pulmonary hypertension that describe PH occurrence by sub-category but specific etiology such as its association with cancer is not listed.8,9,12-15 PH associated with cancer and cancer therapy can fall into any of the 5 sub-categories based upon the effects on the pulmonary arteries, lung parenchyma, or heart. The etiology of PH associated with cancer can be due to several mechanisms which include direct injury, indirect injury and treatment related (Table 3).
Classification of PH by World Symposium on Pulmonary Hypertension 2018 and Associated Oncologic Conditions. 11

Abbreviations: PH, pulmonary hypertension; PTE, pulmonary thromboembolism; PTTM, pulmonary tumor thrombotic microangiopathy.
Associated with: connective tissue disease, HIV, portal hypertension, congenital heart disease, schistosomiasis.
Hemodynamic Classification of Pulmonary Hypertension. 11

Classification groups 1, 3, 4, 5.
Classification groups 2 and 5.
Classification groups 2 and 5.
Modes of Cancer Mechanism of PH Development.

Direct Injury Mechanism of Cancer Associated PH
Direct compromise of the PA’s and proximal airways can occur as a result of tumor mass effect or tumor invasion into bronchial or arterial tissues and create PH. 16 Tumors known to cause compressive/obstructive compromise of the PA’s include benign and malignant tumors such as sarcoma, carcinoma, germ cell tumors, lymphoma, and teratoma which can also lead to PH.16,17
Tumors that can make their way into the circulation can also embolize into the pulmonary arterial system known as pulmonary tumor embolism (PTE) and has been described since the 1930’s as a cause of right heart failure in cancer patients. 3 Cancers noted to be associated with PTE are carcinomas but in pediatrics have also included osteosarcoma,18,19 and Wilms tumor. 20 Some autopsy studies estimate the prevalence of PTE to be 3% to 26% of patients with these tumors and symptoms do not necessarily correlate with tumor burden. 20
In close association with PTE is pulmonary tumor thrombotic microangiopathy (PTTM) which is likely a continuing spectrum of PTE and was recognized for its unique features in 1990. 3 Like PTE, tumor presence in small artery/arterioles occurs but there is additional fibrocellular intimal proliferation. 21 The tumor cells themselves may be capable of creating endothelial cell damage of the arteriole intimal layer which leads to activation of coagulation cascade and growth factor release. 3 These changes are very similar to those of idiopathic PAH. 22
Symptoms associated with direct tumor affects depend upon the proximity of the tumor load to the central pulmonary arteries with more proximal masses creating more acute symptoms. 20 Severe acute right heart failure can occur in 15% to 20% of PTE patients mimicking thrombotic pulmonary embolism (PE), but asymptomatic patients have also been found to have PTE on autopsy.18,20 PTTM tends to present with more progressive dyspnea (87%) and cough (67%) 21 but the condition can quickly thereafter lead to right heart failure and death.21,23 PTTM itself carries a poor prognosis and can be rapidly fatal once symptoms begin.21,23 When PTTM is discovered before death, initiation of chemotherapy may improve survival by several months. 21
Chest x-ray (CXR) can sometimes demonstrate possible oncologic involvement such as cystic lesions, well circumscribed nodules, and multiple calcified lesions as well as chest wall abnormalities such as fusiform expansion of the ribs (eg, Ewing sarcoma).24-26 If CXR is concerning for tumor involvement the next step is chest CT, particularly multidetector CT with angiography to look at central and peripheral lung fields, pulmonary vasculature and airways. 24 Echocardiography is the primary method of diagnosing intracardiac tumors, but can also see proximal intra and extra-pulmonary artery tumors as well look for evidence of PH. 27 The diagnosis of microvascular PTE or PTTM requires a very high index of suspicion antemortem and is often missed. Chest CT and V/Q scan are very non-specific in both conditions.20,21 Pulmonary wedge aspiration via cardiac catheterization can be attempted to withdraw distal tumor cells. 3 A positive CT for any tumor concerns will prompt a biopsy plus or minus bronchoscopy for definitive diagnosis.21,24
Indirect Injury Mechanism of Cancer Associated PH
Cancerous cells can create circumstances that promote elevation of PA pressures as a result of the body’s physiological reaction to the cancer. Two such reactions are thromboembolism and inflammation.
