Abstract
Objectives. Using hair nicotine as the gold standard, this study aimed to establish cutoff points and validate the questionnaire-based environmental tobacco smoke (ETS) exposure and ETS statuses of Indonesian infants. Methods. A cross-sectional study design was conducted among families who were participants of the Peer Health Cohort Study in Jakarta, Indonesia. Households with 6-month-old infants joined this study. The presence and amount of ETS exposure were assessed by both questionnaire and hair sampling for nicotine determination. Head hair samples were collected from 102 infants and measured by optimized gas chromatography-mass spectrometry (GC/MS). Infants were grouped as ETS-exposed if they lived with at least 1 smoker at home. We used the receiver operating characteristic (ROC) curve to assess the sensitivity and specificity of cutoff values of hair nicotine. Results. There were 78 (76.5%) infants exposed to ETS based on the questionnaire. The nicotine concentrations in hair were significantly higher in infants with ETS exposure than in those without ETS exposure (P < .001). The area under the curve for nicotine was 0.774. A hair nicotine cutoff value of 2.37 ng/mg, with a sensitivity of 67.95% and specificity of 83.33%, was identified as the optimal cutoff value for separating exposed from non-exposed to ETS in infants. Conclusion. The hair nicotine value of infants aged 6 months is useful in confirming the questionnaire on smoking in the household and exposure to ETS. Moreover, it also could be used to distinguish ETS-exposed from non-ETS-exposed infants.
Introduction
A significant proportion of children in the low and middle-income countries remain exposed to environmental tobacco smoke (ETS). Based on a study conducted by Mbulo et al., 1 around 84.6% of the children exposed to ETS in China, India, Bangladesh, Indonesia and the Philippines. Indonesia has one of the highest levels of smoking prevalence globally with 89.6 million smokers.2,3 WHO recommendation has been implemented to reduce tobacco consumption in Indonesia, however none of them are yet at the top level of achievement. 3 The occurrence of ETS exposure was higher in countries with higher adult smoking percentages. 1 Worldwide, 1.2 million deaths have been attributed to ETS exposure. Lower respiratory tract infection, otitis media, asthma, sudden infant death syndrome, low birth weight, and adverse growth outcome in children were more common health problems in infants or children associated to ETS.4-6
Due to various highly adverse effects of ETS in infants, it is important to observe the extent of exposure. Questionnaires have usually been used to assess ETS exposure with many concerns about validity, especially underreporting. Some researchers have suggested explanations, including parental denial of exposure, which often regarded as socially unacceptable, inaccurate recall and misclassification of exposure. Biomarkers offer an objective method of determining tobacco smoke exposure since relying on self-report and the numerous biases it introduces may lead to inaccurate measures of the exposure.7-12
Hair nicotine as a biomarker for ETS has several advantages for the study of infants, including the noninvasive nature of sample collection, inexpensive to store and the ease of transport. Moreover, Hair nicotine assures to be a valid and reliable measure of longer-term exposure to tobacco smoke, particularly ETS.13-15 Each 1 cm of hair potentially represents exposure to ETS over 1 month. 16 However, data about the association between hair nicotine and ETS exposure from infants are still limited, especially in 6-month-old infants.
Studies on the correlation of questionnaire information on ETS exposure with hair nicotine concentrations in infants were still limited. Furthermore, the available cutoff values are commonly studied in developed countries setting. Therefore, it is important to specify the appropriate cutoff in developing countries setting such as in Indonesia. Using hair nicotine as the gold standard, the current study contributes to existing knowledge by establishing cutoff points and validating the questionnaire-based ETS exposure and ETS statuses of Indonesian infants.
Methods
Recruitment
A cross-sectional study design was conducted among families who were participants of the PEER Health Cohort Study in Jakarta, Indonesia. Detailed descriptions of PEER Health Cohort Study are available in the previous publication. 17 Briefly, Participants admitted to 7 community health centers in Jakarta were recruited during 2017 to 2019. To be eligible for the study, infants had to be at 6 months of age, have enough hair for hair sampling analysis, at least 1 parent (mother and/or father) had to be available for an interview for the study and willing to sign an informed consent.
Assessment of ETS Exposure
The presence of ETS exposure were evaluated by both questionnaire and hair sampling for nicotine determination. The questionnaire was prepared by PEER Health international team of experts then pilot tested with 10 mothers resulting in no changes. A smoker was defined as a person who are currently smoking at least 1 cigarette per day.15,18 As the definition of ETS includes secondhand and thirdhand smoke, 19 infants grouped as ETS exposed if they lived with at least 1 smoker at home.15,20 The following questions were used to assess cigarette smoking status and exposure: (a) whether the mother smoked, (b) whether the father smoked, (c) whether other family members smoked (d) number of cigarettes smoked daily by mother/father/other family member(s), and (e) frequency of father smoking at home.
Measurement of Hair Nicotine
Head hair samples were collected from 102 infants. Approximately 20 to 40 hair shafts were cut as close as possible to the scalp from each infant and placed in separate, clean plastic bags for storage and later sent for assay at Regional Health Laboratory of DKI Jakarta Province, Jakarta, Indonesia. The formulation of reagents and analysis of hair nicotine was accomplished by the laboratory technician. In the laboratory, each hair sample was cut to exclude hair that was more than 3 cm from the root end which represented the exposure to cigarette smoke for the past 3 months. Since it had been presumed that 1 cm of hair from the scalp would represent last month’s tobacco smoke exposure. 13 Then, the samples were minced into 1 to 2 mm pieces using scissors and weighed approximately 50 mg. Before being extracted with 2.5 ml TBME (Tert-Butyl Methyl Ether), the hair was put in a tube and incubated with 1 mL of 1 M KOH and 10 µl of internal standard DPA (diphenylamine) 40 ppm for 12 hours at 37°C. Hair nicotine analysis was performed using gas chromatography/mass spectrometry (GC Agilent Technologies 7890B/MS 5977B) in selected ion monitoring (SIM) and splitless modes.
For quality control, series of nicotine standards with various concentrations (5, 10, 25, 50, 75, 100, 150 ng/mg) were used. Quantification was achieved by integration of the ion chromatograms and constructing 7-point standard curves of response (Peak area of nicotine height) versus concentration, by linear regression. The concentration of nicotine in hair was calculated by dividing the quantity of nicotine detected in hair (ng) by the mass of hair investigated (mg). The limit of detection (LOD) was 0.2 ng/mg and the limit of quantitation (LOQ) was 0.8 ng/mg for a 50-mg hair sample. The quantifying ions used were m/z 84 for nicotine and m/z 169 for the internal standard. A more comprehensive description of the analytical method can be found elsewhere. 21
Statistical Analysis
Hair samples with “undetectable” levels of nicotine were assumed to have 0.05 ng nicotine/mg hair before the statistical analysis. 22 There were 18 (17.1%) Infants with “undetectable” nicotine in hair. The association between biomarker value, questionnaire-based ETS history in infants, characteristics, and households smoking behavior were assessed using the Mann–Whitney and Kruskal–Wallis tests. Whereas the comparison of characteristics between ETS-exposed and -unexposed groups was measured by Chi-Square test. We used the receiver operating characteristic (ROC) curve to assess the sensitivity and specificity of cutoff values of hair nicotine. The optimal cutoff values to differentiate infants with ETS exposure from non-ETS exposure, were obtained by locating the points with maximum sensitivity and specificity on the curve. A P value <.05 was considered significant.
Ethical Approval and Informed Consent
Informed consent was obtained from all parents of the participating infants. This study has been approved by the ethics committee of the Faculty of Medicine, Universitas Indonesia, with the code of ethics No:895/UN2.F1/ETIK/2015.
Results
Nicotine levels in the infant’s hair ranged from undetectable (0.05 ng/mg hair) to 73.07 ng/mg hair (data not shown). Characteristics of the study population and the distribution of reported smoking in the household by hair nicotine biomarker obtained from the 102 participants are summarized in Table 1. The mean of nicotine concentration was nearly equal based on gender and number of sibling(s). Both father and mother were mostly having a middle education level with percentages of 76.5% and 75.5%, respectively. The education level of parents and family income were not associated with infant hair nicotine. However, there is a tendency that parents with a lower levels of education and lower income had infant with higher hair nicotine level.
Results (Mean ± Standard Deviation) for Hair Nicotine of Questionnaire-based ETS History in Infants, Characteristics, and Households Smoking Behavior (N = 102).

Mann–Whitney test; bKruskal–Wallis test; significant at P < .05.
Based on the questionnaire, most of the infants lived with at least 1 smoker at home (ETS exposure group) (76.5%) and significantly associated with higher mean of hair nicotine concentration compared to non-ETS exposure group (9.96 ± 16.61 ng/mg vs 1.6 ± 1.86 ng/mg; P < .001). There was also a correlation between father smoking status, father smoking frequency at home, and the number of cigarettes smoked daily by all household member(s) and hair nicotine concentrations.