Cancer is a prothrombotic state and a risk factor for venous thromboembolism (VTE) (deep venous thrombosis or PE). 6 The prevalence of VTE in cancer patients is estimated to be 7 times greater compared to the general population. 28 The pathophysiology of hypercoagulation due to cancer is complex. These events together with the release of tumor microparticles will create activation of the clotting cascade and thrombus formation. 28
Cancer patients have twice the risk of developing PH in the setting of any VTE which is increased to 3-fold with PE alone. 6 However not all PE’s found in cancer patients are symptomatic with asymptomatic incidental PE’s found in 4% of adult 29 and 2% of pediatric cancer patients.29-31 Additional risk factors for pediatric PE include coagulation disorders, prior VTE 31 major surgery, central line placement 32 and use of chemotherapy. 30 The “gold standard” for PE diagnosis remains pulmonary angiography, but this has mainly been replaced with CT angiography. 33 V/Q scans can still be used but limited due to frequent low or intermediate probability risk scans. 33 Newer techniques exist for MRI analysis which avoids radiation exposure and can evaluate cardiac function. 33 PE’s that are high risk with cardiovascular compromise may require treatment with thrombolytic or surgical intervention in addition to anticoagulation vs anticoagulation alone in lower risk patients but no set guidelines exist in pediatrics. 33
Cancer and PH both have a prominent inflammatory response that is integral to their pathogenesis.34-37 The innate and adaptive cellular immune response as well as certain cytokines and inflammatory pathways are found to be exaggerated in both conditions.35,36 Macrophages can comprise up to 50% of a tumor’s mass and are known as “tumor-associated macrophages”. 35 These macrophages promote aberrant cell proliferation and increase tumor metastasis through the secretion of cytokines (eg, interleukin (IL)-1RA, IL-8, and granulocyte-macrophage colony stimulating factor.35,37 In PH macrophages are found in pathologic PH arterioles as perivascular infiltration and in occlusive vascular outgrowth vessels called plexiform lesions. 38 T cell proliferation (increase T-helper) but decrease T-regulatory cells is noted to occur in tissue associated with tumor.35,37 In PH T-helper cell types 1 and 17 are involved in vascular remodeling through the secretion of IL-6 and TNF-α likely as a result of immune dysregulation. 38 These inflammatory cells secrete cytokines and chemokines that further propagate inflammation in PH and cancer.35,36,38 Cytokines IL-1B, IL-6, and TNF-α are elevated in PH patients and are associated vascular remodeling. 36 In cancer, these cytokines among others also promote activation of proinflammatory pathways and stimulate cell survival and proliferation as well as angiogenesis. 35 Proinflammatory pathways nuclear factor-κB (NF-κB) and STAT 3 serve to increase local cytokines and promote cell proliferation and survival of tumor cells. 35 In PH the transforming growth factor beta (TGF-β) pathway regulates multiple cellular functions and mutations in the bone morphogenic protein receptor (BMPR) II of this pathway have been found in particular types of PH. 36 Mutations of BMPR II are found in 70% to 80% of inherited PAH and 10% to 20% of sporadic IPAH which affects cell growth and apoptosis. 39 TGF-β pathway is also involved in aberrant immune cell response in tumors while BMPR II mutations promote migration of tumor cells. 40 With strong similarities between cancer and PH in their type of inflammatory response, there is evidence that tumor microenvironment inflammatory process directly promotes vascular changes which elevates PA pressures consistent with PH pathogenesis. 37 Targeting inflammation in cancer such as with IL-6 inhibitor toclizumab may provide novel cancer therapy and possibly also target these PH changes. 35