Table 2 shows the comparison of characteristics between ETS-exposed and -unexposed groups. Compared to infants with ETS exposure, infants unexposed with ETS had significantly higher levels of father education (P = .003) and family income status (P = .043). There were no significant differences in gender, number of children and mother’s educational level between infants with and without ETS.
Comparison of Characteristics Between ETS-Exposed and -Unexposed Groups (N = 102).

chi-square test; significant at P < .05.
ROC curves to validate questionnaire-based ETS history in infants are demonstrated in Figure 1. Before performing the ROC analysis, infants were classified as ETS exposure and non-ETS exposure groups based on the questionnaire. The ROC curve yielded an area under the curve of 0.774 (CI 0.677-0.871; P < .001). There is a 77.4% chance that the model will be able to distinguish between infants exposed to ETS and unexposed to ETS.
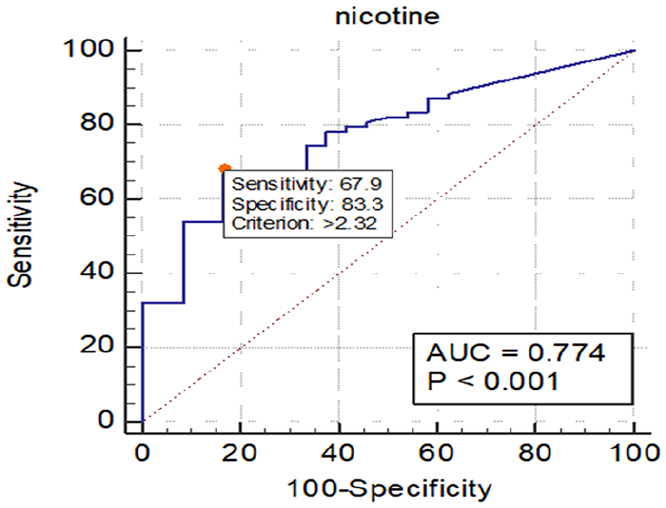
Receiver operated curve (ROC) for hair nicotine versus questionnaire based ETS history in infants (N = 102). Based on the area under the ROC curve (AUC), Youden Index cutoff value that maximized the sum of sensitivity and specificity was determined.
The nicotine cutoff concentration for differentiating exposed from unexposed ETS was 2.37 ng/mg (specificity = 83.33%, sensitivity = 67.95%) (see Table 3). Optimal cutoff values designated on the ROC curves are typically those that simultaneously maximize sensitivity and specificity (see Figure 1). 16.67 % of the infants who indicated that smoker(s) were absent at their home could be classified as being exposed to ETS based on the selected cutoff (2.37 ng/mg). Moreover, about 32% of the infants who indicated exposure to ETS had a hair nicotine concentration below 2.37 ng/mg.
Coordinates of the Receiver Operated Curve (ROC) for Various Cutoff Values of Hair Nicotine for Questionnaire-Based ETS History in Infants (N = 102).

Italic and bold values correspond to the optimal cutoff value.
Discussion
In this study, the measured biomarker nicotine in hair was significantly associated with data obtained from questionnaires (infants ETS exposure, smoking status of the father, frequency of father smoking at home, and the number of cigarettes smoked by all household member(s)), with mean of hair nicotine value 9.96 ng/mg for ETS exposed group; 10.65 ng/mg for infants with father smoker; 12.92 ng/mg for infants with daily/weekly frequency of father smoking at home; and 10.76 ng/mg for infants lived with family who smoked 6 cigarettes or more per day. Father is the main source of ETS exposure in infants and the highest mean of nicotine level is found in the infant lived with a father who consumed cigarettes daily/weekly at home. The results are consistent with previous studies in Indonesia that prevalence of father smoking reached over 70%.23,24
The ranges of nicotine values are in line with reported studies in infants whose both parents were smokers25,26 and infants with daily/weekly ETS exposure. 26 However, those are above the median or mean previously studies in children (not specific in infants) based on number of smokers in the house, 22 parent smoker(s) 27 and smoking inside/outside house. 28 Regarding this, 2 prior studies found that hair nicotine level was higher in younger children than older children.15,29 Potential causes might due to the presence of thirdhand smoke (THS), so that home with a smoker(s) inside indicates high-risk places for ETS exposure in infants, including both environments where active smoker is smoking and environments in which smoker has previously smoked. 30 Moreover, younger children are more vulnerable exposed to ETS due to the longer time they spend at home in the presence of a smoker.15,20
The current finding indicated that ETS-exposed infants had significantly lower levels of father education and family income status compared to ETS-unexposed infants. These results are in line with previous studies showed that low educational level of 1 or both parents were associated with ETS exposure in children20,31,32 and infants from low-income or low-socioeconomic status families were most likely to be exposed to ETS.31-33
Until now, there has no been a cutoff value for distinguishing between the exposed and unexposed ETS in infants. Other studies demonstrated hair nicotine values in infants but not included cutoff point. In our study, the hair nicotine cutoff value to differentiate the exposed from unexposed infants was 2.37 ng/mg, whereas similar studies in adults to distinguish active smokers from passive/non-smokers reported 2.77 ng/mg and 5.68 ng/mg.14,34 It should be highlighted that a cutoff point for ETS exposure in infants or children is less clear and more controversial than the cutoff for active tobacco use. Moreover, it is challenging to define a cutoff for ETS exposure, because of the diversity of exposure sources. 35
Our results provide a reasonable cutoff level to differentiate exposed from non-exposed to ETS in infants with a sensitivity of 67.95% and specificity of 83.33%. A study in passive smokers but different subject (pregnant women), showed the sensitivity of 63% and specificity of 71% for saliva cotinine cutoff value of 1.5 ng/mL. 35
With hair growth stated to be roughly 1 cm per month, 36 a 3 cm of hair from the scalp can possibly characterize tobacco smoke exposure over a 3-month period. 14 The current study showed that hair nicotine has the ability to identify true negative exposure (ETS-unexposed) group better than true positive (ETS-exposed) group. This is likely because ETS-unexposed group was determined from the self-reported non-smoking status of household with zero number of cigarettes smoked per day. It represents a constant behavior in long-term period as it is not an easy process to quit smoking. 37 Whilst ETS-exposed group was based on the self-reported smoking status of household with a daily number of cigarettes smoked that is possibly changes in short-term period. 38 Previous study found that hair nicotine is not as strongly correlated with self-reported recent smoking behavior (ie, daily cigarettes smoking) as salivary cotinine, which represents comparatively current exposure levels. 14
Our study observed misclassification among non-ETS and ETS exposure group. 32% of infants with nicotine concentrations under cutoff point were misclassified as ETS exposed group and 16.67% of infants with nicotine levels over cutoff point were misclassified as unexposed group. This result corresponds with the studies on Japanese pregnant women, 7 and on Aboriginal and Torres Strait Islander women. 9
Significant misclassification could be explained by some reasons. It might due to the definition of ETS exposure that based on the presence of smoker(s) in the household regardless of whether they smoked “indoor or outdoor”. It is possible that the households with smokers had a “smoke free environment” rule and negatively associated with infant ETS exposure. 39 On the other hand, some level of ETS exposure can be detected as the result of exposure from outside home such as in traditional markets, restaurants, public or private transportations and places. Alternatively, through THS exposure by hand-to-mouth activity, ETS exposure pathways may include not only ingesting floor dust, hands or foods containing nicotine but also touching contaminated surfaces (possibly including smoker’s clothing). 40 Perhaps, the substantial proportion of the population with detectable hair nicotine is reflective of the extensive ubiquity of ETS in our environment and the challenges of controlling exposure in several settings. Finally, underreporting of smoking status of family members can also play an important role in underestimating hair nicotine levels. 41
There were some limitations in our study. The cutoff value was acquired from a relatively small number of samples so the result should be interpreted with care as it is unlikely to be representative of the general population. 14 Moreover, information about ETS exposure in infants 6 months old was limited to the presence of smoker(s) in the household, father smoking frequency at home, and number of cigarettes smoked daily by the household member(s). However, the home was the most significant place of such exposure for infants. 42 In a review of 41 studies, parental smoking consistently associated with children’s ETS exposure in the home. 32 Dust and surfaces in smoker’s home were contaminated with ETS and infants were in close physical contact with parents or household member(s) who smoke. 43 Finally, Future research should consider to validate the questionnaire using different age group of children.
Conclusion
In summary, hair nicotine value of infants aged 6 months old is useful in confirming the questionnaire on smoking in household and exposure to ETS. Moreover, it also could be used to distinguish ETS exposed from non-ETS exposed infants.
Footnotes
Author Contributions
Conceptualization: KD. Acquisition, analysis, interpretation and drafted manuscript: SRN. Formal analysis: ARH, SRN. Critically revised manuscript: KD, DMU, ARH. Final approval: KD.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by LPDP (Indonesia Endowment Fund for Education) and Universitas Indonesia.