Pulmonary Hypertension Associated with Cancer Therapy
Hematopoietic stem cell transplant (HSCT) is a life-saving advanced cancer therapy used to treat leukemia, lymphoma and certain tumors. 41 A severe small vessel angiopathy called transplant-associated thrombotic microangiopathy (TA-TMA) can occur as a complication of HSCT in 10% to 35% of patients.42,43 Patients can present with difficult to control hypertension, hypoxia, respiratory failure, heart failure and shock.42-44 PH was found in one cohort in 25% of HSCT patients with respiratory failure. 43 TA-TMA is an endothelial damage syndrome that creates a cascade of procoagulation, hemolytic anemia, thrombocytopenia, and compliment consumption. 42 Histologically, TA-TMA can resemble severe PAH with cellular proliferation and lumen occlusion. 44 There are risk factors for developing TA-TMA which include genetic variants in compliment genes, African American race, female gender, allogeneic HSCT, use of cyclosporine or tacrolimus and prior HSCT. 42 TA-TMA has a high mortality rate of around 44% by 1 year of diagnosis. 43 Addressing PH with TA-TMA first involves treating the TA-TMA. Management of TA-TMA includes managing hypertension, use of compliment blockage, stopping calcineurin inhibitors, managing graft vs host disease and addressing infections. 42 Because of the concern for concomitant PH in the setting of TA-TMA, echocardiographic screening has been proposed every 1 to 2 weeks especially in the setting of severe symptoms of heart or respiratory failure. 44
Several chemotherapeutic agents have the potential to cause PH by their effects on the lungs and heart (Table 4). Survivors of childhood cancer have a 5% incidence of pulmonary adverse events 45 resulting in a 9.2 fold increase in risk of death from lung disease in pediatric cancer survivors. 46 Pulmonary toxicity from chemotherapy can occur at various timeframes with early toxicity resulting in interstitial lung disease to late onset pulmonary fibrosis up to 15 years after treatment. 46 Key offenders include bleomycin which is an antibiotic antitumor medication known to cause interstitial lung disease by free radical creation. 45 Pulmonary vascular injury can be directly induced by tyrosine kinase inhibitors (TKI) with dasatanib most commonly associated with PH and has been labeled by the 6th World Symposium on Pulmonary Hypertension as a definite cause of toxin induced PH.11,47
Chemotherapy Drugs/Radiation Therapy Associated with PH and their Targeted Method.

Strongly associated. **likely associated. *possibly associated.
Cardiotoxicity from chemotherapy can place a patient at risk for developing PH secondary to cardiac dysfunction. Acute cardiotoxicity with heart failure symptoms can occur with treatment in about 1.6% of pediatric patients and delayed onset of heart failure can occur in 2.5% of children treated several years afterward anthracycline usage. 48 However up to 57% of pediatric patients have asymptomatic ventricular dysfunction. 49 Risk factors for cardiotoxicity include use of anthracyclines at a young age (<4 years), high cumulative dose for age (≥200 mg/m2 for age <1 year and ≥300 mg/m2 for >1 year old), concurrent use of radiation therapy, and female gender.48,49 Cardiotoxicity manifests as left or right ventricular systolic or diastolic dysfunction. 50 Because of the risk of cardiotoxicity, echocardiography is obtained frequently with treatment and every 5 years after low dose anthracycline ± radiation therapy (<15 gray) exposure and every 2 years with high dose (>250 mg/m2) or whenever a higher dose (>15 gray) of radiation therapy is used. 51 Cardiomyopathy, including that associated with doxorubicin, is associated with increasing mortality for each 5mmHg increase in mean pulmonary artery pressure. 52
Radiation therapy can significantly affect pulmonary and cardiac function. It has been shown that survivors of pediatric cancer and radiation therapy have a 3.5% incidence of pulmonary fibrosis over a 20 year period and continue to increase beyond 25 years after therapy. 45 A St. Jude Hospital study of adult survivors of pediatric cancer showed that those who received chest radiation therapy had echo estimated abnormal PA pressures in 25% with an odds ratio (OR) of 2 versus those who did not. 53 The prevalence of elevated PA pressures increase at higher radiation doses with >30 gray having about 31% (OR 4.5) showing possible PH. 53
HSCT, Chemotherapy and radiation therapy share a common consequence of cancer treatment known as pulmonary veno-occlusive disease (PVOD) which is part of group I PH classification. PVOD can be found in all ages of cancer patients as a result of therapy. 54 This rare condition can be more prevalent with certain chemotherapy regimens (Table 3) 55 while some studies have shown up to 34% of HSCT patients had pathological evidence of PVOD on autopsy. 56
PVOD results from endothelial cell inflammatory and procoagulant changes that affect the pulmonary venules and small veins. 54 This creates vascular smooth muscle layer hyperplasia, intimal fibrosis with eventual luminal occlusion. 55 Vascular changes consistent with PAH also occur in the pulmonary arterioles making PVOD unique in that it affects all 3 pulmonary vascular compartments. 55
Clinically PVOD demonstrates poor diffusion capacity in pulmonary function tests, CXR with vasculature congestion, bilateral infiltrates and pleural effusions while a chest CT may demonstrate the lungs with ground glass opacities, septal thickening, pleural effusions, and mediastinal lymph nodes.54,55 Definitive diagnosis would require a lung biopsy. 55 However patients are very likely (“highly probable”) to have PVOD if they have evidence of precapillary PH (consistent with PAH) on cardiac catheterization along with typical radiographic abnormalities. 57 A patient can be considered to have “probable” PVOD without catheterization data with all the other clinical and imaging findings. 57
PVOD carries a poor prognosis due to PH with the 1 and 3 year lung transplant-free survival of 75% and 34%, respectively. 55 There are no good treatments for PVOD and therapy is mainly supportive. Preventative therapy with amifostine, a cytoprotective free radical scavenger, can be given when patients are on cyclophosphamide or mitomycin-C.3,57 Lastly lung transplant would be the only definitive therapy if a patient was a surgical candidate.54,55
Evaluation and Management of PH in the Cancer Patient
In the cancer patient, suspicion of possible PH can occur as a result of a surveillance echocardiogram with findings consistent with PH or with a change in clinical status showing symptoms of possible PH. The most common symptoms for pediatric PH include dyspnea on exertion (65%), fatigue (41%), syncope (20%), chest pain (11%), and dizziness (7%). 8 Routine screening may be necessary to identify subclinical PH in asymptomatic at risk patients. Guidelines exist for the diagnosis and management of pediatric PH that should be applied.10,58 The goal of the evaluation is to make a diagnosis of PH, find modifiable causes and to see if pulmonary vasodilators (PVDs) are appropriate. To treat patients with PVDs, they need to meet criteria for PAH with the exception of PVOD. 59 PVDs are generally contraindicated for group 2 classification. 10 Patients who fit closer into group 3 might benefit from PVD if they are not improved with addressing lung disease.60,61 Patients that meet criteria for group 4 or 5 classification should have the underlying issue addressed. An echocardiogram is going to be the initial screening test and this may lead to more advanced testing such as chest CTA to look for parenchyma or pulmonary vessel disease such as tumor metastasis, infection or PE (Figure 1). If there is obvious and addressable etiology for suspected PH then a trial of targeted therapy of the condition maybe reasonable. Re-evaluation by echocardiogram in a short interval (eg, after antibiotics for infection or steroids for inflammation) is needed especially if the patient is worsening. The gold standard for PH diagnosis still remains cardiac catheterization in order to confirm and classify the type of PH present.10,58 However it is also understandable that a cancer patient may not be stable enough to undergo a cardiac catheterization or other testing procedures. 58
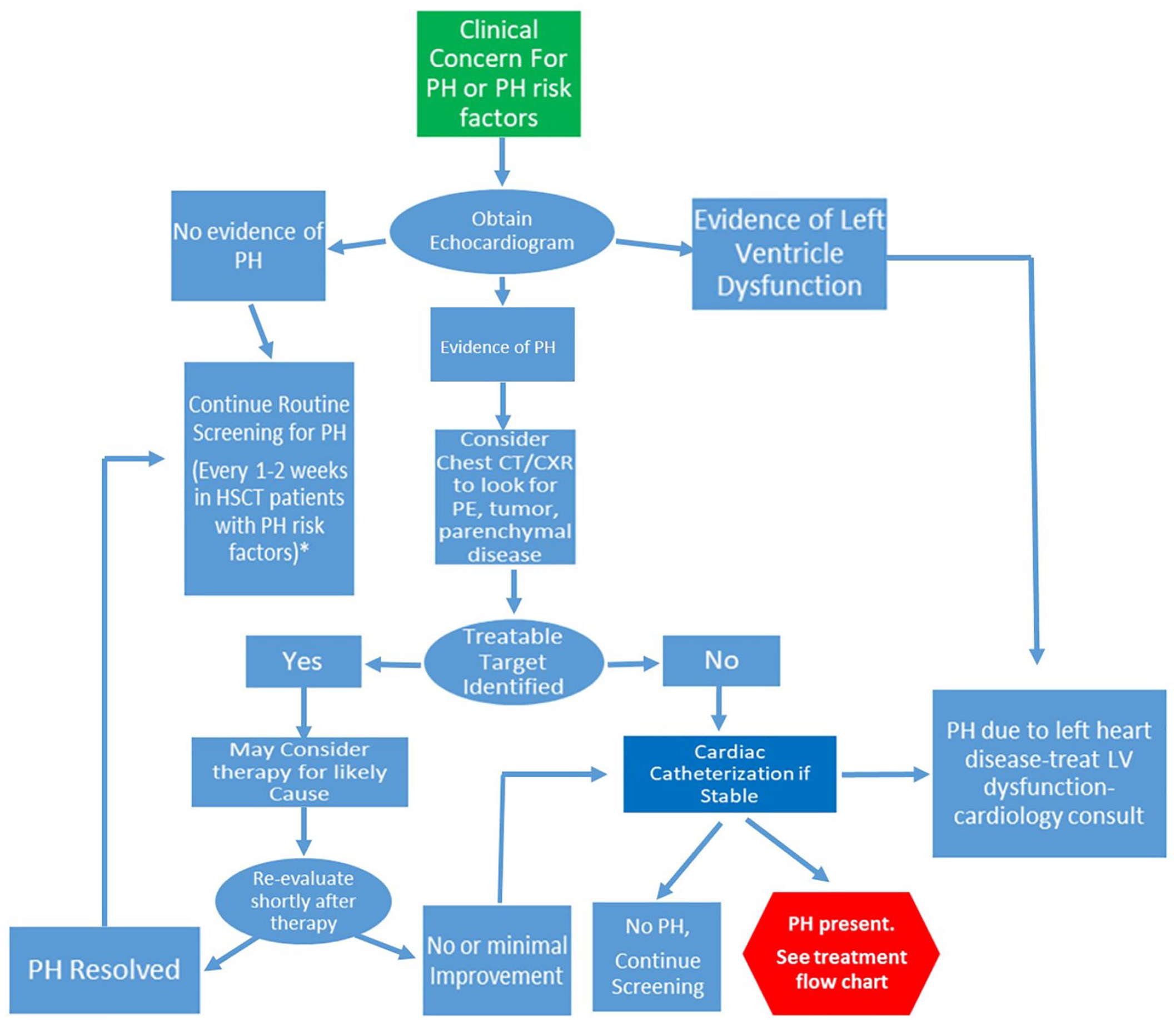
Pulmonary hypertension diagnosis. Algorithm is modified based upon 2019 guidelines 10 to identify etiology and modifiable factors.
Once PAH is confirmed or highly suspected and deemed appropriate to treat with drug therapy, assessment and drug choice should occur with the guidance of a pediatric PH expert as there are currently several drugs with various side-effects/drug interactions and technical aspects that need close monitoring and follow up (Figure 2). Non-PVD drugs are often a part of the management to off load the right ventricle and help improve oxygenation such as diuretics and supplemental oxygen. In certain cases surgical/interventional therapy may be needed if there is continued low cardiac output due to refractory PH. Continuous reassessment and medication adjustment as needed is key to managing PH therapy. Large studies demonstrating outcomes with PH therapy in cancer are not available.

Pulmonary hypertension treatment with pulmonary vasodilators algorithm based upon 2019 guidelines. 10
Conclusion
Pulmonary hypertension is multi-factorial progressive condition that has a high mortality rate. Its occurrence as a result of cancer burden and therapy has been identified but not well studied in pediatrics. Cancer associated PH is unique in that it can be classified into any one or multiple PH subcategories depending upon its presentation. Recognizing symptoms and risks for developing PH can lead to earlier screening by echocardiography to begin prompt treatment. Understanding the pathogenesis of PH in cancer is key to direct treatment to underlying causes such as obstruction, inflammation, drug toxicity or microvascular changes. PH diagnosis should follow established algorithms tailored for cancer patients in order to make sure therapy, including pulmonary vasodilators are appropriate. Because of the disease complexity, management of PH is best under the guidance of a pediatric PH expert.
Footnotes
Authors’ Note
Ethical Approval and Informed Consent statement: This manuscript was created from literature searches in Ovid, Pubmed, Google Scholar and other databases. No human subjects were used for this manuscript and no personal health information was accessed. Therefore ethics approval was not required for the creation of this manuscript.
Author Contributions
Author wrote and revised the entire article.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
